Under identical conditions, aqueous NH2Cl and NHCl2
reacted with 2,4,6-trichlo-m-cresol (17) and 2,4,6-trichloro-
3-methoxyphenol (29) to yield analogous products, 2,6-
dichloro-3-methyl-1,4-benzoquinone-4-(N-chloro)imine (25)
(40%) and 2,6-dichloro-3-methoxy-1,4-benzoquinone-4-(N-
chloro)imine (28) (60%), respectively, and 2,6-dichloro-3-
methyl-1,4-benzoquione (24) (60%) and 2,6-dichloro-3-
methoxy-1,4-benzoquinone (27) (30%), respectively. There
were no minor products in the reaction with 17. The reaction
of 29 had about 10% minor products.
chlorine atoms in the NHCl2 molecule could interact with
the methyl group.
Chlorination of Monochlorinated Phenols with the
Chloram ines. The data in Figure 4 show significant differ-
ences in the position of attack for NH2Cl and NHCl2 toward
2-chlorophenol (7), with NH2Cl attacking primarily at the
4-position to give 9 (77%) and NHCl2 reacting slightly more
rapidly at the 6-position to yield 55% of 10. Similarly, the
results from the chlorination of the monochloro isomers (11,
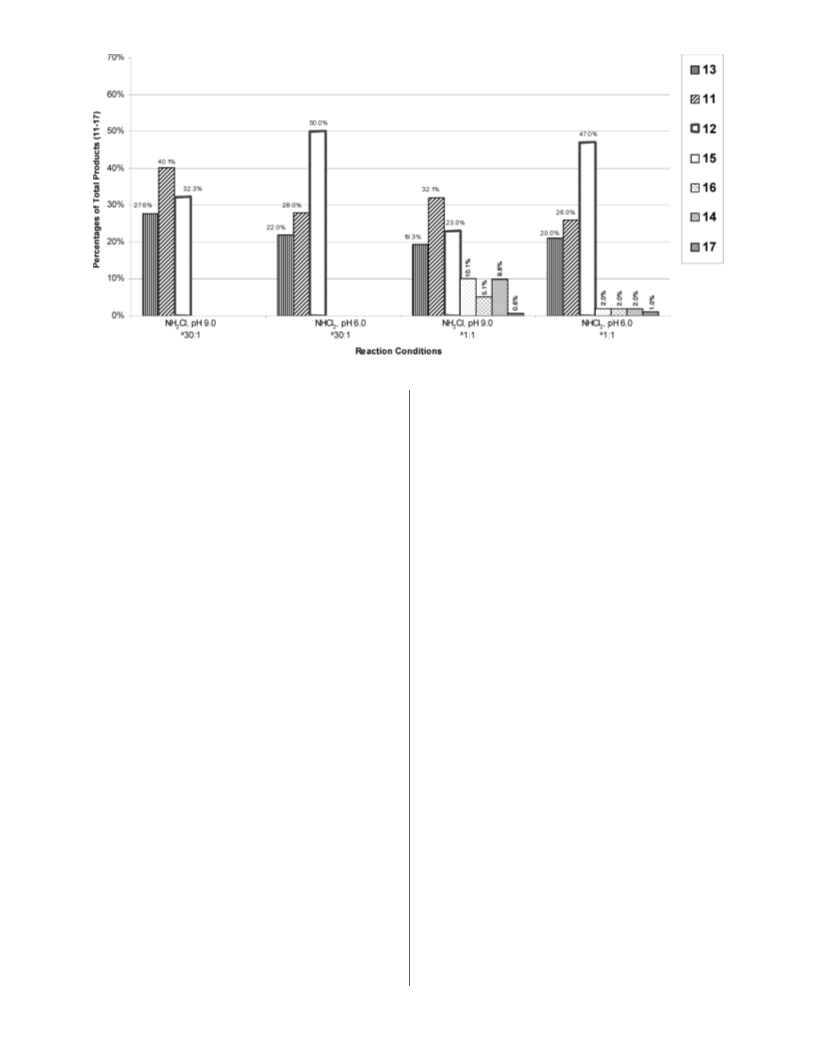
12, and 13) of 3 (Figure 5) indicate differences in the
reactivities of NH2Cl and NHCl2. The data in Figures 4 and
5 are in line with those in Figure 3 which indicate that the
substituted phenols show differences in the reactivities of
NH2Cl and NHCl2; phenol (2), an unsubstituted phenol, as
has already been stated did not show differences in reac-
tivities. These results could have important implications in
the reactions of the humic acids where phenolic substituents,
and perhaps other substituents, could show differences in
reactivities between the two chloramines.
Reaction of 2,4,6-Trichlorophenol (4) with NH2Cl and
NHCl2 in H2O/Glym e. To 0.3 g (1.5 mmol) of 4 dissolved in
15 mL of glyme (1,2-dimethoxyethane) in a flask equipped
with a stirrer was added 15 mL of 0.1 M NHCl2 (1.5 mmol)
at pH 6. Following extraction with dichloromethane as
described previously, GC analysis showed 5 (32%), 18 (20%),
and an additional product 2,4,4,6-tetrachloro-2,5-cyclohexa-
dienone 30 (48%) (39). When this reaction was conducted in
the presence of TEMPO, only 5 and 18 were formed.
A possible explanation for the differences in the positions
of attack by the chloramines on 7 (Figure 4) may arise from
NH2Cl, as an electrophile, reacting at the more electron rich
para-position in 7 (chlorine withdraws electrons from the
ortho(6-)-position), while the reactivity of NHCl2 is still
controlled by complexing with hydrogens with oxygen,
leading to ortho attack. The differences in the reactivities of
the chloramines with the monochlorocresols containing two
substituents (Figure 5), which show different results for NH2-
Cl and NHCl2, probably involve multiple complexities that
defy explanations.
Differences in the Reactivities of Phenols 2 and 3 with
the Chloram ines. The data in Table 1 show that m-cresol (3)
is more reactive than phenol (2) with both NH2Cl and NHCl2.
The greater reactivity of 3 is reasonable since the methyl
group increases the electron density of the aromatic ring
and, therefore, its reactivity toward the chloramines (chlorine
electrophiles).
Considerations on the Form ation of Indophenol (6)
During the Chlorination Process. Our data show that a small
amount of 6, probably present as its salt, was formed when
phenol (2) was chlorinated with NH2Cl at pH 13. (Indophenols
from the substituted phenols were also detected by UV
analysis at this pH, but their amounts were not determined.)
No 6 was detected when 2 and monochloramine reacted at
pH 9. The implications of these results are that 6 might be
present as a contaminant in drinking water, if 6 were formed
during the mixing process where the pH potentially rises
because of hydroxide ion (OH-) formation during the reaction
of ammonia (NH3) and hypochlorite ion to give the chlor-
amines.
Considerations of the Mechanism of the Displacem ent
of the pa ra -Chloride in 4 Resulting in the Form ation of 5
and 18. Direct displacement of the para-chloride in an
aromatic compound like 4 by strong nucleophiles such as
NH3 is without precedent in organic chemistry. Therefore
we were not surprised that aqueous NH3 did not react with
4 to give 2,6-dichloro-4-aminophenol, which is known to
react further with the chloramines to give 5 (32). Since NH2-
Cl is significantly less basic than NH3 (10-10 less basic) (40)
a direct, nucleophilic displacement of chloride (either SN2
or SN1) by the chloramines is inconceivable. We also
established that 5 was not converted directly to 18 under
any of our reaction conditions. Maeda et al. (29) did not
address the mechanism of the displacement of chloride in
4 leading to 5, but Pallagi, Toro, and Farkas (30) have clearly
established that para-substituted phenols (some substituted
with chloride in the para position) react with N-chloro imines
by either an ion radical or a radical mechanism to give
indophenols. They determined that a radical mechanism was
inhibited by TEMPO but that an ion radical reaction was
Results and Discussion
Reaction of Phenols 2 and 3 with the Chloram ines at 1:1
Molar Ratios. Figures 2 and 3 show that 2 and 3, in a 1:1
molar ratio with the chloramines (NH2Cl at pH 9, the mixture
of NH2Cl/ NHCl2 at pH 6.5 and NHCl2 at pH 6.0), have the
chlorinating capacity to chlorinate all the activated positions
(ortho and para) to the hydroxyl groups, producing mono-,
di-, and trichlorophenols leading ultimately to trichlorinated
phenols 4 and 17, respectively. We established that an excess
of NH2Cl and/ or NH2Cl with 2 and 3 converted all of the
chlorinated intermediates to trichlorophenols 4 and 17,
respectively. These data indicate that both NH2Cl and NHCl2
show extensive reactivity toward the phenols, even the
partially chlorinated less reactive intermediates would be
expected to fully chlorinate the activated positions in phenolic
substituents in the humic acids. As shown in Table 1, the
rates would be slow since the reactions of 2 and 3 with the
chloramines are not rapid. The concentrations of both the
humic acids and the chloramines would be low, but relatively
long reaction times would favor chlorination.
Reaction of Phenols 2 and 3 To Give Monochloro
Products. The data in Figure 2 give the ratios of monochloro
isomers that are formed under the conditions of (2):
chloramine of 30:1, respectively. Some of the data in this
figure deserves comment. With phenol (2), both NH2Cl (pH
9) and NHCl2 (pH6) and the mixtures of chloramines (pH
6.5) show the same ratio of isomers within experimental error,
with 2-chlorophenol (7) being the predominant product. This
result is surprising since 2 at pH 9 (NH2Cl), which exists in
a 1/ 0.14 ratio with its phenoxide ion (31), might be expected
to exhibit different orientation than chlorination at pH 6
(NHCl2) where molecular 2 is essentially the sole reactant
(ratio of 2 to 31 is 1:0.00014). Also the anions in the
intermediate ion-pairs following the electrophilic attack,
NH2- and NHCl-, are quite different from NH2Cl and NHCl2,
respectively, and the orientation in the products could have
been affected; apparently this is not the case. The predomi-
nance of the ortho isomer (7) over the para isomer (8) may
result from interactions (complex formation) between the
oxygen of 2 and the hydrogen of the chloramines, resulting
in greater delivery of the chlorine to the ortho position.
The data in Figure 3 indicate that m-cresol (3), in contrast
to 2, reacts differently with the two chlorinating agents
NH2Cl and NHCl2. NH2Cl and 3 continue to give nearly the
same amount of ortho product (11 and 13-68%) as was the
case with 2, but NHCl2 shows a significant increase in para
product (12). Perhaps this is the result of steric hindrance
in the formation of the complex between NHCl2, the oxygen,
and the hydrogen of the hydroxyl group, since the two large
9
VOL. 38, NO. 19, 2004 / ENVIRONMENTAL SCIENCE & TECHNOLOGY 5 0 2 7

 Heasley, Victor L.
Heasley, Victor L.
 Fisher, Audra M.
Fisher, Audra M.
 Herman, Erica E.
Herman, Erica E.
 Jacobsen, Faith E.
Jacobsen, Faith E.
 Miller, Evan W.
Miller, Evan W.
 Ramirez, Ashley M.
Ramirez, Ashley M.
 Royer, Nicole R.
Royer, Nicole R.
 Whisenand, Josh M.
Whisenand, Josh M.
 Zoetewey, David L.
Zoetewey, David L.
 Shellhamer, Dale F.
Shellhamer, Dale F.