8
M. Wu et al. / Food Chemistry 201 (2016) 7–13
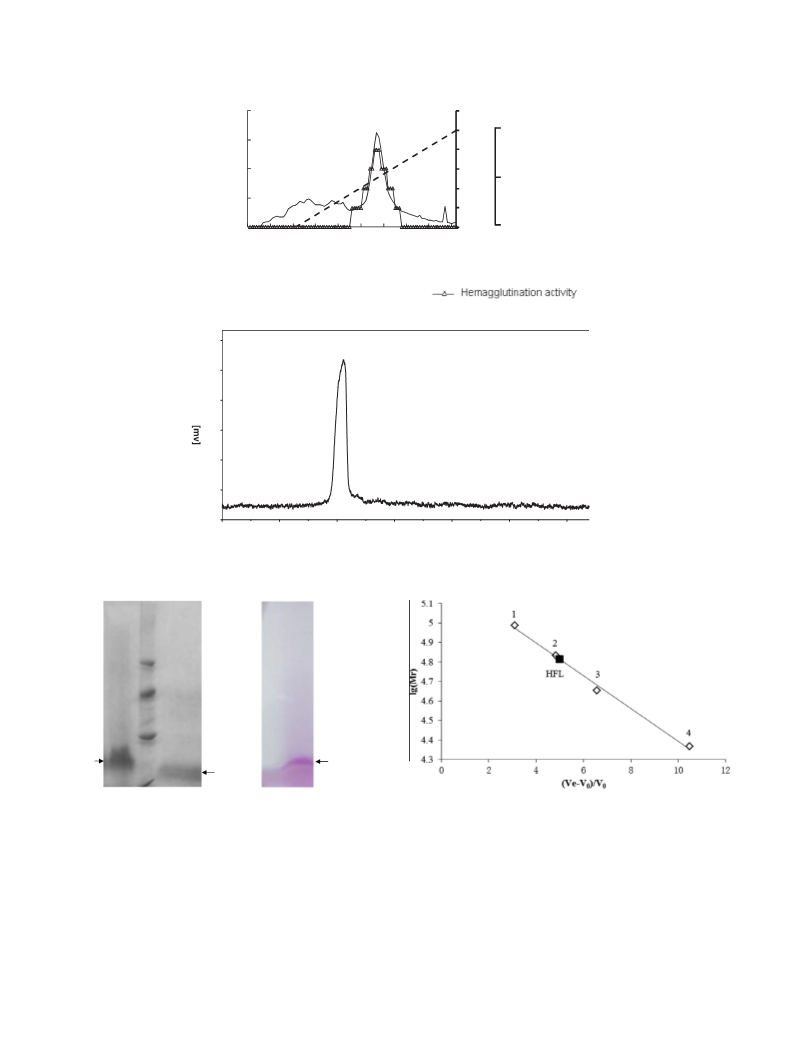
60 kDa (EPL-1) and 59.5 kDa (EPL-2), indicating that EPL-1 and
EPL-2 have a tendency to self-associate into higher order aggre-
gates (Ambrosio, Sanz, Sanchez, Wolfenstein-Todel, & Calvete,
2003). The lectin from the subspecies of the green marine alga,
Codium fragile (CFT), has a molecular mass of 60 kDa and is com-
posed of four 15 kDa subunits joined by disulfide bonds, moreover,
were eluted in TB, adsorbed proteins were eluted with a NaCl gra-
dient (0–1 M) in TB. The protein concentration and hemagglutina-
tion titer of each fraction were measured. Fractions containing
hemagglutinating activity were pooled and lyophilized to yield
powder (45 mg). The purified sample (50
lg) was suspended in
20 L of 0.01 M TB, pH 7.4, and centrifuged at 8000 rpm for
l
the lectin recognized the GalNAc
chains (Wu et al., 1997). The lectin that recognizes
a
1 ? Ser/Thr as carbohydrate side
20 min. The clear supernatant was also purified by high-
performance liquid chromatography (HPLC) on a G4000PWXL col-
umn (7.8 mm ꢀ 30 cm) and eluted with TB.
L
-fucose, fucoi-
dan, porcine stomach mucin and bovine submaxillary gland mucin
was purified from the green marine alga, Ulva lactuca. The molec-
ular weight determined by gel filtration was 8370 Da, and SDS–
PAGE gave a single protein band with a molecular mass of
17120 Da (Sampaio, Rogers, & Barwell, 1998b). Unfortunately,
there is limited data about lectins from brown algae in comparison
with those from red and green algae, and only a few publications
are available. Isolating protein from brown algae is difficult
because of the viscous polysaccharides (Kim, Kong, & Kim, 2010).
Several new compounds from the brown alga, H. fusiformis, have
been isolated and shown to have various bioactivities. Water-
soluble polysaccharides possessing antioxidant activities were iso-
lated from H. fusiformis (Wu, Wu, Qu, Li, & Yan, 2013), the protec-
tive effect of a glycoprotein from H. fusiformis on acetaminophen-
induced liver injury has been investigated (Hwang, Kim, & Nam,
2008), and immune-modulating activities of polysaccharides from
H. fusiformis were studied (Jeong, Jeong, Lee, & Kim, 2015).
Although new compounds have been found in brown algae, H. fusi-
formis, the search for novel lectin remains a challenge.
2.3. Measurement of the molecular mass of the purified native lectin
and the subunit
The molecular mass of the purified native lectin was measured
by passing it through a G4000PWXL column (7.8 mm ꢀ 30 cm) in
0.01 M TBS, pH 7.4 (Tong et al., 2012). The standard proteins used
were rabbit muscle phosphorylase B (97,000), bovine albumin V
(68,000), egg albumin (45,000), and trypsin (23,300).
The molecular mass of the subunit of the purified lectin was
measured by SDS–PAGE according to the Laemmli procedure using
a 15% polyacrylamide separation gel and a 4% polyacrylamide
stacking gel (Laemmli, 1970). The molecular mass of HFL was
determined by SDS–PAGE in the presence and absence of dithio-
threitol. Reduction of HFL was performed by heating at 100 °C for
5 min in sample buffer containing 2% SDS and 2.5% dithiothreitol
(Tong et al., 2012). Gels were calibrated using the following stan-
dard proteins: phosphorylase B (94,000), BSA (67,000), ovalbumin
(45,000), carbonic anhydrase (30,000), trypsin inhibitor (20,100),
and alpha-lactalbumin (14,400). Proteins and glycoproteins were
stained with Coomassie brilliant blue and glycoprotein stain
(ThermoÒ Pierce Glycoprotein Staining Kit), respectively.
In this report, we describe the purification, characterization,
carbohydrate specificity and antioxidant activity of a new Ca2+
-
independent thyroglobulin-binding lectin from the brown alga, H.
fusiformis.
Molecular masses were further investigated by MALDI-TOF
mass spectrometry. The measurements were performed on a Bru-
ker Autoflex time-of-flight mass spectrometer (Bruker, Bremen,
Germany), equipped with a delayed ion-extraction device and a
pulsed nitrogen laser (337 nm; 3 ns). An aqueous solution of lectin
(16 nmol/ml) was diluted 1:1 with aqueous 50% acetonitrile con-
taining sinapinic acid (11 mg/ml). One microliter of the mixture
was deposited on the target and left to air dry. The MALDI was
run in linear mode with an acceleration voltage of 25 kV and the
lag pulse set to 3,200 V. Cytochrome C and BSA were used for
external calibration. Typically, 50 laser shots were averaged per
spectrum.
2. Materials and methods
2.1. Materials
Monosaccharides were obtained from Merck (Darmstadt, Ger-
many). Porcine stomach mucin (PSM, type III), thyroglobulin from
bovine thyroid, 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,20-azi
no-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt
(ABTS) were purchased from Sigma Chemical (USA). Sheep ery-
throcytes were obtained from the Houxin Biotechnology Company
(Nanjing, China). Cellulose DE52 was obtained from the Solarbio
Science and Technology Company (Beijing, China). TSK gel
G4000PWXL columns were purchased from TOSOH (Japan). The
standard proteins used for apparent molecular mass estimation
by SDS–PAGE were purchased from the Beijing Solarbio Science
and Technology Company (Beijing, China). 3-Methyl-1-phenyl-2-
pyrazolin-5-one (PMP) was obtained from J and K Science Ltd. (Bei-
jing, China).
2.4. Hemagglutination assay
To assay hemagglutinating activity, HFL was 2-fold serially
diluted with 0.01 M TBS (25 ll) in microtiter U-plates. An equal
volume of a 2% suspension of sheep erythrocytes was added to
each well, and the mixture was agitated. The hemagglutination
was visually evaluated after 30 min (Belogortseva, Molchanova,
Kurika, Skobun, & Glazkova, 1998; Qu et al., 2015).
For the hemagglutination inhibition assay, the aqueous solu-
tions of various substances were 2-fold serially diluted with TBS.
The brown alga, H. fusiformis, was collected manually along the
coast of Zhoushan, East China Sea in May 2012. The sample was
cleaned and air-dried prior to extraction.
HFL (25
sion (25
l
l
l, 4 doses of agglutination) and a 2% erythrocyte suspen-
2.2. Isolation and purification of HFL
l) were successively added to each sample (25 l). The
l
mixture obtained was gently stirred by pipette and kept for 1 h.
The minimal concentration of each substance required for com-
plete inhibition was determined (Qu et al., 2015).
A 50 g portion of the algal powder was homogenized in the
same volume (w/v) of 0.9% NaCl. The homogenate was stirred for
16 h at 4 °C and centrifuged at 4000 rpm for 20 min. Crude saline
extract (50 ml) was dialyzed for 10 h against water at 4 °C and lyo-
philized to yield powder (870 mg). The powdered sample (100 mg)
was suspended in 2 ml of 0.01 M Tris–HCl buffer (TB), pH 7.4, and
centrifuged at 4000 rpm for 20 min. The clear supernatant was
applied to a cellulose DE52 column (3 ꢀ 11 cm) that had been pre-
viously equilibrated and eluted with TB. After unbound proteins
2.5. Effect of divalent cations
HFL was dialyzed for 24 h against 0.1 M TBS (pH 7.4), containing
50 mM EDTANa2 or 50 mM CaCl2. Sheep erythrocytes were used as
indicator cells.

 Wu, Mingjiang
Wu, Mingjiang
 Tong, Changqing
Tong, Changqing
 Wu, Yue
Wu, Yue
 Liu, Shuai
Liu, Shuai
 Li, Wei
Li, Wei