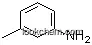
108-44-1Relevant articles and documents
Magnetically nano core–shell Fe3O4@Cu(OH)x: a highly efficient and reusable catalyst for rapid and green reduction of nitro compounds
Shokri, Zahra,Zeynizadeh, Behzad,Hosseini, Seyed Ali,Azizi, Behrooz
, p. 101 - 109 (2017)
Magnetically separable nano core–shell Fe3O4@Cu(OH)x with 22?% Cu content was prepared by the addition of sodium hydroxide to a mixture of CuCl2·2H2O and nano Fe3O4 in water. Characterization of the impregnated copper hydroxide was carried out by X-ray fluorescence (XRF), X-ray diffraction (XRD) atomic absorption spectroscopy (AAS), scanning electron microscopy (SEM), value stream mapping (VSM) and Brunauer–Emmett–Teller (BET) analysis. The core–shell nanocatalyst exhibited the excellent catalytic activity toward reduction of various nitro compounds to the corresponding amines with NaBH4. All reactions were carried out in H2O (55–60?°C) within 3–15?min to afford amines in high to excellent yields. Reusability of core–shell Cu(OH)x catalyst was examined 9?times without significant loss of its catalytic activity.
Studies on carcinogenic azo dyes. I. Synthesis of azo dyes labeled with tritium at the specific position
Baba,Mori,Iwao,Iwahara
, p. 1158 - 1162 (1969)
-
-
Strand,Kovacic
, p. 2977,2981 (1973)
-
-
Kovacic et al.
, p. 1262,1265 (1965)
-
Half-sandwich ruthenium complexes with Schiff base ligands bearing a hydroxyl group: Preparation, characterization and catalytic activities
Jia, Wei-Guo,Wang, Zhi-Bao,Zhi, Xue-Ting
, (2020)
Three half-sandwich ruthenium(II) complexes with hydroxyl group functionalized Schiff-base ligands [Ru(p-cymene)LCl] (2a-2c) have been synthesized and characterized. All ruthenium complexes were fully characterized by 1H and 13C NMR spectra, mass spectrometry and infrared spectrometry. The molecular structure of ruthenium complex 2c was confirmed by single-crystal X-ray diffraction methods. Furthermore, these half-sandwich ruthenium complexes were found to exhibit high catalytic activity for nitro compounds reduction using NaBH4 reducing agent in the presence of cetyltrimethylammonium bromide (CTAB) in water at room temperature.
Synthesis, characterization and catalytic activity of gold complexes with pyridine-based selone ligands
Zhang, Hai-Ning,Jia, Wei-Guo,Xu, Qiu-Tong,Ji, Chang-Chun
, p. 315 - 320 (2016)
Three neutral pyridine-based selone compounds, 2,6-bis(1-methylimidazole-2-selone)pyridine (Bmsp), 2,6-bis(1-ethylimidazole-2-selone)pyridine (Besp) and 2,6-bis(1-isopropylimidazole- 2-selone)pyridine (Bpsp) have been synthesized and characterized. Reactions of HAuCl4 with pyridine-based selone ligands result in the formation of the complexes [Au(L)Cl2]+[AuCl2]- (L = Bmsp (1); L = Besp (2) and L = Bpsp (3)), respectively. All compounds have been characterized by elemental analysis, NMR IR spectra and electrospray ionization mass spectroscopic (ESI-MS). The molecular structure of 2 has been determined by X-ray crystallography. Moreover, the gold complexes are efficiently catalyzed nitroarenes reduction to aromatic amines in the presence of sodium tetrahydroborate reducing agent in water.
Selective Encapsulation and Unusual Stabilization of cis-Isomers by a Spherical Polyaromatic Cavity
Yuasa, Mana,Sumida, Ryuki,Tanaka, Yuya,Yoshizawa, Michito
supporting information, (2022/02/02)
To explore new cavity functions, we herein employed cis-trans stereoisomers with a N=N, C=C, or C=N unit as guest indicators for a polyaromatic capsule. Thanks to the rigid, spherical cavity with a diameter of ~1 nm, azobenzene and stilbene derivatives ar
Selective Carbon-Carbon Bond Amination with Redox-Active Aminating Reagents: A Direct Approach to Anilines?
Qiu, Xu,Wang, Yachong,Su, Lingyu,Jin, Rui,Song, Song,Qin, Qixue,Li, Junhua,Zong, Baoning,Jiao, Ning
, p. 3011 - 3016 (2021/09/13)
Amines are among the most fundamental motifs in chemical synthesis, and the introduction of amine building blocks via selective C—C bond cleavage allows the construction of nitrogen compounds from simple hydrocarbons through direct skeleton modification. Herein, we report a novel method for the preparation of anilines from alkylarenes via Schmidt-type rearrangement using redox-active amination reagents, which are easily prepared from hydroxylamine. Primary amines and secondary amines were prepared from corresponding alkylarenes or benzyl alcohols under mild conditions. Good compatibility and valuable applications of the transformation were also displayed.
Synthesis of Substituted Anilines from Cyclohexanones Using Pd/C-Ethylene System and Its Application to Indole Synthesis
Maeda, Katsumi,Matsubara, Ryosuke,Hayashi, Masahiko
supporting information, p. 1530 - 1534 (2021/03/08)
The synthesis of anilines and indoles from cyclohexanones using a Pd/C-ethylene system is reported. A simple combination of NH4OAc and K2CO3 under nonaerobic conditions was found to be the most suitable to perform this reaction. Hydrogen transfer between cyclohexanone and ethylene generates the desired products. The reaction tolerates a variety of substitutions on the starting cyclohexanones.