5343-92-0Relevant articles and documents
Baeyer-villiger oxidation of acyl carrier protein-tethered thioester to acyl carrier protein-linked thiocarbonate catalyzed by a monooxygenase domain in FR901464 biosynthesis
Tang, Man-Cheng,He, Hai-Yan,Zhang, Feng,Tang, Gong-Li
, p. 444 - 447 (2013)
Baeyer-Villiger monooxygenases (BVMOs), generally catalyzing the transformation of carbonylic compounds into the corresponding esters or lactones known as Baeyer-Villiger oxidation in organic chemistry, are widely distributed among microorganisms and have stimulated great interest as biocatalysts for organic synthesis. The physiological roles of this type of MOs are usually classified as degradation of organic compounds involved in primary metabolism. Recently, increasing numbers of BVMOs have been found to be involved in the biosynthesis of secondary metabolites, especially for postmodification; however, to date, none of them has been reported functionally as a tailoring domain within polyketide synthase (PKS) acting on carrier protein-tethered substrates. FR901464, an antitumor natural product that targets spliceosome and inhibits both splicing and nuclear retention of pre-mRNA, was elucidated to be biosynthesized by a hybrid acyltransferase-less PKS/nonribosomal peptide synthetase (NRPS) system. Within the hybrid system, an unprecedented domain that was proposed to mediate the chain release process was located in the termination module. In this paper, we report the in vitro biochemical characterization of this domain to be a BVMO tailoring domain that catalyzes the BV oxidation of an acyl carrier protein (ACP)-tethered thioester to an ACP-linked thiocarbonate, which represents the first example of BVMOs operating in cis within the PKS and NRPS biosynthetic paradigm.
Unravelling the one-pot conversion of biomass-derived furfural and levulinic acid to 1,4-pentanediol catalysed by supported RANEY Ni-Sn alloy catalysts
Ansyah, Fathur Razi,Astuti, Maria Dewi,Hara, Takayoshi,Husain, Sadang,Mustikasari, Kamilia,Rodiansono,Shimazu, Shogo
, p. 241 - 250 (2022/01/19)
Bimetallic Ni-Sn alloys have been recognised as promising catalysts for the transformation of furanic compounds and their derivatives into valuable chemicals. Herein, we report the utilisation of a supported bimetallic RANEY nickel-tin alloy supported on aluminium hydroxide (RNi-Sn(x)/AlOH; x is Ni/Sn molar ratio) catalysts for the one-pot conversion of biomass-derived furfural and levulinic acid to 1,4-pentanediol (1,4-PeD). The as prepared RNi-Sn(1.4)/AlOH catalyst exhibited the highest yield of 1,4-PeD (78%). The reduction of RNi-Sn(x)/AlOH with H2 at 673-873 K for 1.5 h resulted in the formation of Ni-Sn alloy phases (e.g., Ni3Sn and Ni3Sn2) and caused the transformation of aluminium hydroxide (AlOH) to amorphous alumina (AA). The RNi-Sn(1.4)/AA 673 K/H2 catalyst contained a Ni3Sn2 alloy as the major phase, which exhibited the best yield of 1,4-PeD from furfural (87%) at 433 K, H2 3.0 MPa for 12 h and from levulinic acid (up to 90%) at 503 K, H2 4.0 MPa, for 12 h. Supported RANEY Ni-Sn(1.5)/AC and three types of supported Ni-Sn(1.5) alloy (e.g., Ni-Sn(1.5)/AC, Ni-Sn(1.5)/c-AlOH, and Ni-Sn(1.5)/γ-Al2O3) catalysts afforded high yields of 1,4-PeD (65-87%) both from furfural and levulinic acid under the optimised reaction conditions.
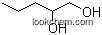
PROCESS OF PREPARATION OF 1,2-PENTANEDIOL FROM FURFURAL
-
Page/Page column 10-13, (2020/08/22)
The present invention relates to hydrogenolysis process for preparation of 1,2-pentanediol from furfural. In particular, the present invention provides a conversion of 1,2-pentanediol from furfural in presence of methanol and using a catalyst based on Rhodium on porous Manganese dioxide octahedral molecular sieve in a single step carried out at temperature range between 130 °C to 170 °C. The advantages of present invention is that it avoids formation of Intermediate-2 and directly give product 1,2-pentanediol with good selectivity over 1,5-pentanediol.