|
Name
|
|
EINECS
|
217-588-1
|
|
CAS No.
|
1897-45-6
|
Density
|
1.71g/cm3
|
|
PSA
|
47.58000
|
LogP
|
4.04356
|
|
Solubility
|
0.6-1.2 mg l-1 (25 °C)
|
Melting Point
|
250-251oC
|
|
Formula
|
C8Cl4N2
|
Boiling Point
|
350.5 °C at 760 mmHg
|
|
Molecular Weight
|
265.913
|
Flash Point
|
153.8 °C
|
|
Transport Information
|
UN 2811
|
Appearance
|
White crystal
|
|
Safety
|
28-36/37/39-45-60-61-36/37-26
|
Risk Codes
|
26-37-40-41-43-50/53-36-20/21/22-11
|
|
Molecular Structure
|
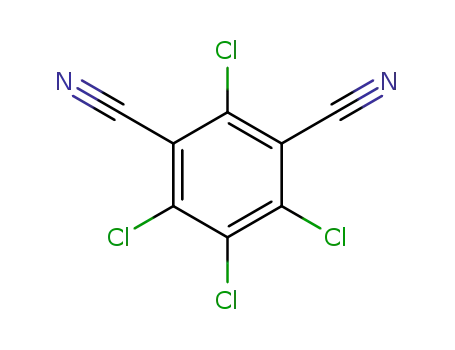
|
Hazard Symbols
|
 T+, T+, N, N, Xn, Xn, F F
|
|
Synonyms
|
Kabiguard 164SA;Nopcocide N 96;Nopcocide 54DB;Vanox;Bravo;Sweep;Dacosoil;Nopcocide;Bravo Ultrex;Termil;Daconil;Nuocide 960;Clortosip;Kavach;Bravo 6F;Thalonil;TPN (pesticide);Daconil M;1,3-Benzenedicarbonitrile,2,4,5,6-tetrachloro-;Daconil 2787;NexGen;Nopcocide N 54D;2,4,5,6-tetrachlorobenzene-1,3-dicarbonitrile;Chlorothalonil (Shandong dacheng pesticide co., ltd);
|
Article Data |
22
|
Chlorothalonil Synthetic route
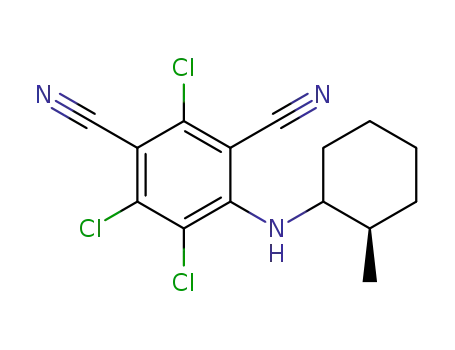
2,4,5-trichloro-6-((2R)-2-methylcyclohexylamino)isophthalonitrile
Conditions
| Conditions | Yield |
|---|
| In 1,4-dioxane at 20℃; for 0.25h; | 98% |
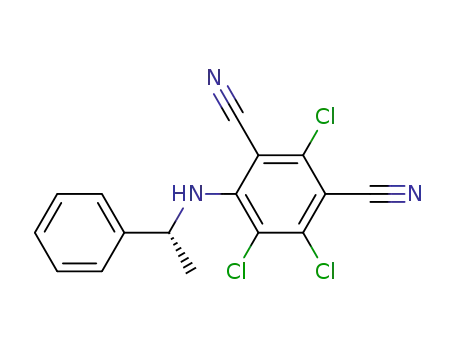
4-N-((R)-1'-phenylethylamino)-2,5,6-trichloro-1,3-dicyanobenzene
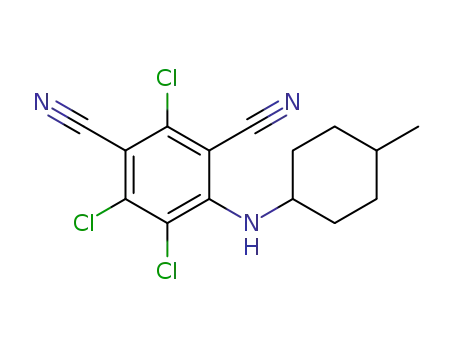
2,4,5-trichloro-6-((4-methylcyclohexyl)amine)isophthalonitrile
Conditions
| Conditions | Yield |
|---|
| In 1,4-dioxane at 20℃; for 0.25h; | 97% |
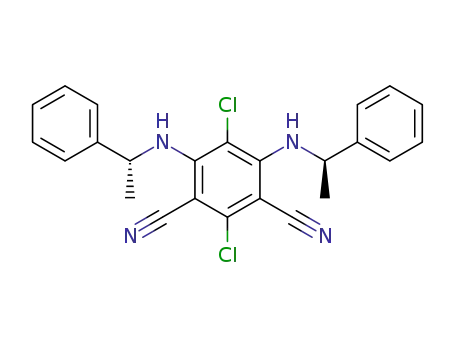
4,6-di-N-((R)-1'-phenylethylamino)-2,5-dichloro-1,3-dicyanobenzene
Conditions
| Conditions | Yield |
|---|
| In methanol at 100℃; for 24h; | 96% |
Conditions
| Conditions | Yield |
|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 8h; Green chemistry; | 95% |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 78% |
| With sodium hydroxide In N,N-dimethyl-formamide at 20℃; for 5h; | |
Conditions
| Conditions | Yield |
|---|
| at 120℃; for 0.2h; Neat (no solvent); Microwave irradiation; regiospecific reaction; | 93% |
| at 20 - 120℃; Neat (no solvent); Microwave irradiation; Combinatorial reaction / High throughput screening (HTS); | |
Conditions
| Conditions | Yield |
|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 8h; Green chemistry; | 93% |
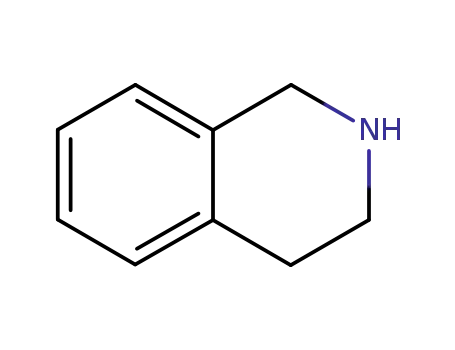
- 91-21-4
1,2,3,4-tetrahydroisoquinoline
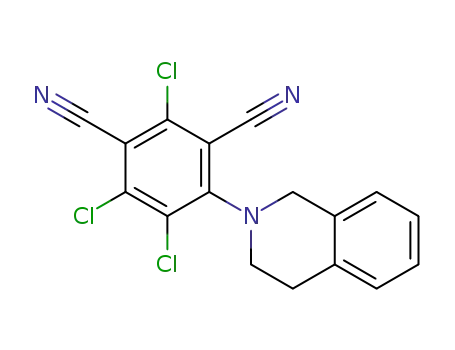
2,5,6-trichloro-6-(3,4-quinolin-2 (1H)-yl)isophthalonitrile
Conditions
| Conditions | Yield |
|---|
| In 1,4-dioxane at 30℃; for 0.166667h; | 93% |
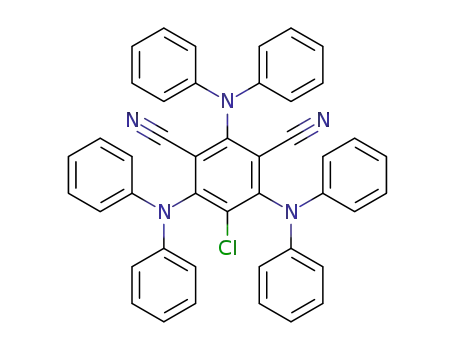
5-chloro-2,4,6-tris(diphenylamino)isophthalonitrile
Conditions
| Conditions | Yield |
|---|
Stage #1: diphenylamine With sodium hydride In tetrahydrofuran at 50℃; for 0.5h; Schlenk technique; Inert atmosphere;
Stage #2: chlorothalonil In tetrahydrofuran at 20℃; for 24h; Schlenk technique; Inert atmosphere; | 93% |
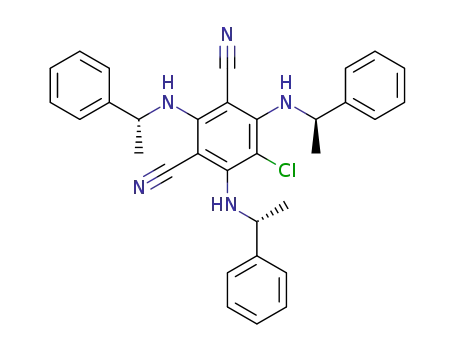
2,4,6-tri-N-((R)-1'-phenylethylamino)-5-chloro-1,3-dicyanobenzene
Chlorothalonil History
CHLOROTHALONIL was first registered for use in the US in 1966. In 1997, the most recent year for which data is available, it was the third most used fungicide in the US, behind only sulfur and copper, with some 12 million lbs used in agricultural alone that year.Including non-agricultural uses, the EPA estimates that on average almost 15 million lbs were used in annually from 1990-1996.
Chlorothalonil Consensus Reports
IARC Cancer Review: Group 3 IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 7 ,1987,p. 56.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Limited Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 30 ,1983,p. 319.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) . NCI Carcinogenesis Bioassay (feed); Clear Evidence: rat NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-41 ,1978. . Cyanide and its compounds are on the Community Right-To-Know List. Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program.
Chlorothalonil Standards and Recommendations
DFG MAK: Confirmed Animal Carcinogen with Unknown Relevance to Humans
Chlorothalonil Specification
The IUPAC name of Chlorothalonil is 2,4,5,6-tetrachlorobenzene-1,3-dicarbonitrile. With the CAS registry number 1897-45-6, it is also named as 1,3-Dicyanotetrachlorobenzene. The product's categories are organics; aromatics pesticides & metabolites; endocrine disruptors (draft); EPA; fungicides; pesticides. It is white crystal with no odor when pure; technical grade has a slightly pungent odor. It is stable in neutral or acidic aqueous media and may react violently with strong oxidizing acids. Additionlly, this chemical should be sealed in the container and stored at the temperature of 0-6 °C.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 2.88; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.88; (4)ACD/LogD (pH 7.4): 2.88; (5)ACD/BCF (pH 5.5): 90.2; (6)ACD/BCF (pH 7.4): 90.2; (7)ACD/KOC (pH 5.5): 873.26; (8)ACD/KOC (pH 7.4): 873.26; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Index of Refraction: 1.632; (13)Molar Refractivity: 55.19 cm3; (14)Molar Volume: 154.6 cm3; (15)Polarizability: 21.88×10-24 cm3; (16)Surface Tension: 71.2 dyne/cm; (17)Enthalpy of Vaporization: 59.52 kJ/mol; (18)Vapour Pressure: 4.36E-05 mmHg at 25°C; (19)Exact Mass: 265.878609; (20)MonoIsotopic Mass: 263.881559; (21)Topological Polar Surface Area: 47.6; (22)Heavy Atom Count: 14; (23)Complexity: 284.
Preparation of Chlorothalonil: It can be obtained by the direct chlorination of isophthalonitrile or by treating tetrachloroisophthaloyl amide with phosphoryl chloride.
Uses of Chlorothalonil: It is a polychlorinated aromatic mainly used as a broad spectrum, non-systemic fungicide, with other uses as a wood protectant, pesticide, acaricide, and to control mold, mildew, bacteria, algae. In addition, it can react with methanol to get 1,3-dicyano-4-methoxy-2,5,6-trichlorobenzene. This reaction needs reagent water and KOH by heating. The reaction time is 1 hours. The yield is 45%.
.gif)
When you are using this chemical, please be cautious about it as the following:
It is highly flammable and harmful by inhalation, in contact with skin and if swallowed. In addition, it is irritating to eyes and respiratory system. Chlorothalonil is very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. After contact with skin, wash immediately with plenty of soap-suds. If you want to contact this product, you must wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) This material and its container must be disposed of as hazardous waste. And people should avoid release to the environment.
People can use the following data to convert to the molecule structure.
1. SMILES:Clc1c(C#N)c(Cl)c(C#N)c(Cl)c1Cl
2. InChI:InChI=1/C8Cl4N2/c9-5-3(1-13)6(10)8(12)7(11)4(5)2-14
The following are the toxicity data which has been tested.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
| dog | LD50 | oral | > 5gm/kg (5000mg/kg) | | "Agrochemicals Handbook," with updates, Hartley, D., and H. Kidd, eds., Nottingham, Royal Soc of Chemistry, 1983-86Vol. A090, Pg. 1984, |
| mouse | LD50 | intraperitoneal | 2500ug/kg (2.5mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY)
BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) | Industrial Health. Vol. 4, Pg. 11, 1966. |
| mouse | LD50 | oral | 3700mg/kg (3700mg/kg) | | Hokkaidoritsu Eisei Kenkyushoho. Report of the Hokkaido Institute of Public Health. Vol. 30, Pg. 53, 1980. |
| rabbit | LD50 | skin | > 10gm/kg (10000mg/kg) | | Pesticide Manual. Vol. 9, Pg. 159, 1991. |
| rat | LC50 | inhalation | 310mg/m3/1H (310mg/m3) | | Farm Chemicals Handbook. Vol. -, Pg. C72, 1991. |
| rat | LD50 | oral | 10gm/kg (10000mg/kg) | | "Agricultural Chemicals," Thomson, W.T., 4 vols., Fresno, CA, Thomson Publications, 1976/77 revisionVol. 4, Pg. 75, 1976/1977. |
| rat | LD50 | skin | > 2500mg/kg (2500mg/kg) | | Fundamental and Applied Toxicology. Vol. 7, Pg. 299, 1986. |





 T+,
T+, N,
N, Xn,
Xn, F
F










.gif)



