
Organic Letters p. 6000 - 6003 (2012)
Update date:2022-08-15
Topics:
 Lauer, Matthew G.
Lauer, Matthew G.
 Henderson, William H.
Henderson, William H.
 Awad, Amneh
Awad, Amneh
 Stambuli, James P.
Stambuli, James P.
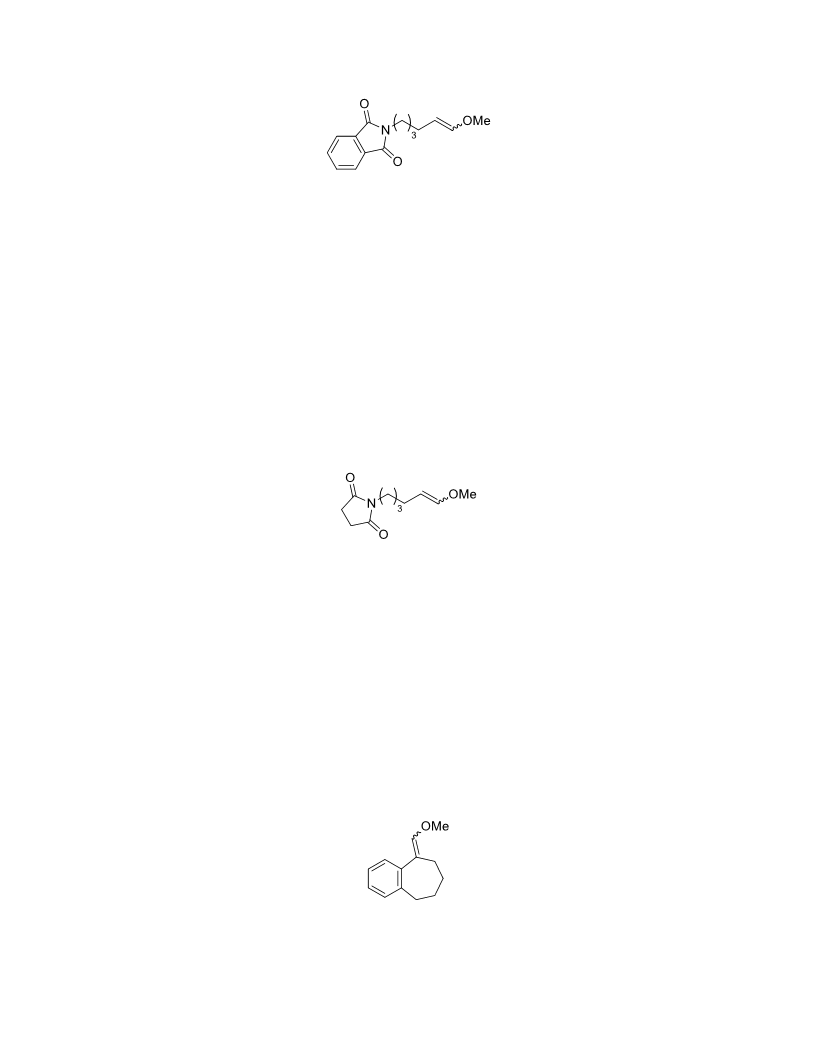
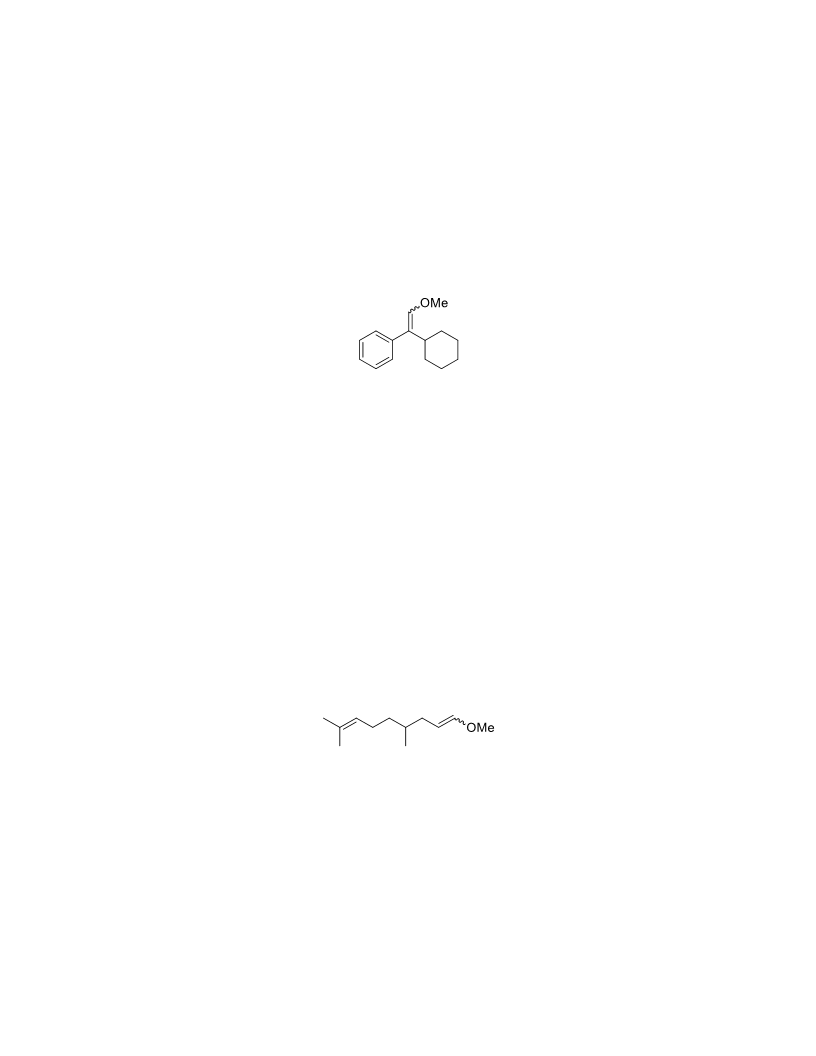
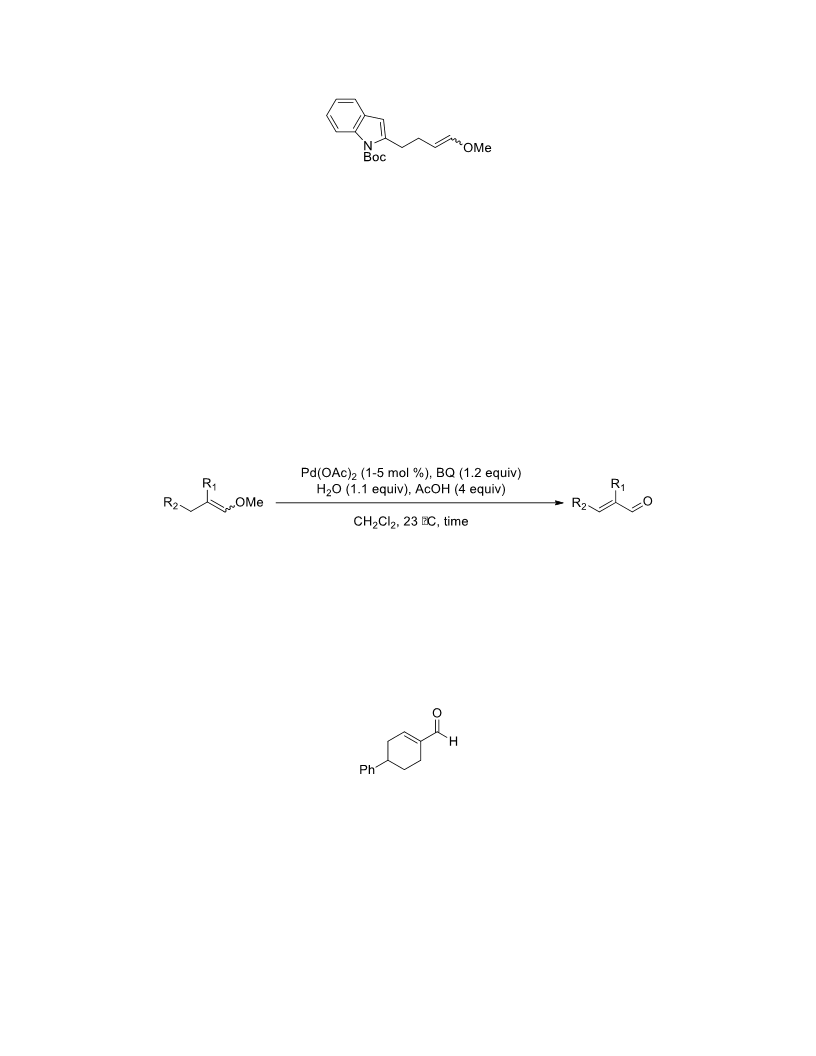
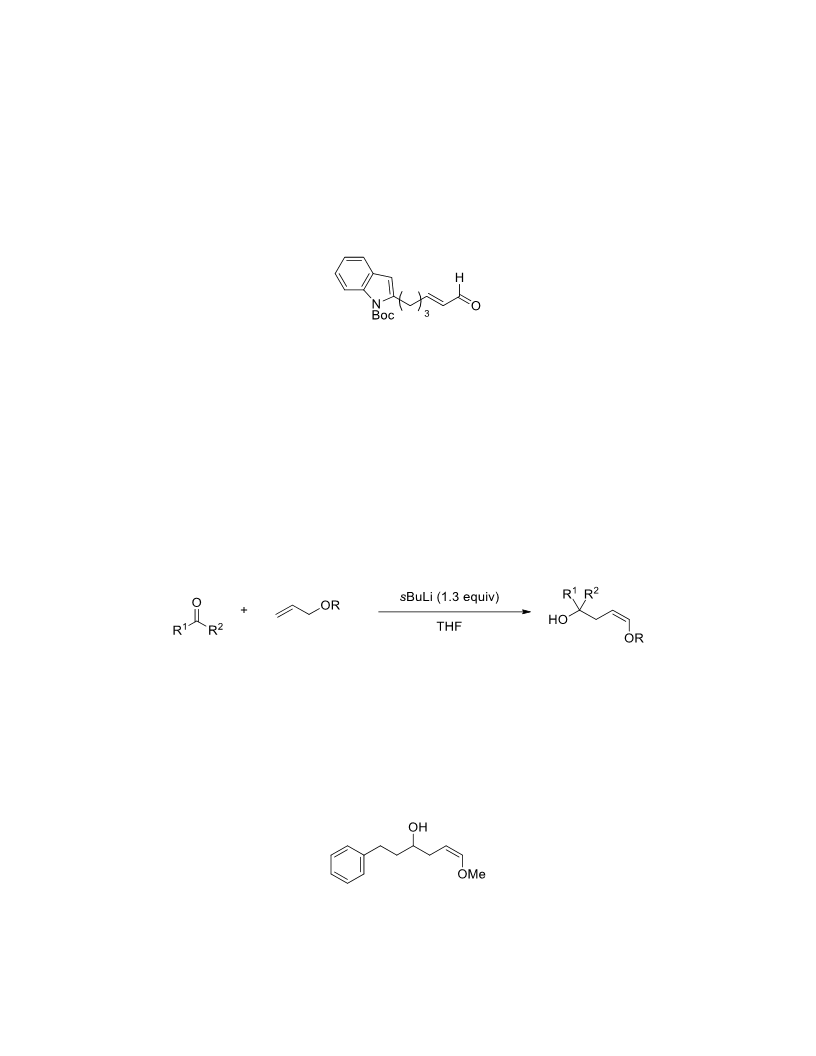
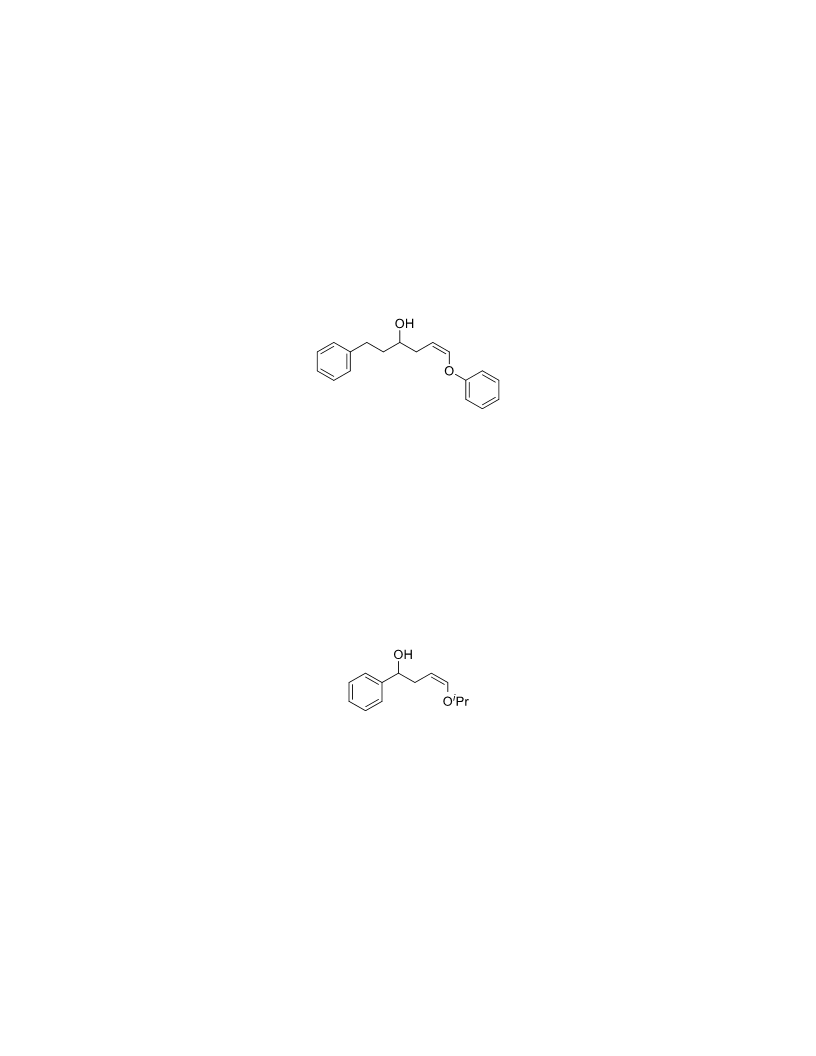
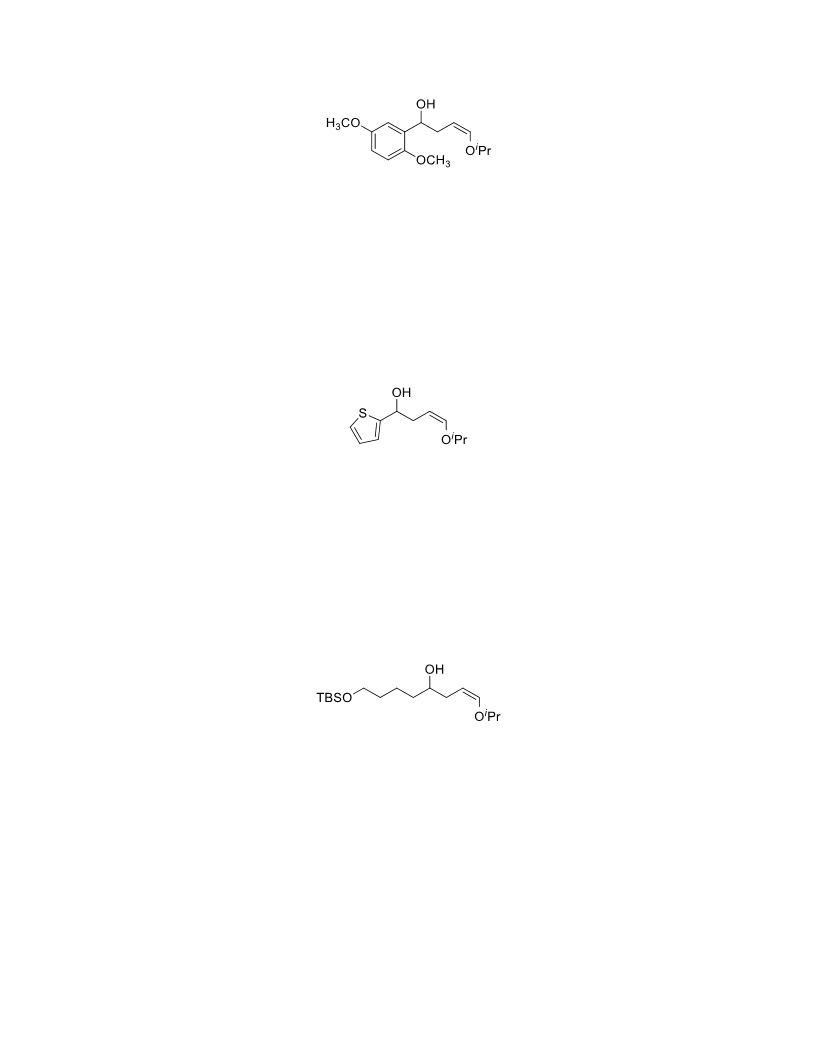
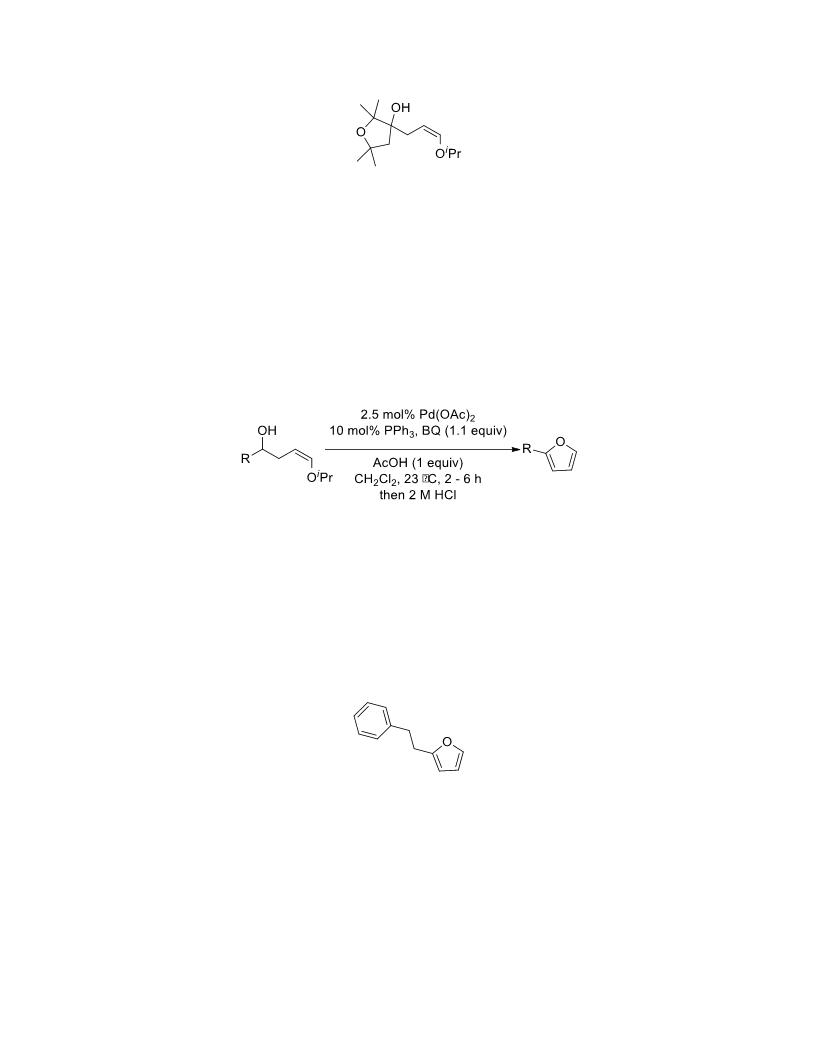
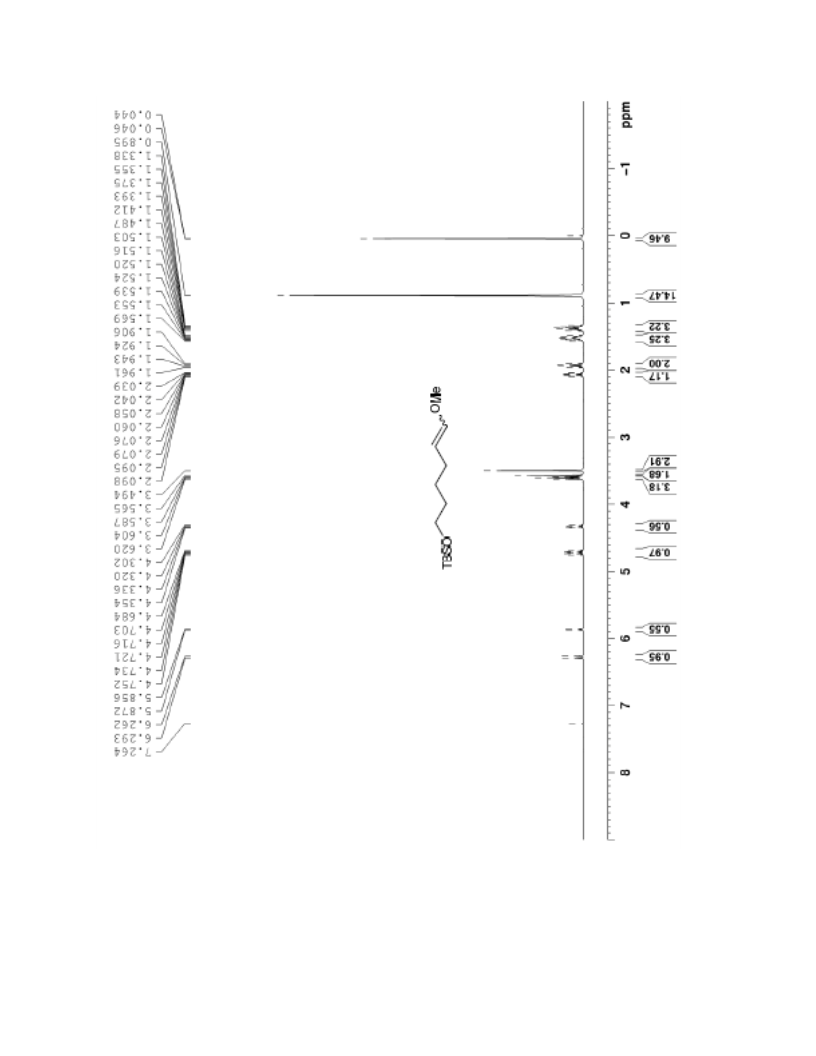
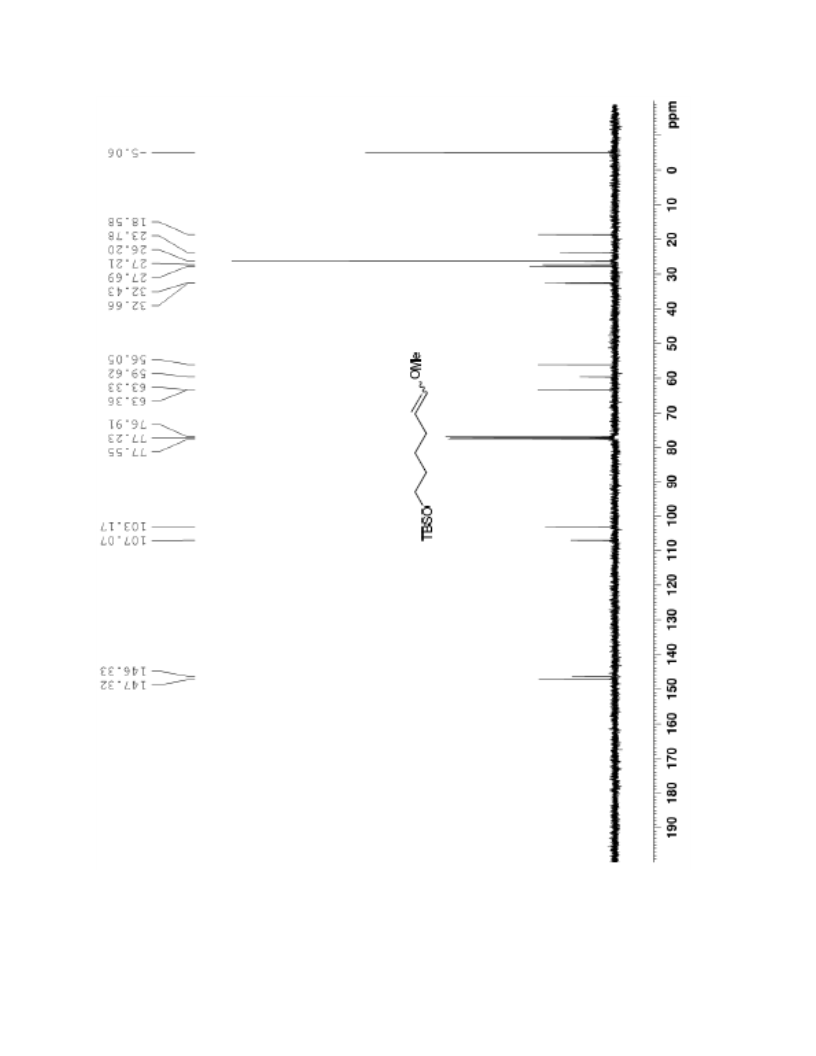
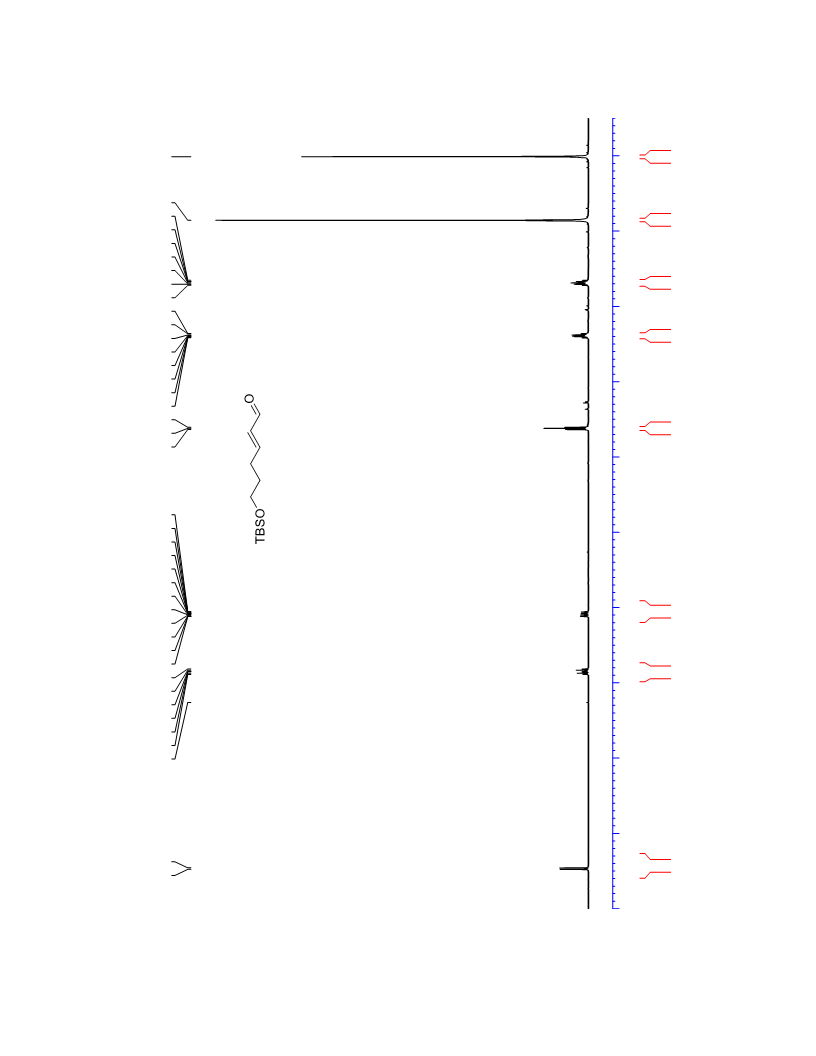
The palladium-catalyzed oxidation of alkyl enol ethers to enals, which employs low loadings of a palladium catalyst, is described. The mild oxidation conditions tolerate a diverse array of functional groups, while allowing the formation of di-, tri-, and tetrasubtituted olefins. The application of this methodology to intramolecular reactions of alkyl enol ethers containing pendant alcohols provides furan and 2,5-dihydrofuran products.
View More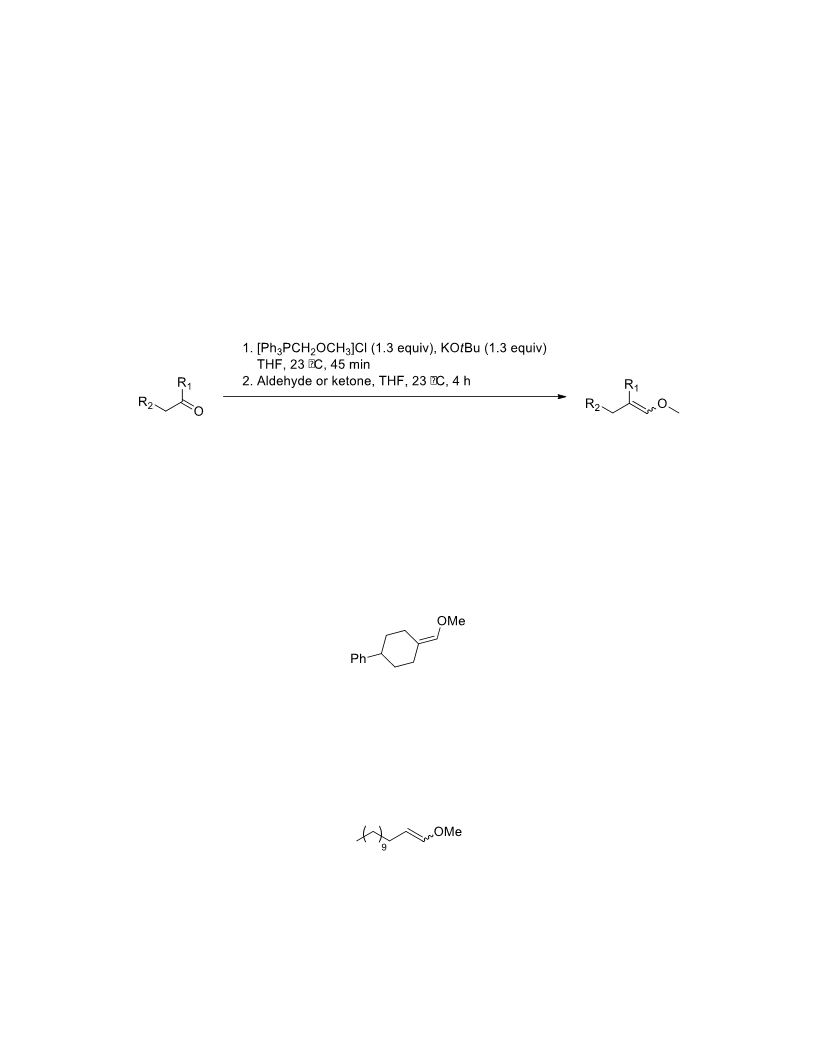











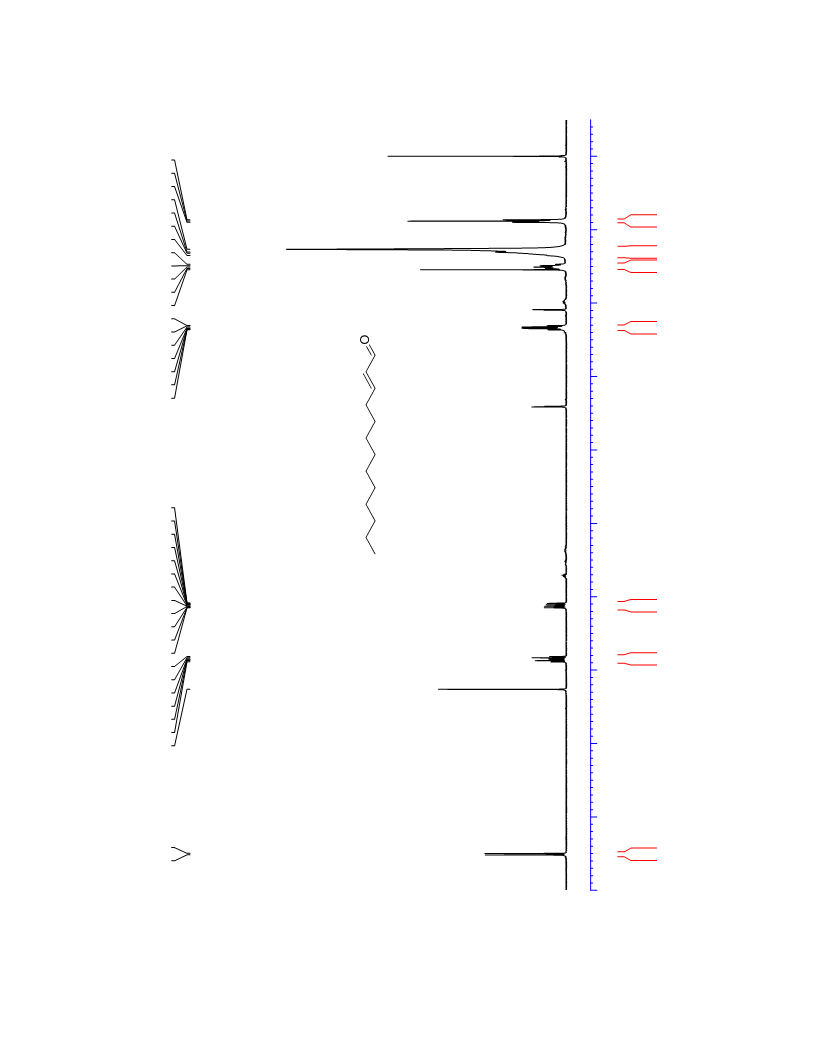
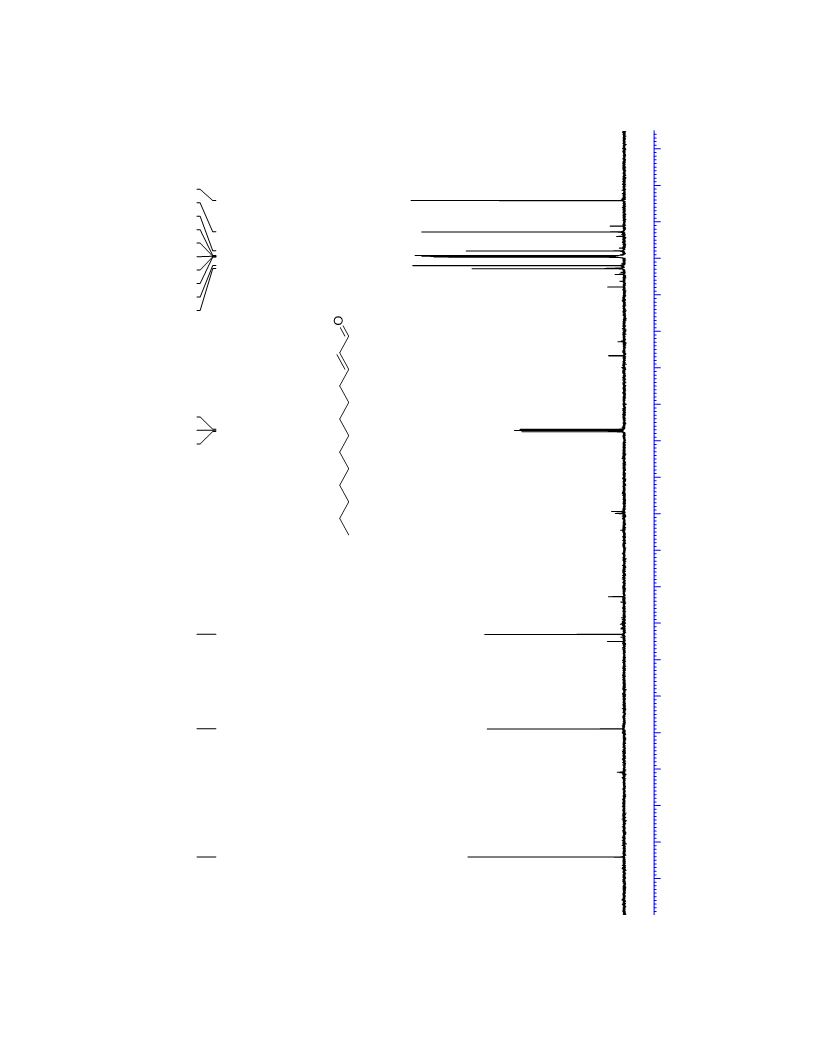

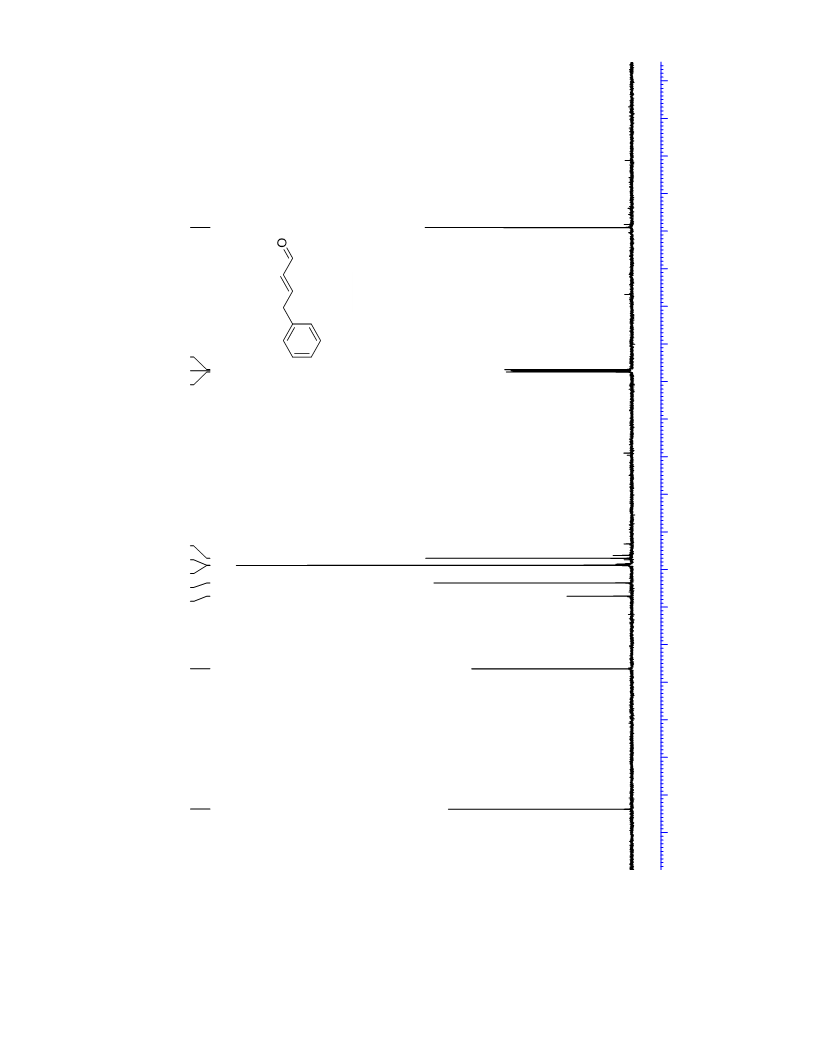

Contact:+86-511-88790000
Address:338 North Yushan Rd, Zhenjiang, Jiangsu 212016
Zhonghao (dalian) Research and Design Institute of Chemical Industry Co., Ltd
Contact:+86 411 84674606
Address:201, Huangpu Road , Shahekou District, Dalian ,116023-China
Frapp's Chemical (NFTZ) Co.,Ltd
Contact:+86-576-86137892
Address:General Chamber of Commercial Building, 159 Wanchang Middle Road, Wenling, Zhejiang, China
Contact:+86-10-67147360/67107388
Address:No.18 Guangming Zhongjie, Chongwen District, Beijing, 100061, China
shanghai Tauto Biotech Co., Ltd
website:http://www.tautobiotech.com/en/index.htm
Contact:+86-21-51320588 ext. 8025
Address:No. 326, Aidisheng Rd , Zhangjiang Hi-tech Park, Shanghai , P.R.CHINA
Doi:10.1055/s-0032-1316753
(2012)Doi:10.1016/j.molstruc.2021.131154
(2021)Doi:10.1016/j.tetlet.2012.09.096
(2012)Doi:10.1016/j.ejmech.2012.09.019
(2012)Doi:10.1248/cpb.42.1784
(1994)Doi:10.1016/j.bmc.2012.10.009
(2012)