Y. Hu et al. / Molecular Catalysis 445 (2018) 316–326
317
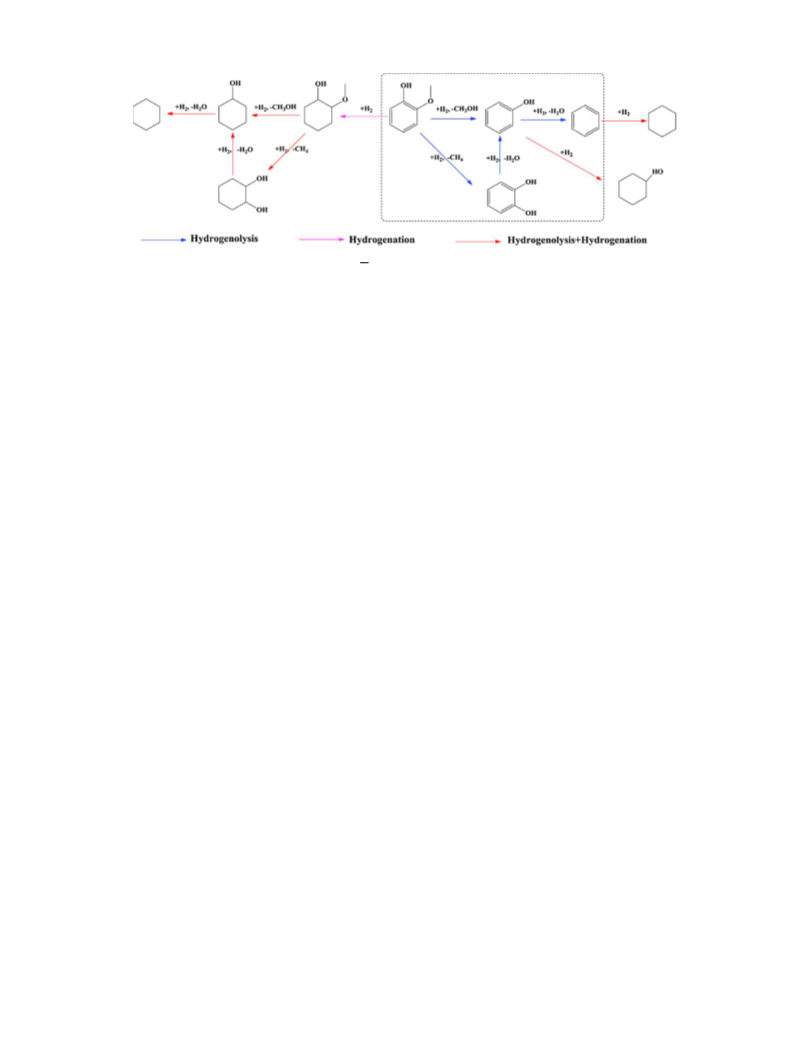
Scheme 1. Cleavage of C O bonds and hydrogenation of aromatic ring.
the stability of Ir-AC/N catalyst improved a lot compared with Ir-AC,
on account of the interaction between N-groups and Ir particles.
In this study, a novel kind of catalysts were prepared by loading
Ru and Ni on nitrogen-doped activated carbon (AC/N), and their
performances in hydrogenolysis of lignin based monomeric sub-
strates (guaiacol, alkyl substituted guaiacol, syringol, etc.) were
into 5 mL water, then 0.75 g nitrogen-doped carbon support was
added into the solution with stirring for 5 h at room tempera-
ture. After that, water was removed by a rotary evaporator and
◦
the obtained powder was dried at 110 C overnight. The dried sam-
◦
ples were reduced in a H2 flow at 200 C for 5 h prior to use. The
obtained catalyst was denoted as x%Ru–y%Ni-AC/N, where x% and
y% refer to the mass percentage of Ru and Ni.
◦
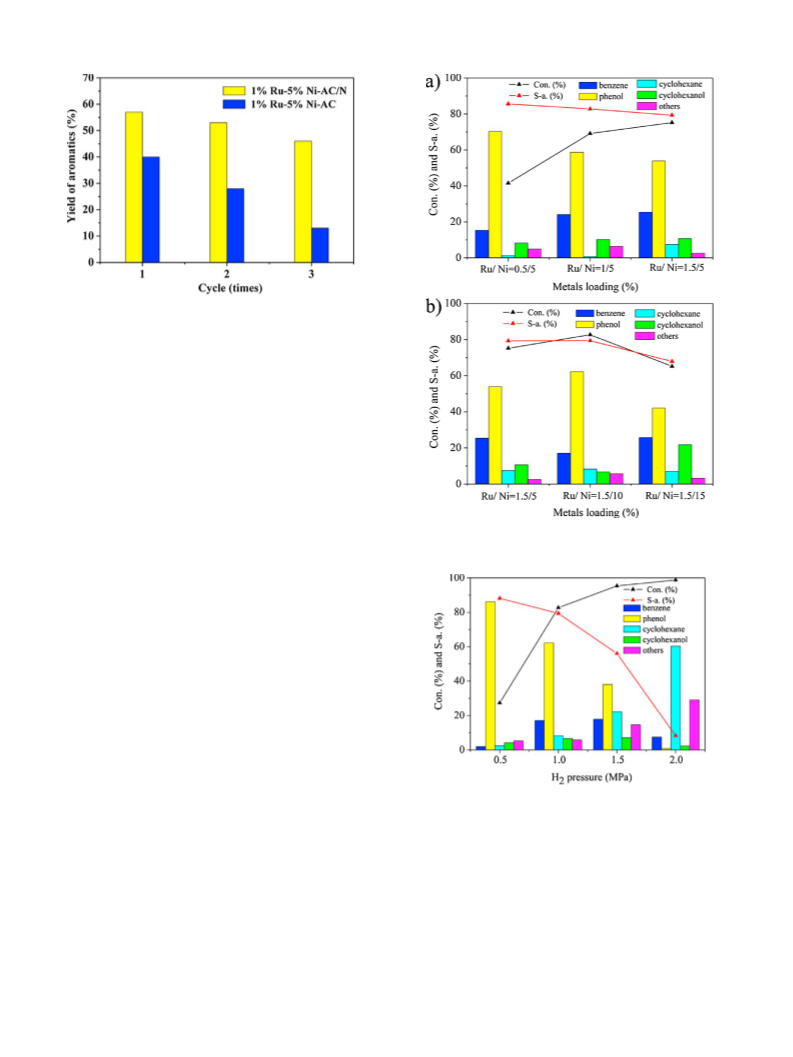
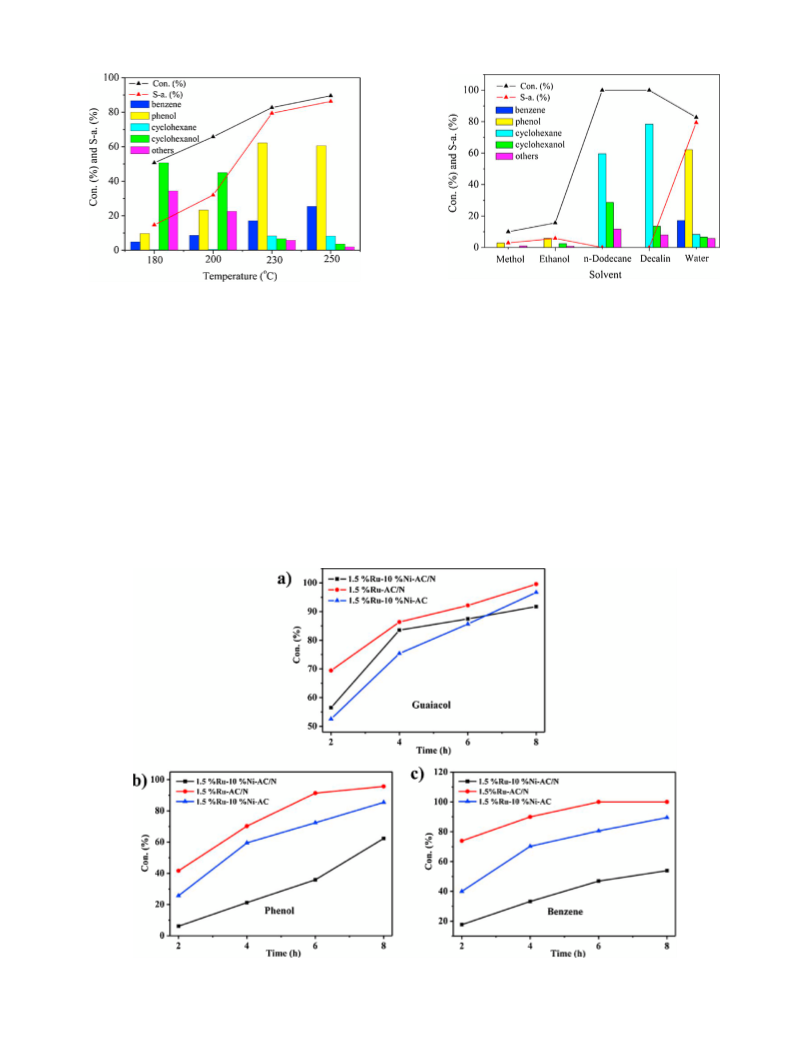
tested under mild reaction conditions (1.0 MPa, 230 C, in aque-
ous). The influence of major reaction parameters, such as metal
loading, H pressure, reaction temperature and solvent, were fully
2
investigated and the possible reaction pathways were also pro-
posed. Moreover, the interaction among Ru, Ni and the electron
2.3. Catalyst characterization
rich N atoms were analyzed in detail by BET, XRD, HRTEM, H -TPR,
2
XPS and ICP-MS to dissect the good aromatics yield and enhanced
durability of the new catalyst.
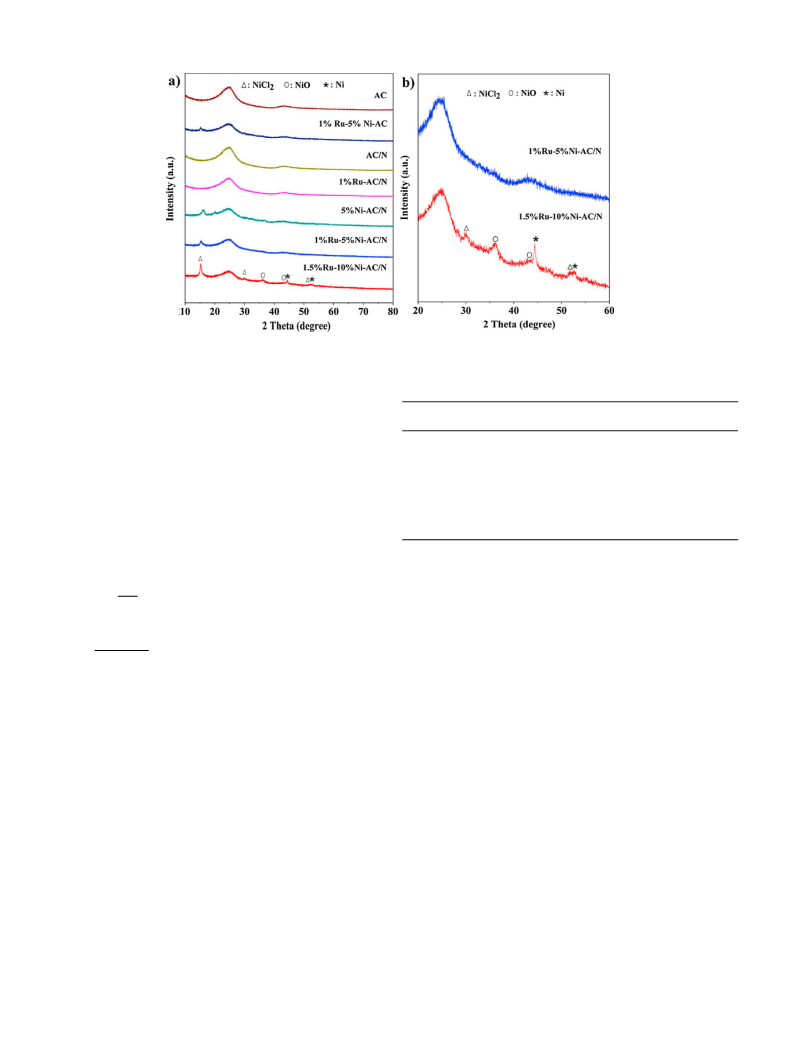
BET (Brunauer-Emmett-Teller) and Barrett-Joyner-Halenda
BJH) were conducted using an ASAP-2020 analyzer. XRD (Powder
(
X-ray diffraction) was recorded at a Bruker D8 diffractometer with
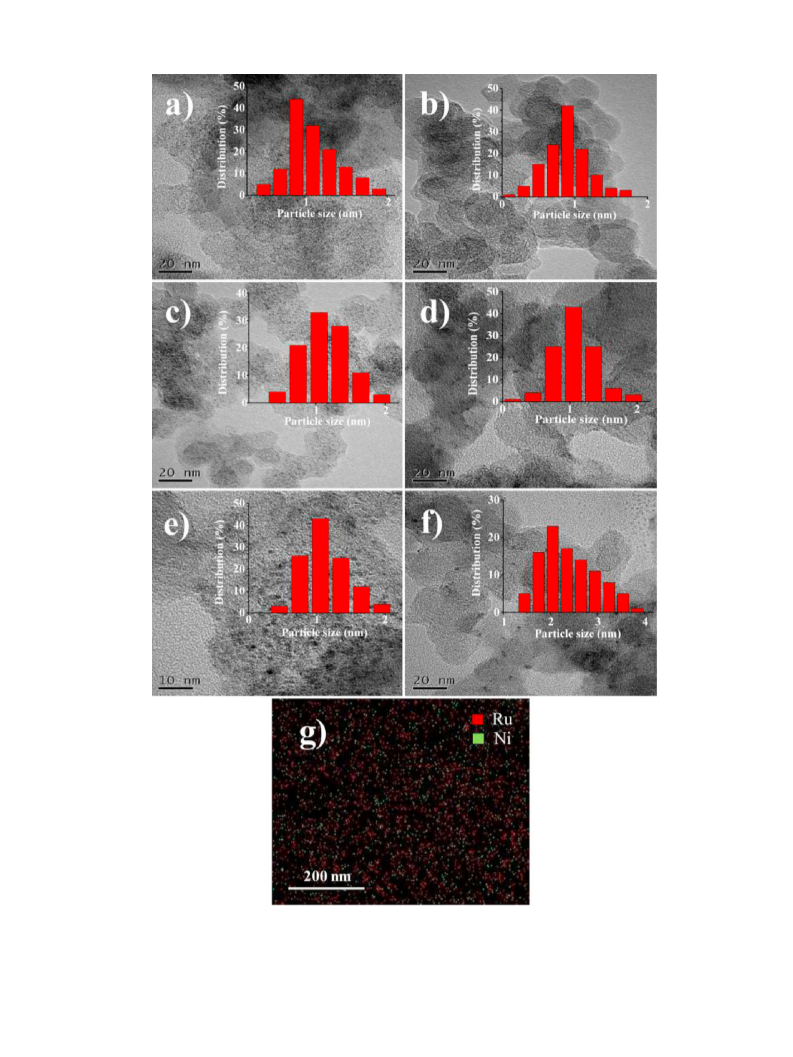
Cu target and K␣ ( = 1.54 Å) radiation. HRTEM (High-resolution
transmission electron microscopy) images were performed with
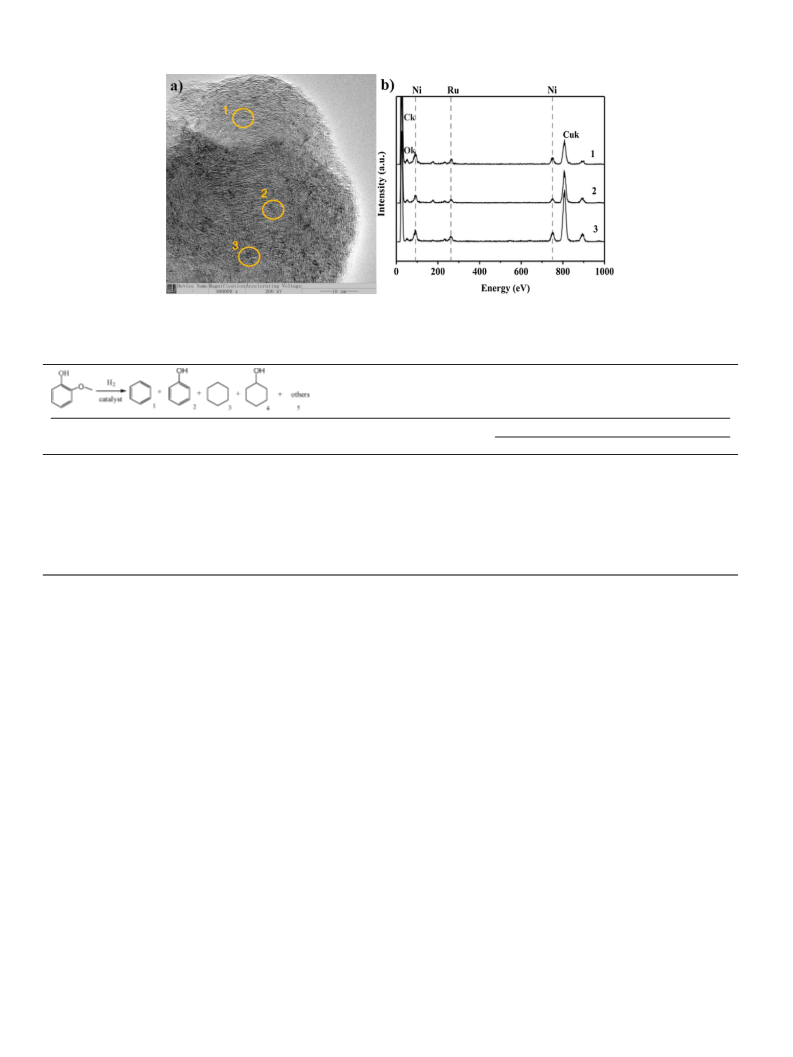
FEI Tecnai G20 instrument. EDS elemental mappings were carried
out on Hitachi SU8000 instrument and XEDS elemental analy-
ses were performed with JEM-2100F electron microscope which
equipped with an Oxford X-MaxN80 T energy dispersive X-ray
spectroscopy. The XEDS elemental analyses were tested by con-
trolling the position of irradiated area within 5 nm. During TEM
samples prepared, the powder samples were dissolved in ethanol
under ultrasonication for 5 min, and then several droplets of the
sample were laid on a copper grid coated with carbon film.
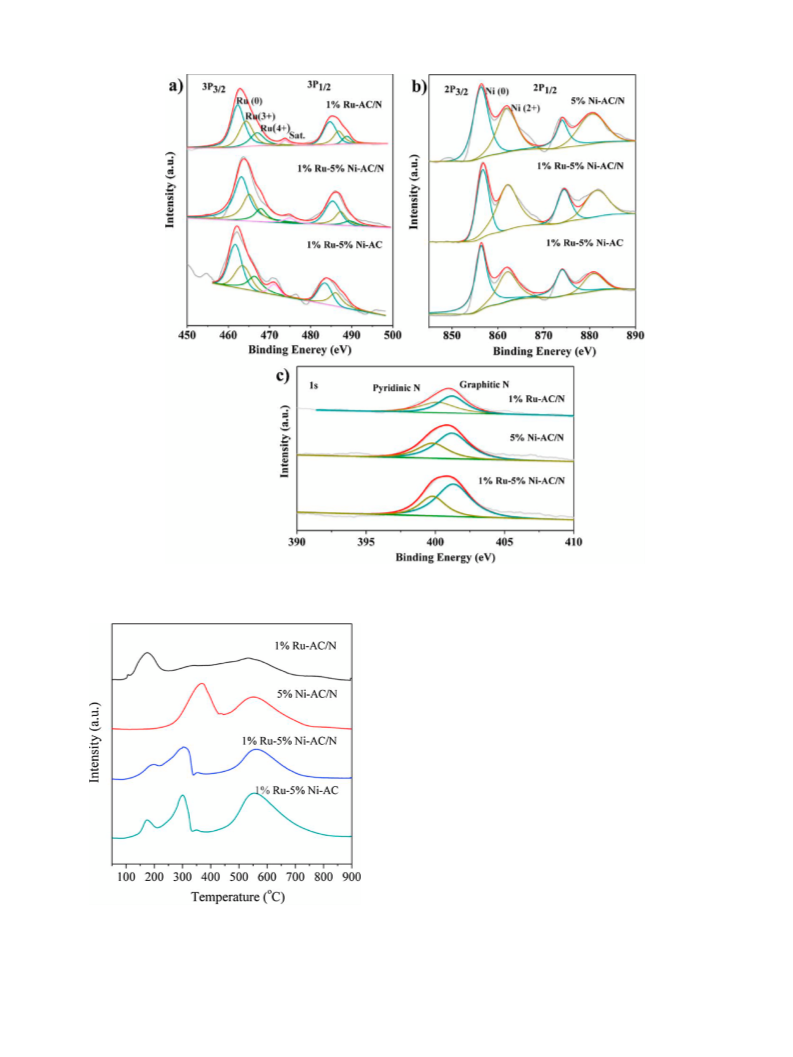
The XPS (X-ray photoelectron spectroscopy) spectra were per-
formed with a Thermo Escalab 250XI system using an Al K␣
(1486.6 eV) as the X-ray source and deconvoluted with XPS PEAK41
software. The binding energies were calibrated using a criterion of
C 1 s peak (284.8 eV) as a reference.
2
. Experimental
2.1. Materials
Commercially available chemicals including metal precursors
of Ru, Ni, Pt and Fe, 1,10-phenanthroline, lignin phenolic model
compounds and other organic reagents were purchased from SCRC,
Alfa Aesar or TCI and used as received. Activated carbon support
(
Vulcan XC72R) was provided by Cabot Corporation.
2.2. Catalyst preparation
2.2.1. The preparation of AC/N support
The AC/N support were prepared according to reference [21].
Generally, 5 g 1,10-phenanthroline monohydrate was dissolved in
5 mL ethanol, and then mixed slowly with aqueous solution of
H SO (5 g 98% H SO diluted with 25 mL deionized water). Ethanol
2
H -TPR (H -temperature programmed reduction) was carried
2
4
2
4
2
2
and water were removed by distillation after the mixture stirred for
h. Followed, 2.5 g of the synthesized phenanthroline sulfuric acid
out on a Chemisorption Analyzer (Micromeritics AutoChem II 2920)
4
equipped with a thermal conductivity detector (TCD). For H -TPR
2
salt, 5 g activated carbon (Vulcan XC72R), 80 mL of deionized water
and 50 mL of ethanol were added into a 250 mL round-bottom flask
and stirred at room temperature for 12 h. After all solvents were
analysis, about 100 mg of sample was used for each measurement
◦
and pre-treated in a Ar flow at 200 C for 2 h. TPR was initiated from
◦
◦
−1
room temperature to 900 C at 10 C min with a mixture flow of
◦
−1
evaporated, the obtained black solids were calcined at 900 C in a
10% H /Ar (20 mL min ).
2
tube furnace under N atmosphere for 6 h with a heating rate of
ICP-MS (inductively coupled plasma mass spectrometry) was
performed on an Agilent 7700ce system. After each reaction, the
aqueous solution was diluted with water to 100.0 mL prior to sub-
ject to the analyzer.
2
◦
−1
5
C min . Finally, the nitrogen-doped activated carbon support
was obtained.
For H -TPD (H -temperature programmed desorption) analy-
2.2.2. The preparation of mono- and bi- metallic catalysts
2
2
ses, Micromeritics AutoChem II 2920 Chemisorption Analyzer was
used as instrument. The sample (100 mg) was pre-treated under Ar
All catalysts were prepared by incipient wetness impregnation
method. Chloride nickel hydrate (NiCl ·6H O), rhodium chloride
2
2
◦
◦
flow at 300 C for 2 h, and then cooled to 230 C for 2 h. After exposed
hydrate (RuCl ·6H O), palladium chloride hydrate (PdCl ·H O),
3
2
2
2
−
1
to a 10% H /Ar flow (20 mL min ), the physical adsorption of H2
iron chloride hydrate (FeCl ·6H O), and activated carbon (AC),
2
3
2
was purged by Ar flow for 2 h until pulses base line became sta-
nitrogen-doped activated carbon (AC/N) were used as metal pre-
cursors and supports, respectively. Take 1%Ru-5%Ni-AC/N as an
example, 0.02 g RuCl ·3H O and 0.15 g NiCl ·6H O were dissolved
◦
ble. Furnace temperature was increased from 100 to 800 C with a
◦
−1
heating rate of 10 C min in a Ar flow.
3
2
2
2

 Hu, Yinghui
Hu, Yinghui
 Jiang, Guangce
Jiang, Guangce
 Xu, Guoqiang
Xu, Guoqiang
 Mu, Xindong
Mu, Xindong