2
558
STEPOVIK et al.
2
8
6.60 [ОС(СН ) ], 39.57 [С(СН ) ], 81.20 [ОС(СН ) ],
9.02 [С–ООBu-t], 133.05 [НС=], 198.19 [C=O]. The
REFERENCES
3
3
3 3
3 3
1
2
3
4
. Dodonov, V.А., Stepovik, L.P., Sofronova, S.M., and
Mukhina, T.V., Zh. Obshch. Khim., 1988, vol. 58, no. 7,
p. 1578.
. Stepovik, L.P., Gulenova, M.V., and Martynova, I.M.,
Russ. J. Gen. Chem., 2005, vol. 75, no. 4, p. 507. DOI:
second fraction (0.116 g) contained the unreacted
quinone. After washing it with isooctane, 0.098 g of
3
2
,6-di-tert-butyl-о-benzoquinone was obtained, mp
08–209°С.
1
0.1007/s11176-005-0262-6.
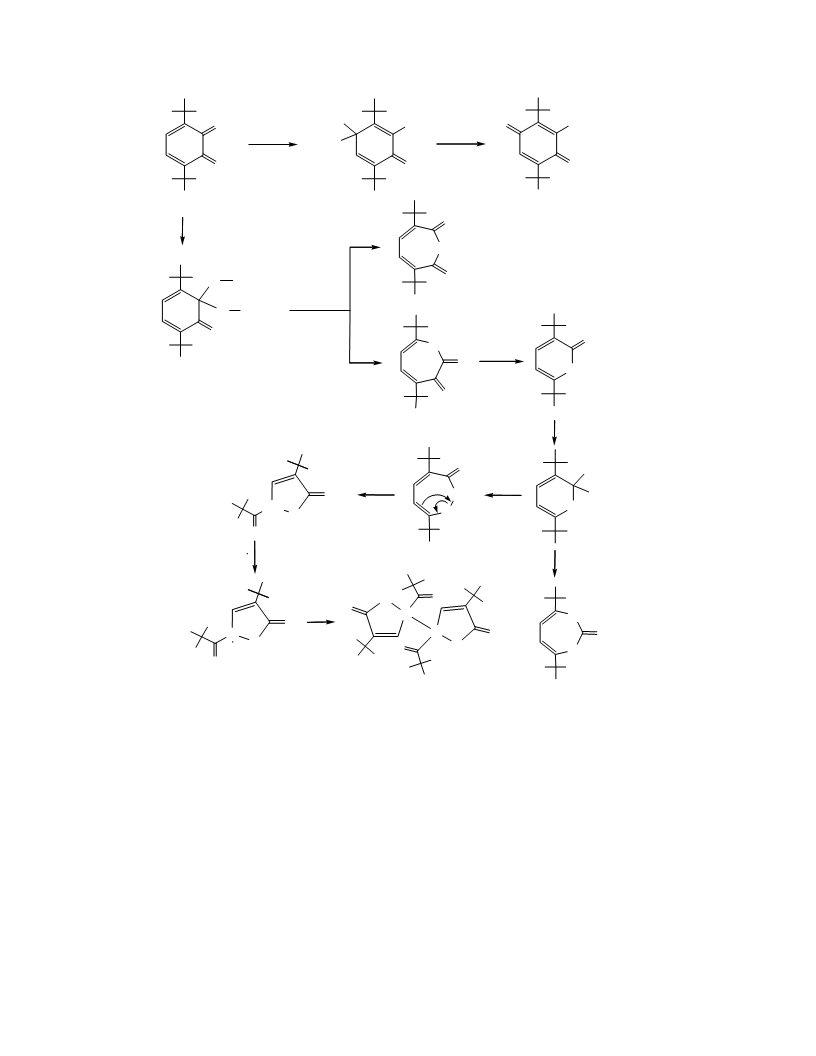
Reaction of 3,6-di-tert-butyl-о-benzoquinone
. Stepovik, L.P., Gulenova, M.V., Tishkina, A.N., and
Cherkasov, V.K., Russ. J. Gen. Chem., 2007, vol. 77,
no. 7, p. 1254. DOI: 10.1134/S1070363207070183.
. Stepovik, L.P., Potkina, A.Yu., and Poddelskii, A.I.,
Russ. J. Gen. Chem., 2013, vol. 83, no. 11, p. 2005.
DOI: 10.1134/S1070363213110066.
with tert-butylhydroperoxide in the presence of
cobalt acetylacetonate (1 : 5:1, С Н , 20°С). 0.20 g of
quinone 4 and 0.41 g of the hydroperoxide 1 were
added to a solution of 0.233 g of the cobalt alkoxide in
1
6
6
8 mL of benzene. The green reaction mixture turned
dark-brown after several minutes, and finally moss
green solution was formed with brown precipitate at
the bottom after 20 h. Carbon oxide CO was detected
in the flask over the solution. 2 days later the solvent
and volatile products were condensed in a trap cooled
with liquid nitrogen. 0.28 g of t-BuOH was detected in
the solution, the starting hydroperoxide was absent.
5
6
7
8
. Dodonov, V.А., Stepovik, L.P., and Sofronova, S.M.,
Zh. Obshch. Khim., 1981, vol. 51, no. 12, p. 2730.
. Muzart, J., Chem. Rev., 1992, vol. 92, no. 1, p. 113.
DOI: 10.1021/cr00009a005.
. Muzart, J., Tetrahedron Lett., 1987, vol. 28, no. 19,
p. 2133. DOI: 10.1016/S0040-4039(00)96062-3.
. Riahi, A. Henin, Fand ., Muzart, J., Tetrahedron Lett.,
1
4
999, vol. 40, no. 12, p. 2303. DOI: 10.1016/S0040-
039(99)00168-9.
The residue, dark-green mass, crystallized upon
incubation and was then purified of the metal via
column chromatography (petroleum ether : diethyl
ether = 22 : 1). The eluate was concentrated and
analyzed by chromatography. The starting benzo-
quinone was absent, and traces of hydroxyquinone 5,
pyrone 7, and anhydride 8 were found. After removal
of the solvent, 3 mL of petroleum ether was added to
the residue, and the dissolved part was decanted. The
remained colorless crystals were washed with 1 mL of
petroleum ether to give 0.089 g of crystalline
9
. Alyea, E.C., Basi, J.S., Bradley, D.C., and Chisholm, M.H.,
J. Chem. Soc. (A), 1971, p. 772. DOI: 10.1039/
J19710000772.
0. Gulenova, M.V., Stepovik, L.P., and Cherkasov, V.K.,
Russ. J. Gen. Chem., 2006, vol. 76, no. 6, p. 980. DOI:
10.1134/S1070363206060235.
11. Tsuji, Y., Ohta, T., Ydo, T., and Minbu, H., J. Organo-
metal. Chem., 1984, vol. 270, no. 2, p. 333. DOI:
10.1016/0022-328X(84)80381-2.
12. Stepovik, L.P., Gulenova, M.V., Shavyrin, A.S., and
Fukin, G.K., Russ. J. Gen. Chem., 2006, vol. 76, no. 8,
p. 1275. DOI: 10.1134/S1070363206080202.
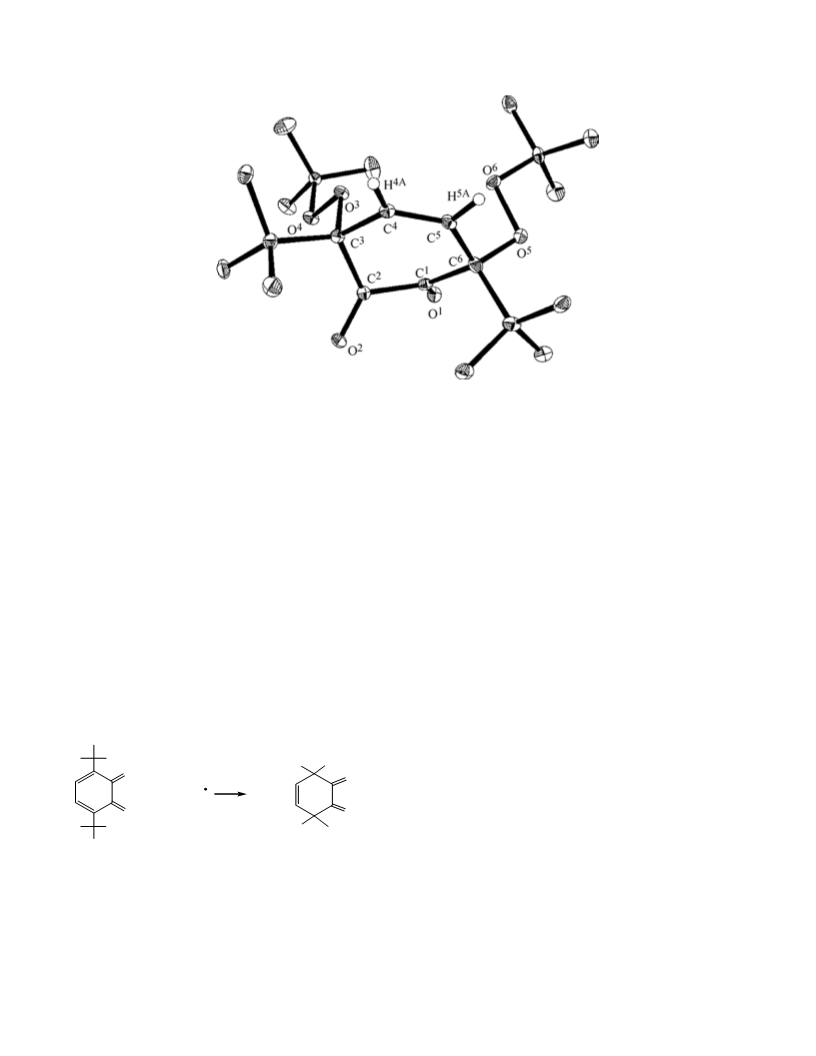
1
–
1
compound 10, mp 189°С. IR spectrum (KBr), ν, cm :
2
1
1
1
975, 2965, 2934, 2873, 1777, 1775, 1702, 1694,
481, 1462, 1396, 1318, 1260, 1131, 1097, 1032,
1
1
1
3. Stepovik, L.P., Potkina, A.Yu., and Poddelskii, A.I.,
Russ. J. Gen. Chem., 2012, vol. 82, no. 9, p. 1805. DOI:
10.1134/S1070363213110066.
1
004, 985, 934, 887. Н NMR spectrum, δ, ppm: 1.16,
1
3
.25, [18Н, С(СН ) ], 6.90 (1Н, СН=). С NMR
3
3
4. Stepovik, L.P., Malysheva, Yu.B., and Fukin, G.K.,
spectrum, δ , ppm: 27.96 [(СН ) С–СО], 29.16 [(СН )
С
3 3
3
Russ. J. Gen. Chem., 2015, vol. 85, no. 6, p. 1401. DOI:
1
5. Glushakova, V.N., Skorodumova, N.A., Nevodchi-
kov, V.I., Abakumova, L.G., Makarenko, N.P., Cherka-
sov, V.K., and Druzhkov, N.O., Russ. Chem. Bull.,
С], 32.05 [С(СН ) ], 46.30 [(СН ) С–СО], 94.77
3
3 3
3 3
0.1134/S1070363215060080.
[
С С–О], 144.54, 144.59 [НС=С]. 170.19 [C=O,
3
furan], 206.01 [C=O, ketone].
According to the IR spectroscopy data, the oily
1
999, vol. 48, no. 5, p. 934. DOI: 10.1007/BF02494640.
6. Matsumoto, M. and Kuroda, K., J. Am. Chem. Soc.,
982, vol. 104, no. 5, p. 1433. DOI: 10.1021/ja00369a052.
residue contained compound 10 along with non-
1
1
–
1
identified carbonyl-containing compounds (ν, cm :
723, 1740).
1
1
7. Funabiki, T., Mizoguchi, A., Sugimoto, T., Tada, S.,
Tsuji, M., Sakamoto, H., and Yohida, S., J. Am. Chem.
Soc., 1986, vol. 108, no. 11, p. 2921. DOI: 10.1021/
ja00271a022.
ACKNOWLEDGMENTS
The authors are thankful to Yu. B. Malysheva for
registration and interpretation of NMR spectra.
1
8. Sawaki, Y. and Foote, C.S., J. Am. Chem. Soc., 1983,
vol. 105, no. 15, p. 5035. DOI: 10.1021/ja00353a030.
RUSSIAN JOURNAL OF GENERAL CHEMISTRY Vol. 85 No. 11 2015

 Stepovik
Stepovik
 Zaburdaeva
Zaburdaeva
 Fukin
Fukin
 Karaghiosoff
Karaghiosoff