DOI: 10.1002/cplu.201402049
Communications
PEG600-Carboxylates as Efficient Reusable Reaction Media
and Acylating Agents for the Resolution of sec-Alcohols
Carlos M. Monteiro,[a] Nuno M. T. LourenÅo,*[b] Frederico C. Ferreira,[b] and
Carlos A. M. Afonso*[a]
Herein is presented a simple, attractive, and reusable method-
ology for one-pot resolution/separation of free sec-alcohols
with enantiomeric excess (ee) values over 90% by the combi-
nation of sustainable acylating agents/solvents (polyethylene
glycol derivatives) and an easily available and common biocat-
alyst (Candida antarctica lipase B, or CAL B) under irreversible
conditions, along with a separation process by extraction or
distillation. A scale-up reaction was carried out with the Fluox-
etine precursor with ee values close to 90% for the R enantio-
mer.
mation of inclusion complexes is the only described method
for resolution of sec-alcohols by crystallization.[3]
The preferred method for sec-alcohol resolution has been ki-
netic resolution (KR). There are a few instances of chemical KR
methods,[4] but the majority of applications focus on the enzy-
matic kinetic resolution (EKR), either by acylation of an alcohol
or by hydrolysis of the acylated product. EKR is a well-estab-
lished method given that the biocatalysts employed accept
a broad range of substrates and afford very high enantioselec-
tivity. The success of an EKR is dependent on three factors:
1) high efficiency of the enzyme, the stability of which can be
increased in selected organic solvents, ionic liquids (ILs),[5] and
eutectic solvents;[6] 2) the occurrence of EKR under irreversible
conditions achieved by either using vinyl esters or anhydrides
as acylating agents,[7] or drying agents for water removal or
product removal (water or volatile alcohol) by evaporation
under vacuum;[8] and 3) effective separation of the two enan-
tiomers as free alcohol and ester.[9] The separation step is an
important factor for upscaling a process. Separation methods
are based on the type of acylating agent. Simple nonfunction-
alized acyl groups are commonly separated by flash chroma-
tography on silica and more rarely by distillation.[8,9d,10] Separa-
tion by extraction is a very common method applied to a varie-
ty of acylating agents and esters including succinic
esters,[9a,10e,11] amine-based acylating agents,[10e] carbonate
esters that contain a pendant polyethylene glycol (PEG) unit,[12]
and a fluorinated pendant acylating agent in combination with
a fluorinated extraction phase.[9c,13] Extraction techniques have
also used ILs as membranes,[14] extraction with supercritical
CO2 (sc-CO2), and sc-CO2 in combination with ILs.[15] Other sep-
aration methods have also been reported such as distillation,[8]
precipitation,[9d,12b,16] and sublimation.[17]
Enantiomerically pure sec-alcohols are an important class of
biologically relevant compounds amenable to facile and selec-
tive functional-group transformation. Stereoselective synthesis
of a desired enantiomer can be achieved through asymmetric
reduction of ketones, stereoselective nucleophilic addition to
aldehydes, dynamic kinetic resolution of racemic sec-alcohols,
and modification of enantiomeric natural products available
from the chiral pool.
Resolution of racemic alcohols is an appealing strategy, par-
ticularly when both enantiomers are valuable. Standard chro-
matographic methods such as chiral high-performance liquid
chromatography (HPLC) and gas–liquid chromatography (GLC)
excel for analytical purposes and small-scale preparative pur-
poses but are not viable for any meaningful scale-up. Simulat-
ed moving-bed chromatography has allowed for continuous
operation but the high cost of chiral stationary phases and
need for careful optimization for each substrate has reserved
this method for more intensive resolutions.[1] Membrane tech-
nology using chiral selectors is another continuous resolution
method.[2] Crystallization has been the standard for racemic
resolution, particularly for substrates that can form organic
salts, such as amines and carboxylic acids. By contrast, the for-
The extraction methods listed above do possess certain limi-
tations such as the need for expensive and poorly reusable re-
action media (e.g., ILs or fluorinated solvents), quite technical
complex procedures (e.g., sc-CO2) and non-reusable enzymes
or acylating reagents. Also, the isolation of the other enantio-
mer can require even further chemical modification (e.g., hy-
drolysis, transesterification of the ester or deprotection of the
tert-butyloxycarbonyl (Boc) group).[9a,11a]
[a] Dr. C. M. Monteiro, Prof. Dr. C. A. M. Afonso
Instituto de Investigażo do Medicamento(iMed.ULisboa)Faculdade de
Farmꢀcia da Universidade de Lisboa
Av. Prof. Gama Pinto, 1649-003 Lisboa(Portugal)
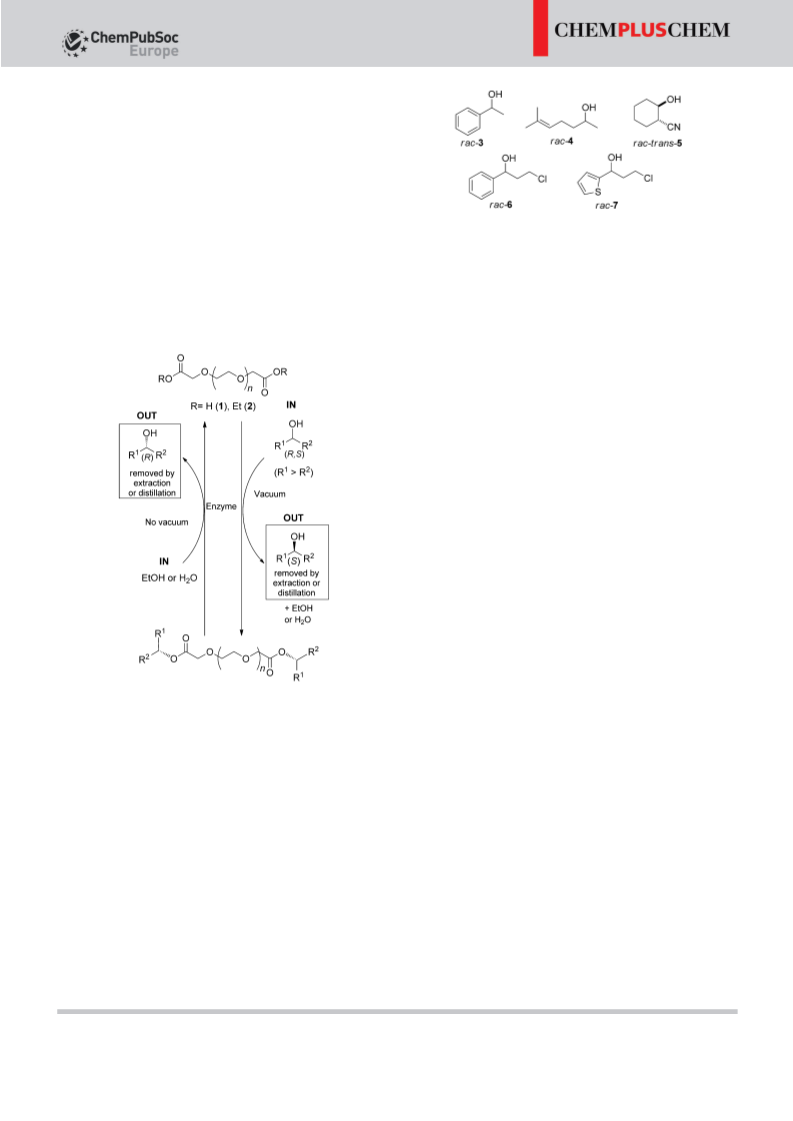
To overcome these limitations, we have developed a meth-
odology with the following features: 1) The use of low-volatile
reaction media such as ILs[9b,18] or fatty esters to avoid the use
of vinyl esters[10b,19] and to enable continuous removal of the
volatile alcohol (transesterification) or water (esterification)
under vacuum. These conditions should drastically minimize
EKR reversibility and afford high yields. 2) The use of the same
[b] Dr. N. M. T. LourenÅo, Dr. F. C. Ferreira
Department of Bioengineering
IBB (Institute for Biotechnology and Bioengineering)
Centre for Biological and Chemical Engineering
Instituto Superior Tꢁcnico, 1049-001 Lisboa(Portugal)
Supporting information for this article is available on the WWW under
ChemPlusChem 2015, 80, 42 – 46
42
ꢀ 2015 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Monteiro, Carlos M.
Monteiro, Carlos M.
 Lourenco, Nuno M. T.
Lourenco, Nuno M. T.
 Ferreira, Frederico C.
Ferreira, Frederico C.
 Afonso, Carlos A. M.
Afonso, Carlos A. M.