D.R. Mohbiya et al.
JournalofPhotochemistry&PhotobiologyA:Chemistry364(2018)40–52
applications.
[15] F. Tancini, Y.-L. Wu, W.B. Schweizer, J.-P. Gisselbrecht, C. Boudon, P.D. Jarowski,
M.T. Beels, I. Biaggio, F. Diederich, 1,1-Dicyano-4-[4-(diethylamino)phenyl]buta-
1,3-dienes: structure-property relationships, Eur. J. Org. Chem. 2012 (2012)
5. Conclusion
[16] L. Zhang, D. Duan, Y. Liu, C. Ge, X. Cui, J. Sun, J. Fang, Highly selective off–on
fluorescent probe for imaging thioredoxin reductase in living cells, J. Am. Chem.
[17] Pavel A. Panchenko, Antonina N. Arkhipova, Olga A. Fedorova, Yuri V. Fedorov,
Marina A. Zakharko, Jonusauskas Gediminas, Controlling photophysics of styr-
ylnaphthalimides through TICT, fluorescence and E,Z-photoisomerization interplay,
[18] L.K. Aschenbach, F.R. Knight, R.A.M. Randall, D.B. Cordes, A. Baggott, M. Bühl,
A.M.Z. Slawin, J.D. Woollins, Onset of three-centre, four-electron bonding in peri-
substituted acenaphthenes: a structural and computational investigation, Dalton
[19] P. Alaei, S. Rouhani, K. Gharanjig, J. Ghasemi, A new polymerizable fluorescent
PET chemosensor of fluoride (F-) based on naphthalimide-thiourea dye,
Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 90 (2012) 85–92, http://dx.doi.
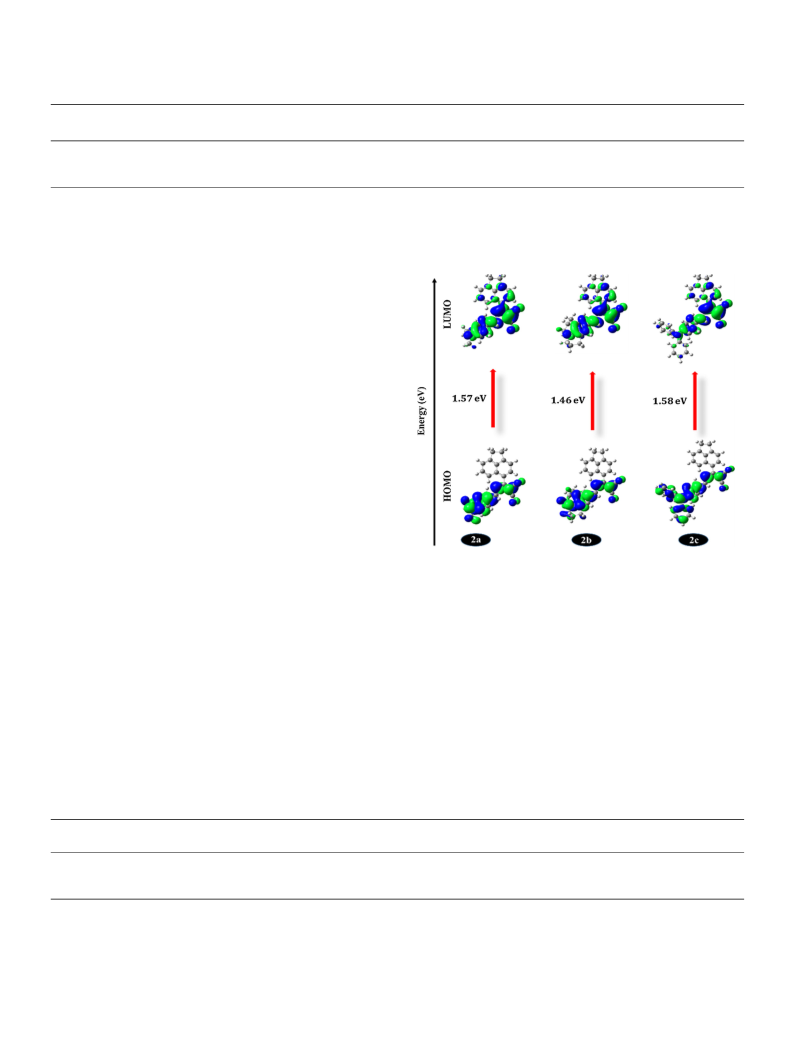
In summary, we have successfully designed and synthesized novel
acenaphthene based D-π-A derivatives with N-substituted donors and
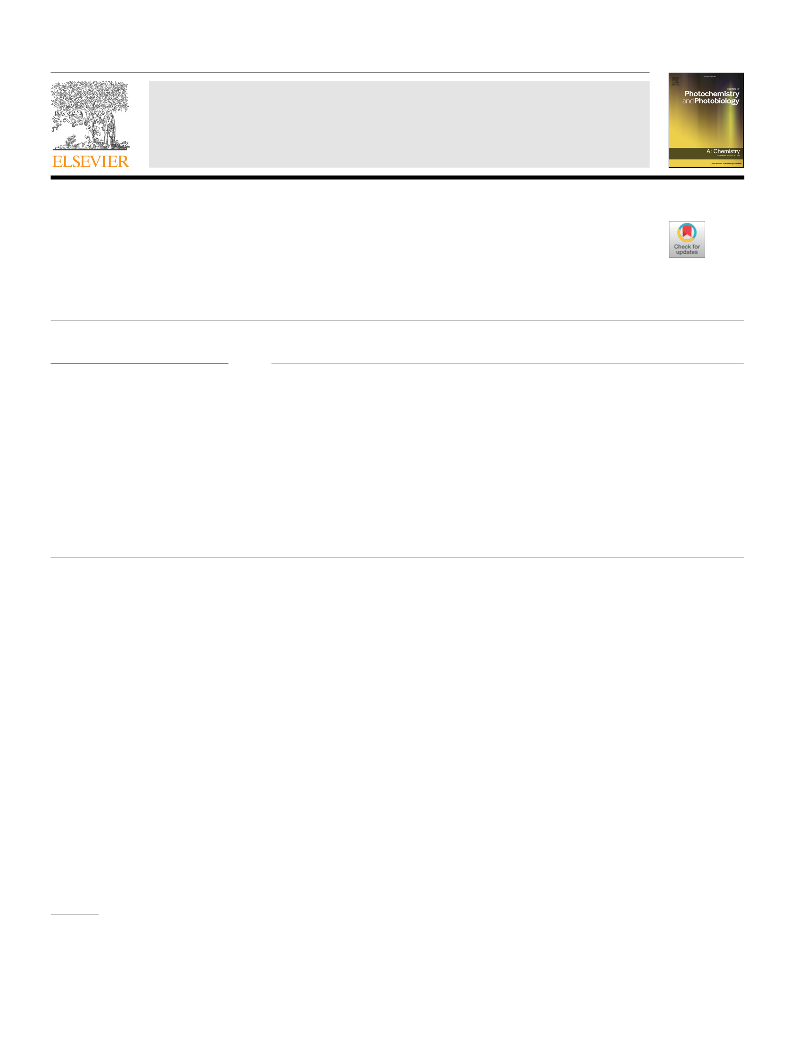
DCMN as an acceptor. All the dyes showed positive solvatochromism
from non-polar (toluene) to polar (DMF) environment. The polarity
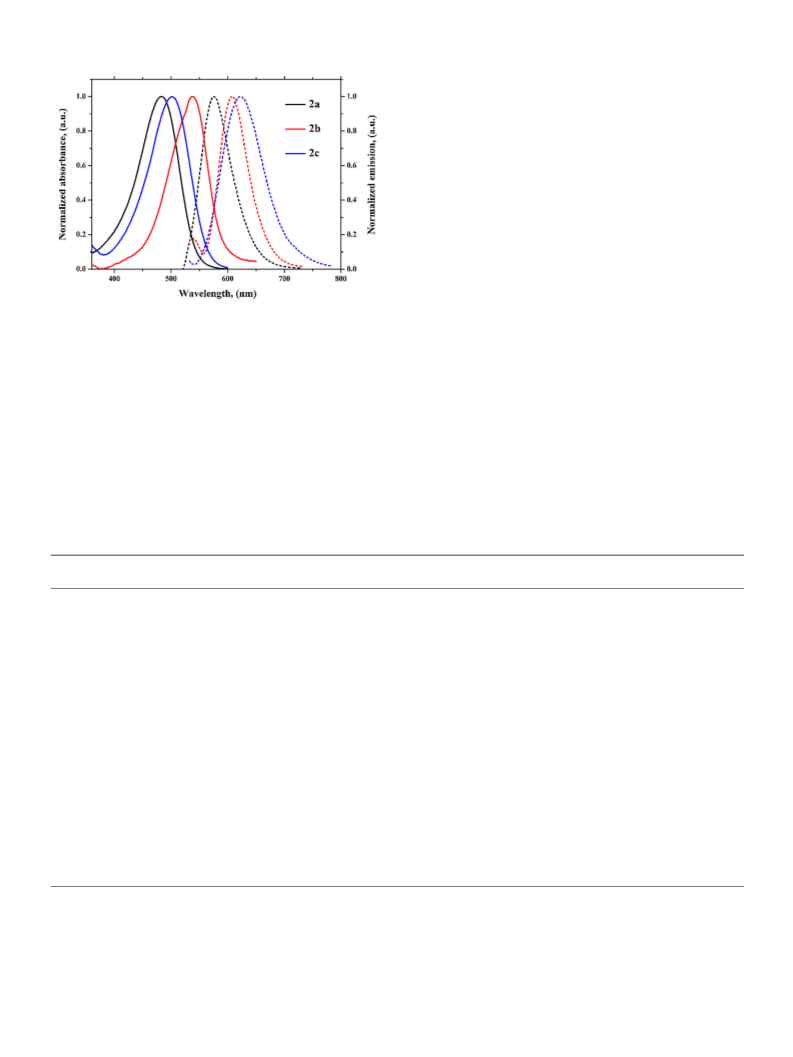
plots drawn on the basis of Lippert-Mataga, McRae and Weller model
confer reasonable sign of charge transfer characteristics whereas, the
Rettig model furnishes TICT state for dyes 2a-c which is been used for
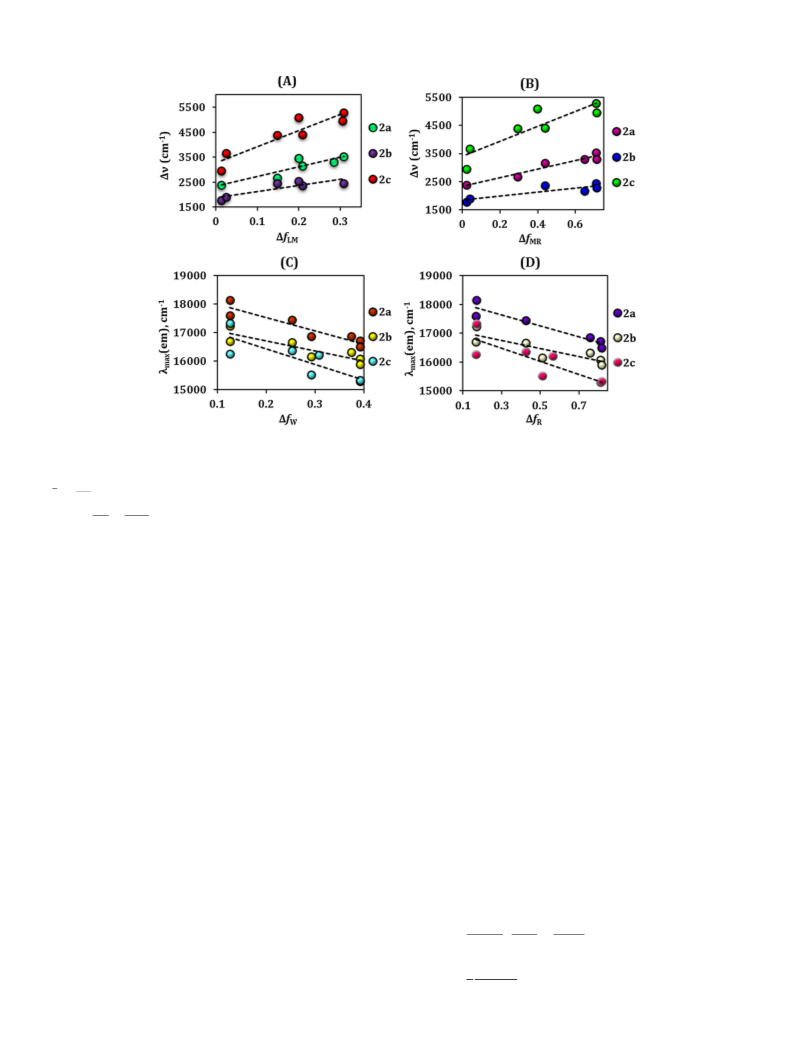
the FMR application. Among all the dyes, 2b was found to have the
higher viscosity sensitivity (x = 0.50) as compared to other dyes as well
as traditional FMR (DCVJ, x = 0.41) dye. The DFT calculations re-
ported here deliver
a significantly increased fundamental under-
standing of dyes 2a-c. On the other hand, the polarizability and hy-
perpolarizability values estimated for dyes 2a-c were found be superior
than that of urea which makes the dyes suitable for the non-linear
optical materials.
[20] T.A. Alhujran, L.N. Dawe, P.E. Georghiou, Synthesis of functionalized ace-
naphthenes and a new class of homooxacalixarenes, Org. Lett. 14 (2012)
[21] H.W.W. Ehrlich, A refinement of the crystal and molecular structure of ace-
[22] Chao Xu, Wen-Feng Luo, Li-Jun Li, Synthesis and enantiomer separation of 5-(1-
(3,5-dinitrobenzoylamino)pent-4-enyl)acenaphthene, Asian J. Chem. 22 (2010)
[23] M. Kumar, S. Roy, M.S.H. Faizi, S. Kumar, M.K. Singh, S. Kishor, S.C. Peter,
R.P. John, Synthesis, crystal structure and luminescence properties of acenaphthene
benzohydrazide based ligand and its zinc(II) complex, J. Mol. Struct. 1128 (2017)
Appendix A. Supplementary data
Supplementary material related to this article can be found, in the
[24] M.P. Cava, K.E. Merkel, R.H. Schlessinger, Pleiadene systems-II : on the mechanism
of acepleiadylene formation - a vinylogous elimination in the acenaphthene series,
[25] G. Felix, M. Laguerre, J. Dunogues, R. Calas, Synthesis of new functional ace-
naphthylene derivatives. 2. Regioselective electrophilic substitution of silylated
acenaphthenes and acenaphthylenes, J. Org. Chem. 47 (1982) 1423–1427, http://
[26] D.A. Evans, L.M. Lee, I. Vargas-Baca, A.H. Cowley, Photophysical tuning of the
aggregation-induced emission of a series of para-substituted aryl bis(imino)ace-
naphthene zinc complexes, Dalton Trans. 44 (2015) 11984–11996, http://dx.doi.
[27] D.A. Evans, L.M. Lee, I. Vargas-Baca, A.H. Cowley, Aggregation-induced emission of
bis(imino)acenaphthene zinc complexes: photophysical tuning via methylation of
the flanking aryl substituents, Organometallics 34 (2015) 2422–2428, http://dx.
[28] P. Majumdar, J. Mack, T. Nyokong, Synthesis, characterization and photophysical
properties of an acenaphthalene fused-ring-expanded NIR absorbing aza-BODIPY
[29] L. Liu, C. Zhang, J. Zhao, The effect of the regioisomeric naphthalimide acetylide
ligands on the photophysical properties of N^N Pt(ii) bisacetylide complexes, Dalton
[30] A.D. Kitchin, S. Velate, M. Chen, K.P. Ghiggino, T.A. Smith, R.P. Steer, Photophysics
of a dithioester RAFT polymerization agent and the acenaphthenyl model light-
harvesting chromophore, Photochem. Photobiol. Sci. 6 (2007) 853, http://dx.doi.
[31] U. El-Ayaan, A.A.M. Abdel-Aziz, S. Al-Shihry, Solvatochromism, DNA binding, an-
titumor activity and molecular modeling study of mixed-ligand copper(II) com-
plexes containing the bulky ligand: Bis[N-(p-tolyl)imino]acenaphthene, Eur. J.
[32] M. Michalska, K. Grudzień, P. Małecki, K. Grela, Gold(I)-catalyzed formation of
Naphthalene/Acenaphthene heterocyclic acetals, Org. Lett. 20 (2018) 954–957,
[33] M. Shahid, S.S. Razi, P. Srivastava, R. Ali, B. Maiti, A. Misra, A useful scaffold based
on acenaphthene exhibiting Cu2+induced excimer fluorescence and sensing cya-
nide via Cu2+displacement approach, Tetrahedron 68 (2012) 9076–9084, http://
References
[1] M. Blanchard-Desce, R. Wortmann, S. Lebus, J.M. Lehn, P. Krämer, Intramolecular
charge transfer in elongated donor-acceptor conjugated polyenes, Chem. Phys. Lett.
[2] A. Wiessner, W. Ku, T. Fiebig, H. Staerk, Intramolecular charge transfer (ICT) and
solvation of a rigidly linked pyrene / N-methylindolino derivative and related
compounds in linear alcohols, J. Phys. Chem. A 101 (1997) 350–359, http://dx.doi.
[3] K.G. Thorat, A.K. Ray, N. Sekar, Modulating TICT to ICT characteristics of acid
switchable red emitting boradiazaindacene chromophores: perspectives from
synthesis, photophysical, hyperpolarizability and TD-DFT studies, Dye Pigm. 136
[4] S.I. Druzhinin, S.A. Kovalenko, T. Senyushkina, K.A. Zachariasse, Dynamics of ul-
trafast intramolecular charge transfer with 1-tert-butyl-6-cyano-1,2,3,4-tetra-
hydroquinoline (NTC6) in n-hexane and acetonitrile, J. Phys. Chem. A 111 (2007)
[5] L.S. Kocsis, K.M. Elbel, B.A. Hardigree, K.M. Brummond, M.A. Haidekker,
E.A. Theodorakis, Cyclopenta[b]naphthalene cyanoacrylate dyes: synthesis and
evaluation as fluorescent molecular rotors, Org. Biomol. Chem. 13 (2015)
[6] R.D. Telore, M.A. Satam, N. Sekar, Push-pull fluorophores with viscosity dependent
and aggregation induced emissions insensitive to polarity, Dye Pigm. 122 (2015)
[7] H. Qian, M.E. Cousins, E.H. Horak, A. Wakefield, M.D. Liptak, I. Aprahamian,
Suppression of Kasha’s rule as a mechanism for fluorescent molecular rotors and
[8] S. Mohamed, D. Demeter, J.-A. Laffitte, P. Blanchard, J. Roncali, Structure-prop-
erties relationships in triarylamine-based donor-acceptor molecules containing
naphtyl groups as donor material for organic solar cells, Sci. Rep. 5 (2015) 9031,
[9] Y. Wang, C. Shu, E.M. Breitung, R.J. Mcmahon, Synthesis and characterization of
thiazole-containing chromophores for second-order nonlinear optics, J. Mater.
[10] K.Y. Suponitsky, Y. Liao, A.E. Masunov, Electronic hyperpolarizabilities for donor-
acceptor molecules with long conjugated bridges: calculations versus experiment, J.
[12] P.K. Nandi, N. Panja, T.K. Ghanty, Heterocycle-based isomeric chromophores with
substantially varying NLO properties: a new structure-property correlation study, J.
[13] T. Zhu, J. Du, W. Cao, J. Fan, X. Peng, Microenvironment-sensitive fluorescent dyes
for recognition of serum albumin in urine and imaging in living cells, Ind. Eng.
[14] M. Cui, M. Ono, H. Watanabe, H. Kimura, B. Liu, H. Saji, Smart near-infrared
fluorescence probes with donor-acceptor structure for in vivo detection of β-amy-
[34] L.A. Gifford, F.T.K. Owusu-Daaku, A.J. Stevens, Acenaphthene fluorescence deri-
vatisation reagents for use in high-performance liquid chromatography, J.
[35] Y.M. Xie, Y. Deng, X.Y. Dai, J. Liu, L. Ouyang, Y.Q. Wei, Y.L. Zhao, Synthesis and
biological evaluation of novel acenaphthene derivatives as potential antitumor
50

 Mohbiya, Dhanraj R.
Mohbiya, Dhanraj R.
 Mallah, Ramnath R.
Mallah, Ramnath R.
 Sekar, Nagaiyan
Sekar, Nagaiyan