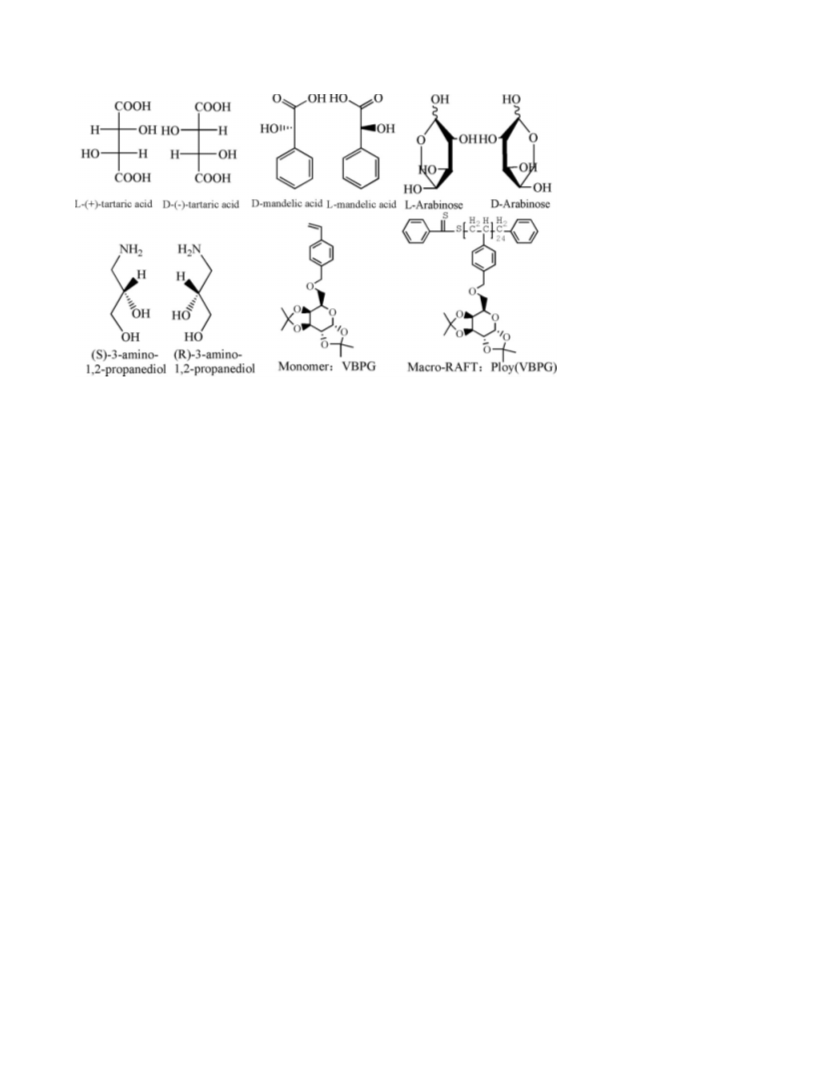
ARTICLE
applied to specific rotation measurement while the chiral
nanoparticles were washed with ethanol three times before
dried for the next use. Each time after separation, we
deducted the lost nanoparticles out then suspended the
remaining in a corresponding amount of ethanol dissolved
D,L-tartaric acid. In this way four separation–abstersion
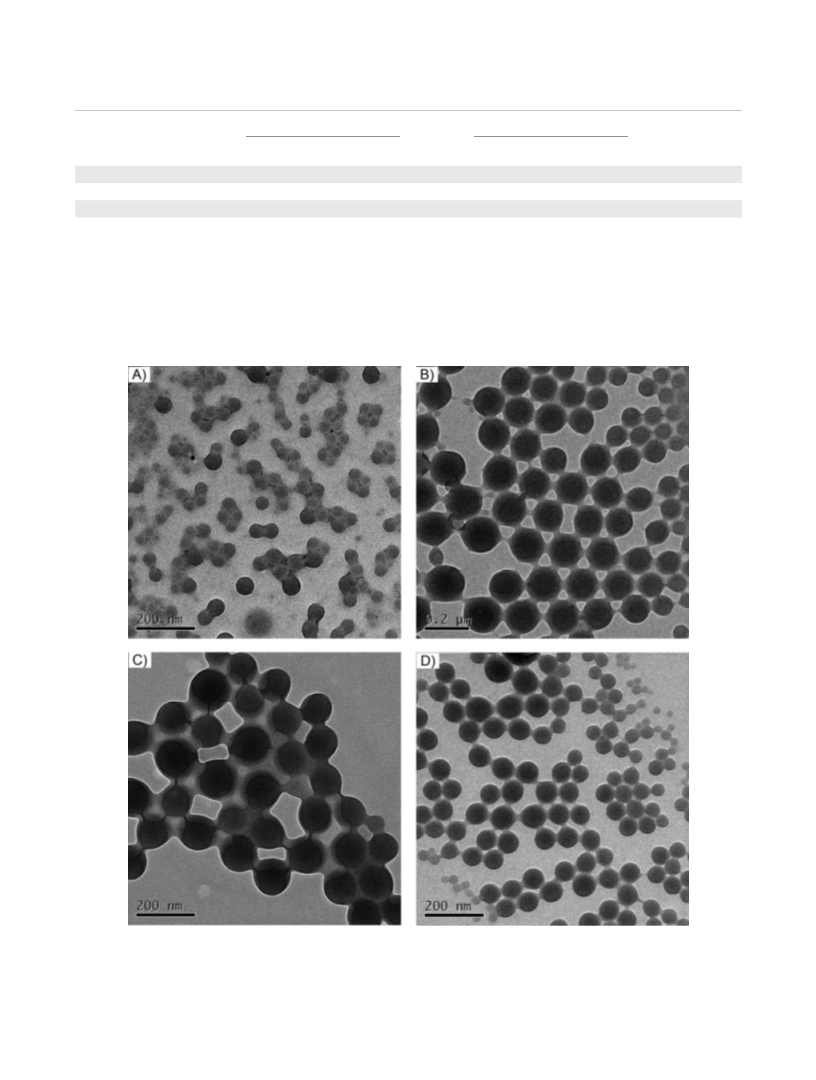
cycles were performed. As shown in Figure 6, the nanopar-
ticles can be reused well; however, the separation ability has
somewhat loss after each cycle. This is attributable to the
fact that the crosslinked nanoparticles could not disperse
well in ethanol solution when dried and reused to next cycle,
due to some of the agglomerations or collapsed nanopar-
ticles was occurred while most of the nanoparticles still kept
the same morphology after four times of chiral separation,
which resulted in some loss of the specific area.
6 Hoffmann, C. V.; Pell, R.; L a¨ mmerhofer, M.; Lindner, W. Anal
Chem 2008, 80, 8780–8789.
7
Okamoto, Y. J Polym Sci Part A: Polym Chem 2009, 47,
731–1739.
1
8
Ikai, T.; Okamoto, Y. Chem Rev 2009, 109, 6077–6101.
9
Yamamoto, C.; Okamoto, Y. Bull Chem Soc Jpn 2004, 77,
27–257.
2
10 Miyako, E.; Maruyama, T.; Kamiya, N.; Goto, M. J Am Chem
Soc 2004, 126, 8622–8623.
1
1 Cichelli, J.; Zharov, I. J Am Chem Soc 2006, 128, 8130–8131.
2 Horvath, J. D.; Koritnik, A.; Kamakoti, P.; Sholl, D. S.; Gell-
1
man, A. J. J Am Chem Soc 2004, 126, 14988–14994.
3 Behzadi, B.; Romer, S.; Fasel, R.; Ernst, K. H. J Am Chem
Soc 2004, 126, 9176–9177.
1
CONCLUSIONS
1
4 Sciannamea, V.; J e´ r oˆ me, R.; Detrembleur, C. Chem Rev
2
008, 108, 1104–1126.
A novel way for the synthesis of well-defined chiral poly-
(
VBPG) homopolymers and corresponding crosslinked nano-
15 Matyjaszewski, K.; Tsarevsky, N. V. Nat Chem 2009, 1,
particles with different crosslinking density, via RAFT minie-
mulsion polymerization using linear poly(VBPG) as the
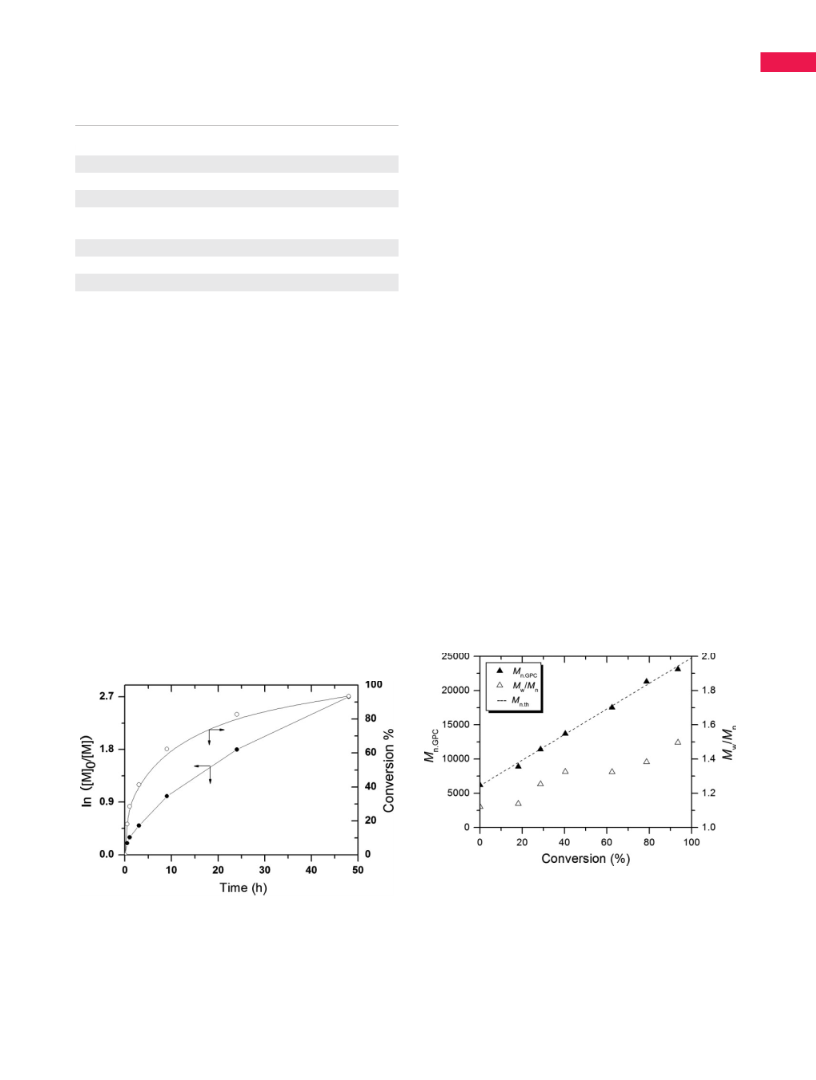
macro-RAFT agent, was demonstrated. The kinetics of RAFT
miniemulsion polymerization of VBPG suggested that the
polymerization proceeded in a living fashion. The amount of
crosslinker had a significant effect on the size and chiral sep-
aration ability of the obtained crosslinked nanoparticles. It
was found that the efficiency of chiral separation was greatly
improved after transferring the crosslinked structure into
the nanoparticles when compared with the chiral linear
chains and that 3–6 wt % of DVB relative to the chiral
monomer VBPG could give the crosslinked chiral nanopar-
ticles better chiral separation ability for the different kinds
of racemates such as D,L-arabinose and (6)-3-amino-1,2-pro-
panediol; if continued to increase the amount of crosslinker
up to 9 wt %, the ability decreased rapidly.
276–288.
1
6 Moad, G.; Rizzardo, E.; Thang, S. H. Polymer 2008, 49,
1
079–1131.
1
7 Zetterlund, P. B.; Kagawa, Y.; Okubo, M. Chem Rev 2008,
1
08, 3747–3794.
1
8 Cunningham, M. F. Prog Polym Sci 2008, 33, 365–398.
1
9 Luo, Y. W.; Tsavalas, J.; Schork, F. J. Macromolecules 2001,
34, 5501–5507.
0 (a) Pham, B. T. T.; Nguyen, D.; Ferguson, C. J.; Hawkett, B.
S.; Serelis, A. K.; Such, C. H. Macromolecules 2003, 36,
907–8909; (b) Lu, F. J.; Luo , Y. W.; Li, B. G. Macromol Rapid
Commun 2007, 28, 868–874.
1 Al-Bagoury, M.; Buchholz, K.; Yaacoub, E. J. Polym Adv
Technol 2007, 18, 313–322.
2
8
2
2
2 Lowe, A. B.; Sumerlin, B. S.; McCormick, C. L. Polymer
The financial supports of this work by the National Natural Sci-
ence Foundation of China (Nos. 20874069, 50803044,
2
003, 44, 6761–6765.
2
3 Okada, M. Prog Polym Sci 2001, 26, 67–104.
2
0904036, and 20974071), the Specialized Research Fund for
the Doctoral Program of Higher Education contract grant (No.
00802850005), the Program of Innovative Research Team of
24 Wulff, G.; Schmid, J.; Venhoff, T. Macromol Chem Phys
2
1996, 197, 259–274.
Soochow University and the Qing Lan Project are gratefully
acknowledged.
2
5 Hazzazi, O. A.; Attard, G. A.; Wells, P. B. J Mol Catal A:
Chem 2004, 216, 247–255.
2
6 Wang, J.; Zhu, X. L.; Cheng, Z. P.; Zhang, Z. P.; Zhu, J. J
Polym Sci Part A: Polym Chem 2007, 45, 3788–3797.
REFERENCES AND NOTES
2
7 (a) Schweitz, L.; Sp e´ gel, P.; Nilsson, S. Analyst 2000, 125,
1
Rmaile, H. H.; Schlenoff, J. B. J Am Chem Soc 2003, 125,
1899–1901; (b) Sp e´ gel, P.; Schweitz, L.; Nilsson, S. Electropho-
resis 2001, 22, 3833–3841.
6602–6603.
2
Crosby, J. Tetrahedron 1991, 47, 4789–4846.
28 (a) Quaglia, M.; De Lorenzi, E.; Sulitzky, C.; Massolini, G.,
Sellergren, B. Analyst 2001, 126, 1495–1498; (b) Quaglia, M.; De
Lorenzi, E.; Sulitzky, C.; Caccialanza, G.; Sellergren, B. Electro-
phoresis 2003, 24, 952–957.
3
Di e´ guez, M.; P a` mies, O.; Claver, C. Chem Rev 2004, 104,
3189–3215.
4
Ward, T. J.; Baker, B. A. Anal Chem 2008, 80, 4363–4372.
2
9 Zhou, X. D.; Ni, P. H.; Yu Z. Q. Polymer 2007, 48, 6262–6271.
5
G u¨ bitz, G.; Schmid, M. G. Biopharm Drug Dispos 2001, 22,
91–336.
30 Paci, I.; Szleifer, I.; Ratner, M. A. J Am Chem Soc 2007, 129,
2
3545–3555.
CROSSLINKED CHIRAL NANOPARTICLES, XU ET AL.
1331

 Xu, Wenliang
Xu, Wenliang
 Cheng, Zhenping
Cheng, Zhenping
 Zhang, Lifen
Zhang, Lifen
 Zhang, Zhengbiao
Zhang, Zhengbiao
 Zhu, Jian
Zhu, Jian
 Zhou, Nianchen
Zhou, Nianchen
 Zhu, Xiulin
Zhu, Xiulin