3
04
W. K. Yoshito et al.
-
0 9 10 moles for 0.2 g of YSZ, confirming that YSZ is
6
1
For the study of reduction kinetic influence of NiO on
porosity, samples of NiO–YSZ were prepared with corn
starch as pore former. The concentration of pore former
was fixed at 10, 15 and 20 mass% of NiO in the composite.
The powder was mixed in ethanol and oven dried at 80 °C
and then deagglomerated in agate mortar. These samples
were pressed using the same procedure used for the sam-
ples without pores formers.
not easily reduced by H2.
The sintering of composites prepared from small sized
powders (\1 lm) promote no formation of enough open
porosity for percolation of gases through the anode. In this
case, pores formers agents that decompose into gases
during the heat treatment are usually added. Haslam et al.
[
9] presented a study of the influence of pore formers in the
diffusion of H gas in the reducing kinetics of the NiO
2
In the sintering process it was maintained an isotherm of
600 °C for 30 min to ensure that all corn starch had been
decomposed and removed from the sample and finally held
in an isotherm of 1450 °C for 1 h. All sintering stages were
performed in air.
0
to Ni .
There are several methods for preparation of the Ni–
YSZ composite as mechanical alloying [10], gel precipi-
tation [11], combustion [12] and co-precipitation synthesis
[
13]. Among them co-precipitation is a large alternative in
TG/DTG data were obtained using Thermobalance TGA
51 model (Shimadzu), under dynamic air, N and H /N
2
the manufacturing of the anode, because it allows a mixture
of precursors already in the solution, resulting in a mixture
of gel with a high degree of chemical homogeneity that
ensures a uniform distribution of nickel particles in the
ceramics matrix [14]. With regard to these comments, the
purpose of this study is to evaluate the use of powders
obtained by the co-precipitation technique, taking into
account the effect of temperature in each stage of powders
processing and finally the influence of porosity in the
2
2
-
1
mixture atmospheres and heating rate of 10 °C min to
-
1
powders and 20 °C min to ceramics. All experiments
were performed in a platinum crucible.
Mass loss and thermal decomposition of the powders
and corn starch was evaluated under heating up to 900 °C,
dynamic atmosphere of air at a total flow rate of
-
1
50 mL min . The reduction kinetics of powders was
carried out in dynamic 33% H /N mixture atmosphere at a
2
2
0
-1
total flow rate of 100 mL min .
reduction kinetics of NiO to Ni .
The reduction kinetic studies of the ceramic bodies
were performed in dynamic 50% H /N mixture of atmo-
2
2
-
1
Experimental procedure
sphere at a total flow rate of 100 mL min , under heating
up to 900 °C and holding isothermal in this temperature
for 1 h.
Zirconium oxychloride prepared from the caustic fusion of
zircon sand (IPEN, Brazil), yttrium chloride (prepared by
hydrochloric acid dissolution of 99,99% Y O —Aldrich
Powders were characterized by X-ray diffraction
(DMAX 2000, Rigaku) for crystal structure analysis,
scanning electron microscopy (SEM) (XL30, Phillips) for
particle and agglomerate morphology observation, trans-
mission electron microscope (TEM) (200-C, JEOL) for the
morphology and particle size evaluation, laser diffraction
(granulometer 1064, Cilas) for agglomerate size distribu-
tion determination, N2 gas absorption technique (BET)
(Nova 1200, Quantachrome) for measurement of surface
area and Helium pycnometry (Micromeritics, AccuPyc
1330) for density measurement.
2
3
Chemical Co, USA and nickel chloride (obtained by
aqueous dissolution of 98% nickel chloride hexahydrate—
Merck) were the metallic ions precursors. Ammonium
hydroxide (CAAL Brazil) was the precipitating agent.
The relative amounts of yttrium and zirconium were
chosen to produce a final composition corresponding to
8
mol% yttria stabilized zirconia and the concentration of
NiO was set for a final composition of the powders of NiO–
YSZ with 56 mass% of NiO.
The coprecipitation reactions were carried out by
spraying the mixed metal chlorides solution (heated at
The porosity of the samples after the reduction was
determined by the immersion method described in ASTM
C20-00 [16].
95 °C), into vigorously stirred ammonium hydroxide
solution. The precipitates were filtered in vacuum and
-
washed with distilled water to remove Cl ions. To avoid
the formation of hard agglomerates the precipitates were
washed with ethanol, followed by an azeotropic distillation
with butanol. The resulting powders were oven-dried at
Results and discussion
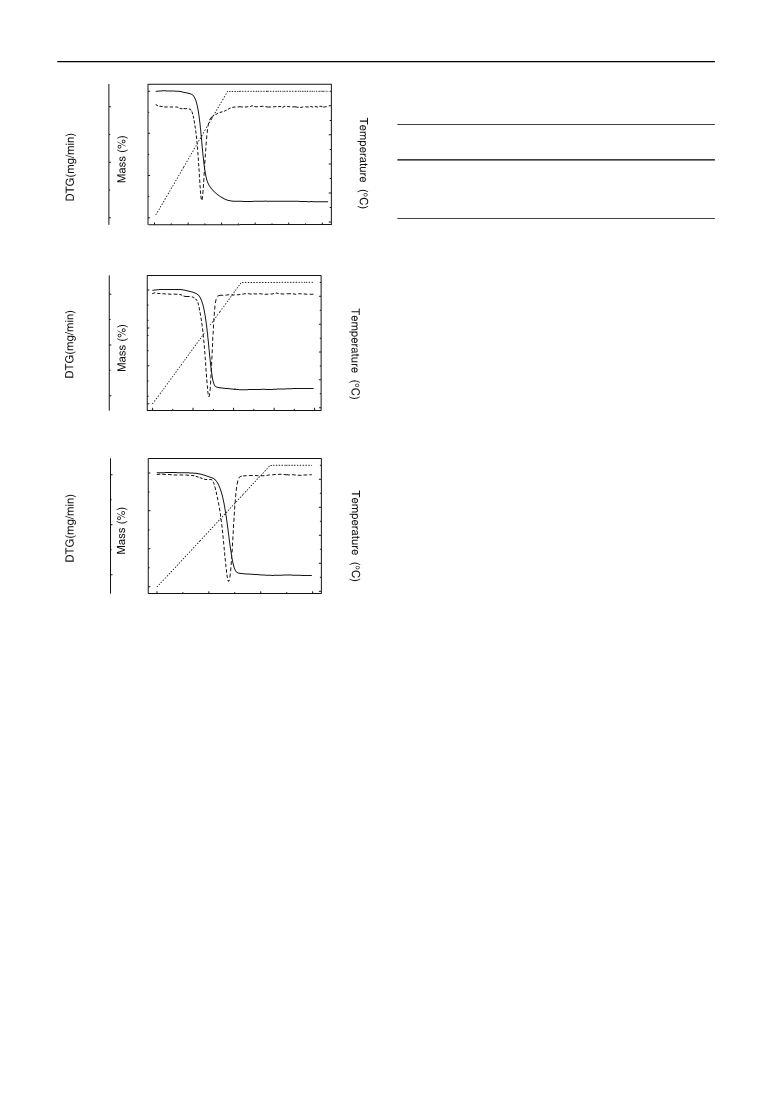
Figure 1 shows the thermal decomposition of powders
synthesized by co-precipitation. Two different stages were
observed: dehydration and further decomposition to NiO–
YSZ. The first mass loss event corresponds to the removal
of free water molecules and organic solvents, occurs slowly
and gradually between 25 and 280 °C. The second event
80 °C for 24 h, calcined at 800 °C for 1 h and milled in
ethanol for 15 h using zirconia balls [15].
Powders were uniaxially pressed at 100 MPa as cylin-
drical pellets of 15 mm diameter and 1 mm height and
sintered in air at 1450 °C for 1 h.
1
23

 Yoshito
Yoshito
 Matos
Matos
 Ussui
Ussui
 Lazar
Lazar
 Paschoal
Paschoal