Please do not adjust margins
Organic & Biomolecular Chemistry
Page 8 of 10
ARTICLE
Journal Name
(100 MHz, CDCl3) δ 192.4, 136.5, 134.7, 134.8, 132.7, 129.6,
129.2, 128.0, 126.9, 122.8.
Requirements for Registration of Pharmaceuticals for
DOI: 10.1039/D0OB01790F
Human Use (ICH): Geneva, Switzerland, October 2006.
(a) P. Venkatesan and K. J. Valliappan, Pharm. Sci. & Res.,
2014, 6, 254-259 and references cited therein; (b) S. B.
Bari B. R. Kadam,Y. S. Jaiswal and A. A. Shirkhedkar,
Eurasian. J. Anal. Chem., 2007,2, 32-53.
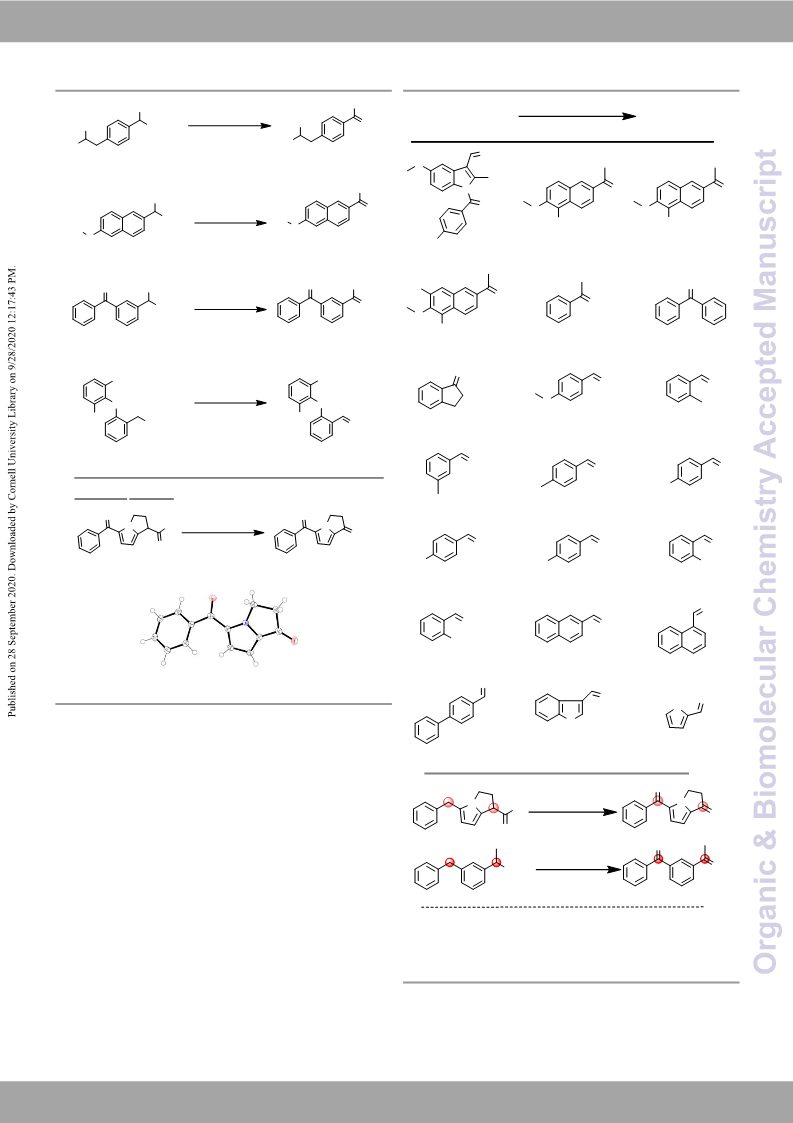
1-Naphthaldehyde (24).5a It was synthesized from 1-
Naphthaleneacetic acid by following an analogous procedure
described for 2. Thick colorless oil. Yield: 0.129 g, 77%. IR
2
1
(neat) 1740, 1515, 1214, 1058, 964 cm-1. H NMR (400 MHz,
CDCl3) δ 10.33 (s, 1H), 9.24 (d, J = 8.6 Hz, 1H), 8.00 (d, J = 8.2
Hz, 1H), 7.86 (dd, J = 12.5, 8.0 Hz, 2H), 7.64 (t, J = 7.7 Hz, 1H),
7.53 (dd, J = 12.0, 7.7 Hz, 2H); 13C NMR (100MHz, CDCl3) δ
193.8, 136.8, 135.3, 133.7, 131.4, 130.5, 129.1, 128.5, 127.0,
124.9.
3
4
P. Shefali and A. Madhavi, Res. J. Pharm. Dosage.
Forms.Tech.,2016, 8, 31-36.
(a) S. Caron, R. W. Dugger, S. G.Ruggeri, J. A. Ragan and D.
H. Brown Ripin, Chem. Rev., 2006, 106, 2943-2989; (b) Y.
Sakakibara, P. Cooper, K. Murakami and K. Itami, Chem.
Asian J., 2018, 13, 2410-2413; (c) T. B. Mete, T. M.
Khopade and R. G. Bhat, Tetrahedron Lett., 2017, 58,
2822-2825.
(a) Q. Feng and Q. Song, J. Org. Chem., 2014, 79, 1867-
1871; (b) Q. Song, Q. Feng and K. Yang, Org. Lett., 2014,
16, 624-627; (c) V. N. Telvekar and K. A. Sasane, Synlett.,
2010, 2778-2779.
(a) S. M. Ataei, Chem. Research., 2000, 148-149; (b) S.;
Tangestaninejad and V. Mirkhani, J. Chem. Res., 1998,
820-821; (c) M. Moghadam, S. Tangestaninejad, V.
Mirkhani, I. Mohammadpoor-baltork, N. Sirjanian and S.
Parand, Bioorg. Med. Chem., 2009, 17, 3394–3398; (d) V.
Mirkhani, S. Tangestaninejad, M. Moghadam and M.
Moghbel, Bioorg. Med. Chem., 2004, 12, 903–906.
(a) V. Mirkhani, S. Tangestaninejad, M. Moghadam, Z.
Karimian, Bioorg. Med. Chem. Lett., 2003, 13, 3433-3435;
(b) V. Mirkhani, S. Tangestaninejad, M. Moghadam, M.
Moghbel, Bioorg. Med. Chem., 2004, 12, 903-906; (c) M.
Moghadam, S. Tangestaninejad, V. Mirkhani, I.
Mohammadpoor-baltrok, N. Sirjanian, S. Parand, Biorg.
Med. Chem., 2009, 17, 3394-3398.
[1,1'-biphenyl]-4-carbaldehyde (25).20 It was synthesized from
4-Biphenylacetic acid by following an analogous procedure
described for 2. Thick colorless oil. Yield: 0.105 g, 61%. IR
5
6
1
(neat) 2911, 2852, 1687, 1366, 831 cm-1. H NMR (400 MHz,
CDCl3) δ 10.01 (s, 1H), 7.90 (d, J = 8.1 Hz, 2H), 7.70 (d, J = 8.3
Hz, 2H), 7.62 – 7.56 (m, 2H), 7.48 – 7.35 (m, 3H); 13C NMR
(100MHz, CDCl3) δ 191.9, 147.2, 139.7, 135.3, 130.3, 129.1,
128.5, 127.7, 127.4.
1H-indole-3-carbaldehyde (26).21 It was synthesized from
Indole-3-acetic acid by following an analogous procedure
described for 2. Dark brown solid. Mp: 201-203 °C. Yield:
0.086g, 52%. IR (neat) 3435, 3203, 2966, 2934, 2878, 1644,
1
1453, 1216, 1055, 966 cm-1. H NMR (200 MHz, DMSO-d6) δ
7
12.19 (s, 1H), 9.98 (s, 1H), 8.32 (d, J = 3.1 Hz, 1H), 8.15 (dd, J =
5.7, 2.6 Hz, 1H), 7.56 (dd, J = 6.0, 2.5 Hz, 1H), 7.39 – 7.14 (m,
2H); 13C NMR (125 MHz, DMSO-d6) δ 185.0, 138.5, 137.1,
124.2, 123.5, 122.2, 120.9, 118.2, 112.5.
Thiophene-2-carbaldehyde (27).4c It was synthesized from 2-
Thiopheneacetic acid by following an analogous procedure
described for 2. Thick greenish oil. Yield: 0.090g, 57%. IR (neat)
3097, 2826, 1654, 1516, 1415, 1208, 1043, 855 cm-1. 1H NMR
(400 MHz, CDCl3) δ 9.83 (d, J = 1.2 Hz, 1H), 7.68 (dd, J = 10.1,
4.5 Hz, 2H), 7.11 (t, J = 8.8 Hz, 1H); 13C NMR (100 MHz, CDCl3) δ
183.2, 144.0, 136.7, 135.3, 128.3.
8
9
P. Gandeepan, T. Müller, D. Zell, G. Cera, S. Warratz, L.
Ackermann, Chem. Rev., 2019, 119, 2192−2452.
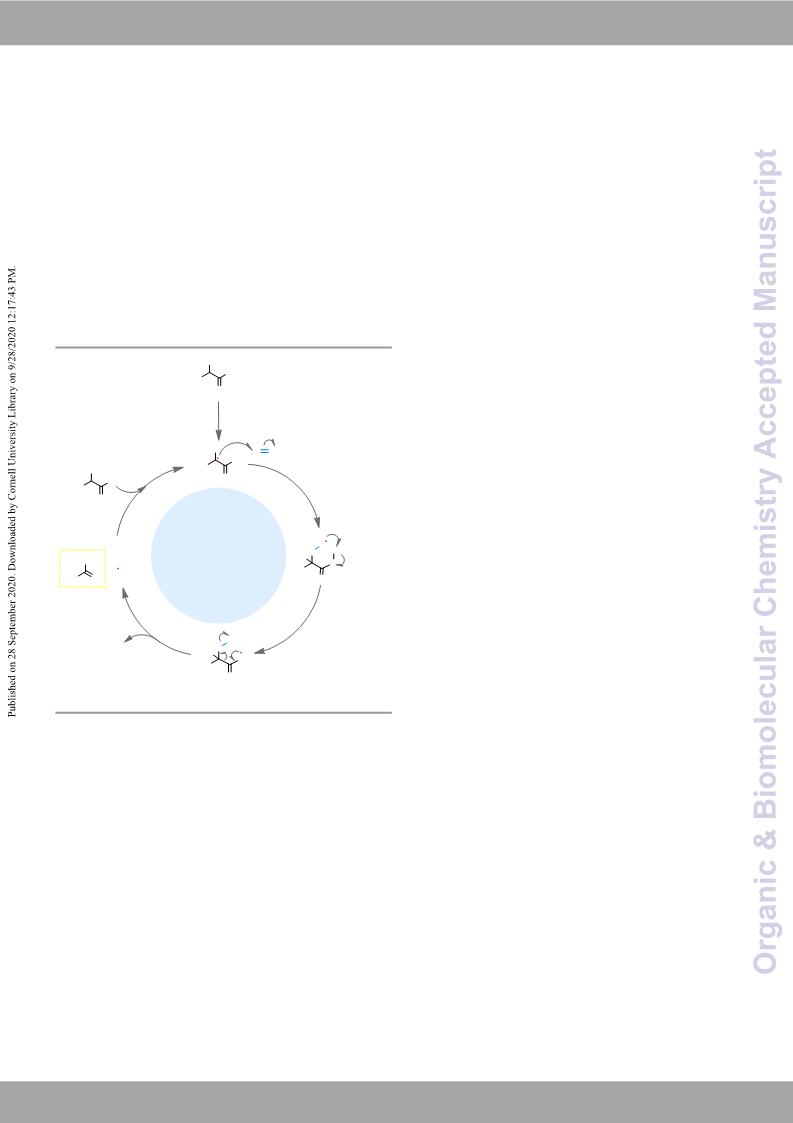
(a) M. L. Neidig, S. H. Carpenter, D. J. Curran, J. C.
DeMuth, V. E. Fleischauer, T. E. Iannuzzi, P. G. N. Neate, J.
D. Sears, N. J. Wolford, Acc. Chem. Res., 2019, 52,
140−150; (b) A. Fürstner, ACS Cent. Sci. 2016, 2, 778−789;
(c) I. Bauer, H.-J. Knölker, Chem. Rev., 2015, 115,
3170−3387; (d) S. Shaik, Nat. Chem., 2010, 2, 347−349;
(e) A. Correa, O. GarcíaMancheño, C. Bolm, Chem. Soc.
Rev., 2008, 37, 1108−1117.
Conflicts of interest
There are no conflicts to declare.
10
11
K. S. Egorova, V. P. Angew. Chem., Int. Ed., 2016, 55,
12150−12162.
(a) N. O’ Connor, M. Geary, M. Wharton, L. Curtin, J. Appl.
Pharm. Sci., 2012, 2, 15-21; (b) S. Orlandini, S.Furlanetto,
S. Pinzauti, G.D’Orazio and S. Fanali, J. Chromatogr A.,
2004, 1044, 295-303; (c) L. Gu, H-S. Chiang and D.
Johnson, Int. J. Pharm., 1988, 41, 105-113. This report
documented the light-induced degradation of ketorolac
where one of the products is oxidative decarboxylation.
However, this method lacks practicality and generality.
(a) C. Bolm, J. Legros, J.Le Paih and L. Zani, Chem. Rev.,
2004, 104, 6217-6254. (b) I. Bauer and H.-J. Knölker,
Chem. Rev., 2015, 115, 3170-3387.
Acknowledgements
We acknowledge the support of Indian Pharmacopoeia
Commission, Govt. of India, Ghaziabad-201002. Dr. Rajesh
Gonnade of the Centre for Materials Characterization, CSIR-
NCL, Pune, for the single crystal X-ray analysis. C.G. thanks
CSIR, New Delhi, for the award of research associate fellowship
(31/11(1087)/2019-EMR-I.)
12
References
1
ICH Q3A(R2) Impurities in New Drug Substances,
8 | J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Gangadurai, Chinnakuzhanthai
Gangadurai, Chinnakuzhanthai
 Illa, Giri Teja
Illa, Giri Teja
 Reddy, D. Srinivasa
Reddy, D. Srinivasa