
Tetrahedron Letters p. 5005 - 5008 (2003)
Update date:2022-08-11
Topics:
 Kim, Geon-Joong
Kim, Geon-Joong
 Lee, Hosung
Lee, Hosung
 Kim, Seong-Jin
Kim, Seong-Jin
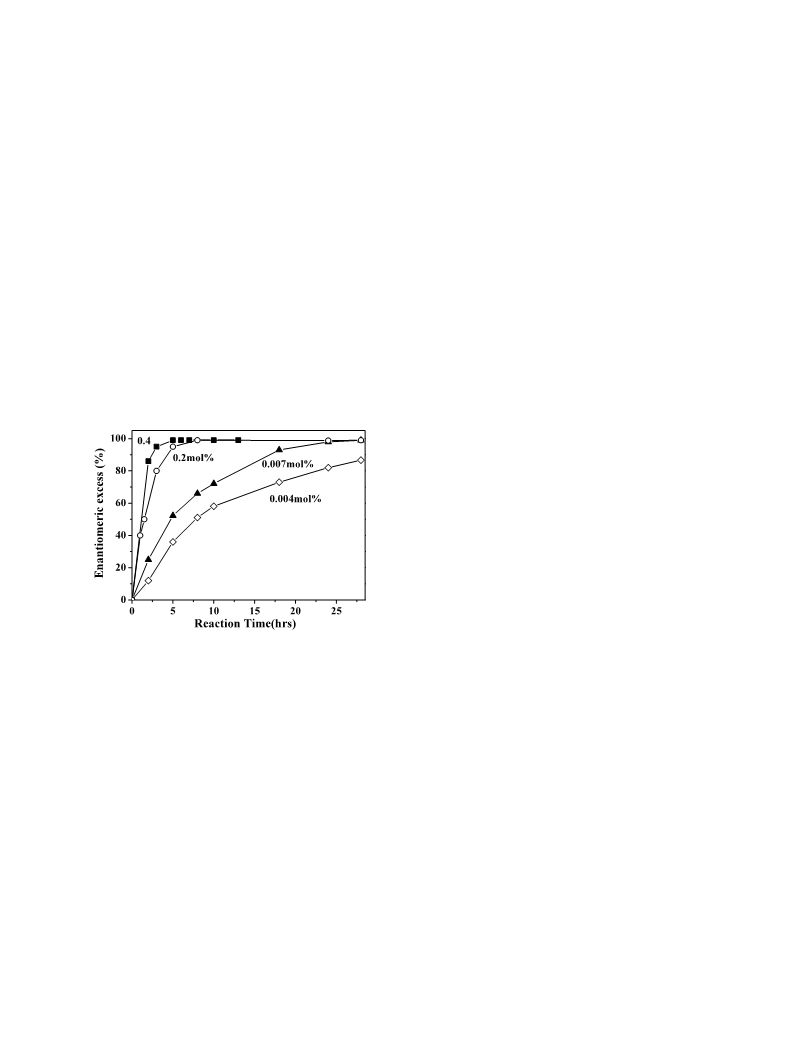
Chiral Co(III) salen catalysts bearing PF6, BF4 or Br counterions proved to be reactive and enantioselective in the hydrolytic resolution of terminal epoxides. The catalysts could be recovered and reused several times without further
View More


Tianjin Anda North Industrial & Business Co.Ltd.
Contact:86-22-24999306
Address:No.11 Erwei Road,Dongli Development Area,Tianjin,China
Contact:0512-62706911
Address:No.289 Yinzhong South Road Wuzhong Economic Development District,Suzhou,China
Weifang Arylchem Chemical Co., LTD
Contact:86-536-5217866
Address:Development Zone, Shouguang, Shandong Province
Hangzhou Dingyan Chem Co., Ltd
website:http://www.dingyanchem.com
Contact:86-571-87157530-8001
Address:RM.1118,NO.1 Building, Baiyun Tower,Jianggan Area, Hangzhou city, China,310004
Zhejiang Haizhou Pharmaceutical Co., Ltd.
website:https://www.haizhoupharma.com/
Contact:+86 576 88221016
Address:No. 19, Donghai 5th Avenue, Yanhai Industrial Zone, Linhai, Zhejiang, China
Doi:10.1021/ic0259890
(2003)Doi:10.1039/c5ob01228g
(2015)Doi:10.1016/j.molliq.2015.06.006
(2015)Doi:10.1016/j.molcatb.2014.07.015
(2014)Doi:10.1002/anie.202004017
(2020)Doi:10.1021/jm00254a015
(1974)