OXIDATIVE ALKOXYLATION OF PHOSPHINE IN ALCOHOL SOLUTIONS OF COPPER
1543
tion to phosphonite PH(OR)2, trialkylphosphite
Р(OR)3, dialkylphosphite P(O)H(OR)2, and phos-
phate Р(O)(OR)3 proceed in a manner similar to the
formation of phosphinites, but more quickly. Replac-
ing the H atoms with OR-groups facilitates the transi-
tion of a P-atom from the 3p3 to the 3sp3 configuration
and leads to sharp increases in the basic properties of
the P-atom, the acidity of P–H bonds, the polarity of
P(III) derivatives, and the tendency to form com-
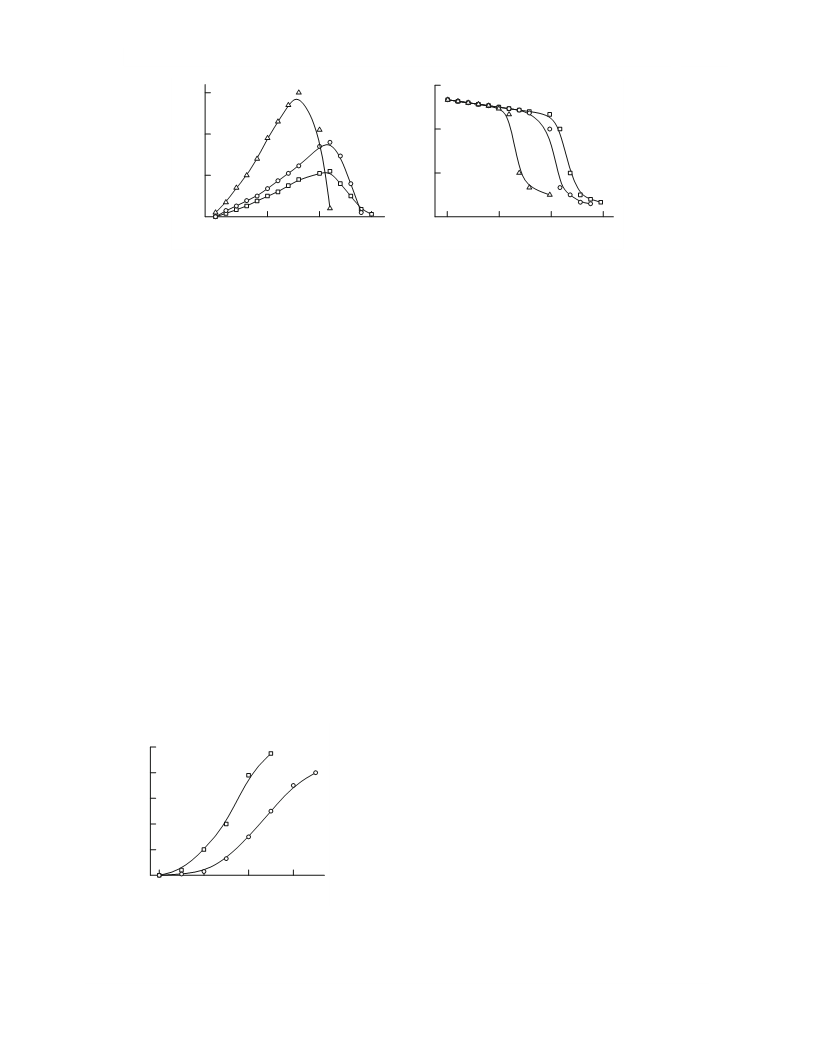
plexes. Primary alcohols primarily form trialkylphos-
phates; secondary alcohols, dialkylphophites, which
do not undergo further oxidation to trialkylphos-
phates. The formation of P–Cu(I) bonds with di-
isoalkylphosphites are hindered by relatively weak
acidity of P–H bonds and the large size of the alkyl
group of the alcohols.
The coordination mechanism is favored by low
activation energies, compared to those of the breaking
Р–Н (322 kJ/mol) and Cu–OR (260 kJ/mol) bonds,
and by negative entropies of activation. The driving
forces behind the reactions of inserting PH3 along
Cu–OR bonds are the high energy of the forming
P‒O and P=O bonds (360 and 612 kJ/mol, respec-
tively) and the substantial redox potential of the
Cu(II) to Cu transition (0.34 V).
Copper (0), which forms as a result of the redox
decomposition of the intermediate complexes, is rap-
idly oxidized by Cu(II) to Cu(I). The presence of oxy-
gen in the PH3–Ar gas mixture promotes the regener-
ation of Cu(II) and allows us to conduct the process
continuously with accumulation of the target product.
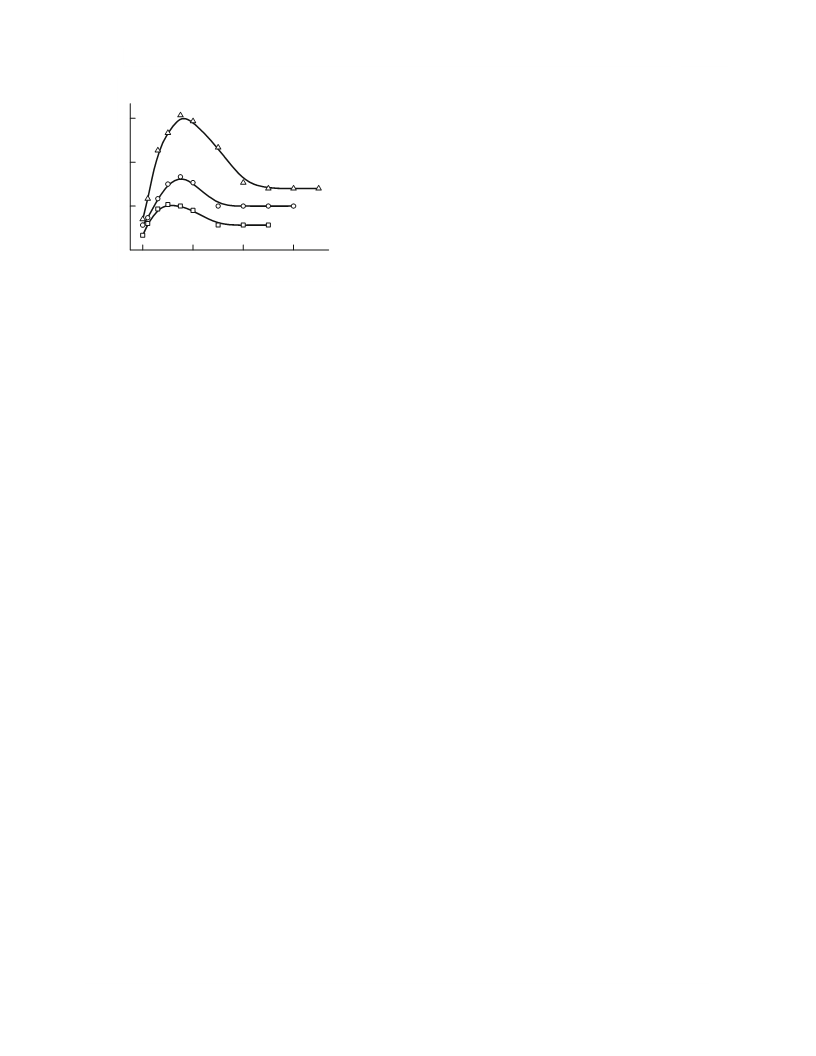
In an atmosphere of oxygen, the absorption curves of
phosphine are characterized by a maximum with a
transition to the stationary mode in terms of both the
rate (Fig. 4) and the potential of the reaction solution.
Reactions (1) and (2) can be conducted periodically
by first feeding the PH3–Ar mixture into the reactor,
and then oxygen.
W × 104, mol/(L min)
3
9
6
2
1
3
0
0
10
20
30
Q × 102, mol/L
Fig. 4. Oxidation of phosphine with oxygen in a butanol
solution of copper chloride: [BuOH] = 10.9 mol/L, [PH ] =
3
0.012 vol %, [O ] = 2.8 vol %, 25°C; [CuCl ]: (1) 0.9,
2
2
(2) 1.4, and (3) 1.9 mol/L.
2−
phides of copper,
[15]. PH3 is activated as
CuX2PH2
a result of the formation of these complexes: the bond
angle grows from 93.8 to 160°, the dipole moment rises
from 0.5 to 1.5 D, the p-character of the lone pair of
electrons increases, and a negative effective charge
appears on the P-atom. Unlike phosphine, phosphi-
des react with alkoxyhalides CuX(OR) to form the
mixed binuclear metal complex [X2Cu–PH2–CuH
(OR)], which facilitates electron delocalization and
the transfer of the
−-ion from the Cu(I) atom to the
PH2
Cu(II) atom. The product-defining complex haloge-
−
nalkoxyphophide of copper (II)
forms
CuХ(OR)PH2
as a result of the transmethylation reaction. The reac-
tion proceeds quickly through the bridging ligand,
resulting in an exchange of electrons between the
π-orbitals of complexes Cu(I) (d10) and Cu(II) (d9).
The high rate of transmethylation is due to the
biphility of the P-atom and the character of single-
electron exchange. In the coordination sphere of
Cu(II), the low-polar PH3 molecule becomes a strong
acceptor as a result of the directed weakening of the
P–H bond, and is capable of the electrophilic bonding
of coordinated alkoxy ions. The limiting step of the
reaction is the oxidative heterolysis of P–Cu-bonds, in
which the transfer of two electrons occurs with the for-
mation of phosphinites PH2(OR) and copper (0).
There is no homolytic decomposition, since the addi-
tive of radical inhibitor benzoquinone does not inter-
fere with the process. The oxidative heterolysis of P–
Cu-bonds is facilitated in the presence of X−-ligands,
which form complexes with copper. The multielectron
oxidation of low valent phosphorus P (−3; 0) proceeds
through a series of energetically advantageous two-
electron redox reactions; as a result, phosphorus forms
stable intermediate compounds Р–, Р+, Р3, and Р5 [16,
17]. Subsequent steps of phosphinite PH2(OR) oxida-
To estimate the thermodynamic probability of pro-
cesses (1), (2) in the system CuX2–РН3–ROH–O2
the values of Gibbs free energy (ΔG°, kJ) of individual
redox stages of the proposed mechanism were calcu-
lated using the equation
°
°
(6)
ΔG° = −zeF(Eox − Ered),
where ze is the number of electrons transferred from
the reducing agent to the oxidizing agent in the reac-
°
Eox
°
tion;
is the standard potential of the oxidant;
Ered
is the standard potential of the reducing agent; and F
is the Faraday constant (96485 C). The reference val-
ues of the standard redox potentials in aqueous solu-
tions without considering the effect of the organic
medium were used in our calculations (Table 2).
Being strong two-electron oxidizing agents, chlo-
rides and bromides of Cu(II) are almost equally capa-
ble of oxidizing phosphorus compound Р−3, Р+, Р3
[16, 17]. The oxidation step of Cu(0) to Cu(I) is faster
RUSSIAN JOURNAL OF PHYSICAL CHEMISTRY A Vol. 90 No. 8 2016

 Polimbetova
Polimbetova
 Borangazieva
Borangazieva
 Ibraimova, Zh. U.
Ibraimova, Zh. U.
 Bugubaeva
Bugubaeva
 Keynbay
Keynbay