Catalysis Science & Technology
Paper
Table 3 Catalytic performance characteristics of cyclohexane oxidation
over a variety of different catalyst materials in the presence of a radical
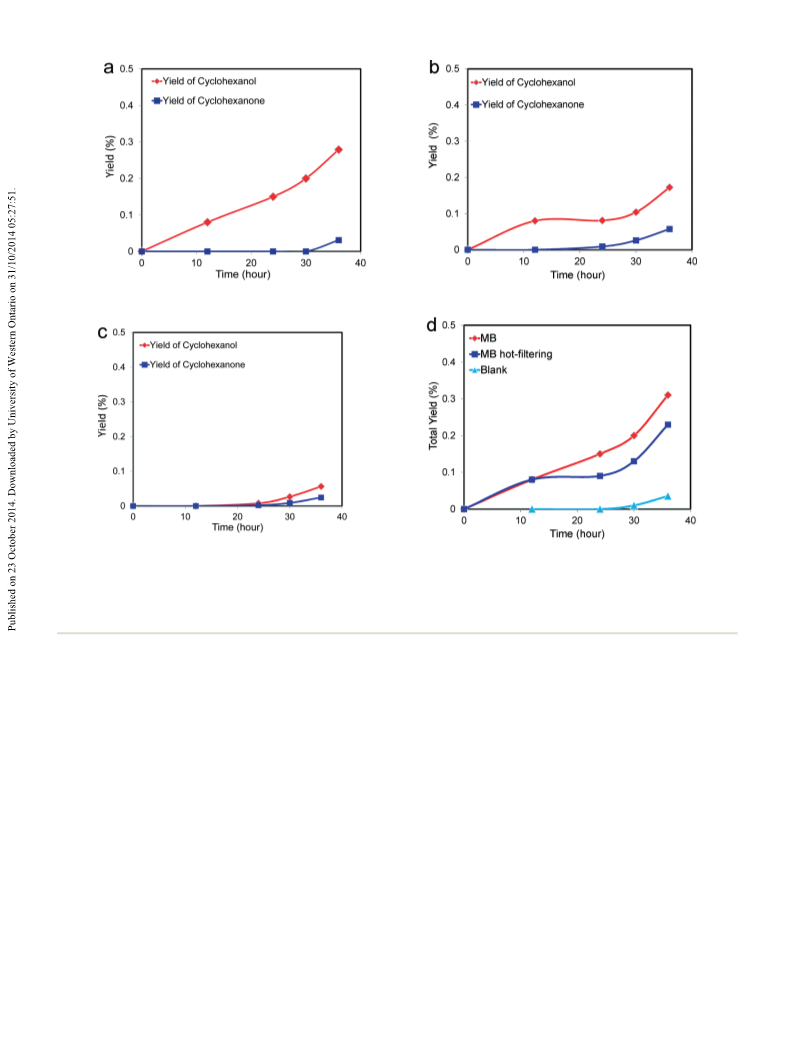
catalytic activity was observed, and the conversion remained
unmodified at ca. 0.08%. The hot filtration experiment
therefore provides further evidence that the presence of MB
plays a key role in the catalytic oxidation of cyclohexane. As
no further reaction was observed in the absence of the MB
catalyst, it is clear that MB is required to observe conversion,
and hence the reaction that is observed is not due to an
underlying homogeneously catalysed pathway from any
solubilised Mo species. On the other hand the apparent
similarities between oxidation reactions carried out in the
presence or absence of MB (Fig. 9d), i.e. an increase in
activity after ca. 25 h reaction time, can be ascribed to
autoxidation processes which are invariably operating for
these class of reaction. In fact, for our experiments, in the
presence of MB the increase in conversion after 25 h is due
to an increase in alcohol. Whereas in absence of MB the
increase in activity is due to an increase in cyclohexanone
formation.
3
scavenger (CBrCl )
Selectivity (%)
CHHP BCH AA Total
a
Catalyst
MB
Co naphthenate 0.9
FeIJacac)
Conversion (%)
A
K
7
58 26
—
—
—
—
9
—
—
—
—
93
90
47
95
3
5
12
7
75
35
10
3
0.8
6.7
SiMo12
O
40
50 35
a
Reaction conditions: 8.5 g cyclohexane, catalyst (6 mg), 140 °C,
bar O , 17 hour; FeIJacac) represents Fe(III) acetylacetonate. In the
3
2
3
shorthand notation used, A, K, CHHP, AA and BCH represent
cyclohexanol, cyclohexanone, cyclohexyl hydroperoxide, adipic acid
and bromocyclohexane respectively.
3
2
ketone, and we wanted to minimize the possibility of undesir-
able parallel reaction pathways existing in these radical
quenching experiments.
3.2.3 Tests on fresh and thermally treated MB. The
inference from the body of experimental evidence presented
so far (i.e. conversion in presence of scavenger, reusability,
selectivity effect, and hot filtration) is that MB operates the
oxidation of cyclohexane via a different pathway if compared
3
In the presence of CBrCl , we observed that the activity of
MB remains basically unaltered by the addition of the scaven-
ger, whereas the activity of Co-naphthenate and FeIJacac) is
3
severely suppressed, with their conversions reduced by about
one order of magnitude (Table 3). Even more significant, in
presence of the scavenger the selectivity for Co-naphthenate
to FeIJacac)
3
and Co-naphthenate. In particular, Fe- and
Co-based materials are promoters of the autoxidation route,
whereas MB would appear to create a new reaction pathway
by means of a real catalytic process enhancing alcohol forma-
tion. This could have important consequences for obtaining
a catalyst capable of introducing a real selectivity control to
such an important reaction.
3
and Fe(acac) is now shifted towards cyclohexanone as the
major product (ca. 10%), whereas, in case of MB the selectiv-
ity is shifted even more towards the more desirable cyclo-
hexanol ( i.e. from 52% to 58%), which would suggest a real
catalytic control exists in the MB system.
3
.2.2 Control tests for solubilized Mo species. In view of
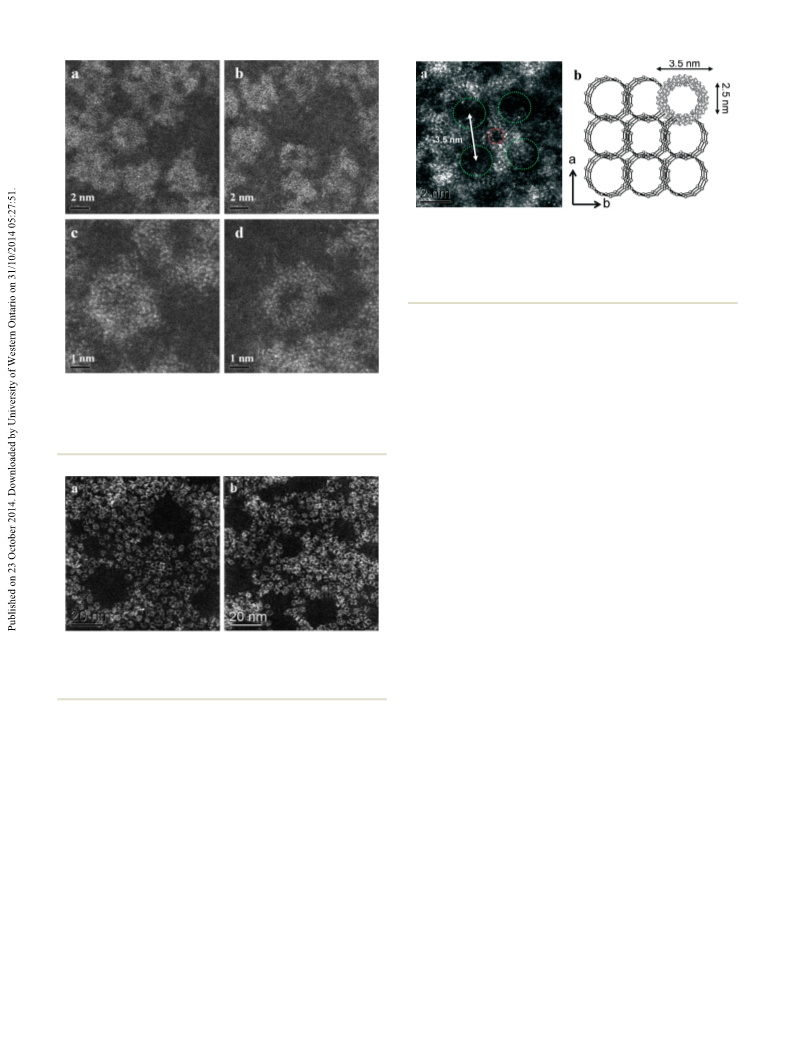
In order to identify the underlying source of this behav-
iour, we have systematically studied the reactivity of MB
nanorings, especially considering the specific structure of MB
described in section 3.1. The first comparison was a control
these data, control tests combining a hot filtration step with
a lower reaction temperature were carried out. The aim here
was two-fold: (i) to further discriminate between a catalytic
route induced by MB, and a pure autoxidation pathway; and
reaction using molybdate silicate blue IJSiMo12
O40) which is
(
ii) to rule out any activity by oligomer molybdenum species
if present. In this experiment (Fig. 9), the catalytic oxidation
of cyclohexane was performed at lower reaction
stable in our reaction environment and also exhibits a mixed
valence Mo –O–Mo structure. The first observation is that
this compound also shows similar conversion values of MB
V
VI
33
a
temperature (i.e. 120 °C instead of 140 °C) in order to
suppress the autoxidation component. As expected the yield
of products was significantly lower (ca. 0.4%), although it
was observed that cyclohexanol was the sole product over a
and also is not quenched by adding CBrCl
3
(see Tables 1 and 3).
Moreover, the SiMo O is also capable of creating a higher
1
2
40
selectivity towards the alcohol, unlike Co-naphthenate and
Fe(acac) , thus suggesting that the structural motif of mixed
valence Mo –O–Mo is important for the catalysis. Although
it should be stressed that this effect for SiMo12 40 is fully
3
V
VI
3
0 h reaction time at the lower temperature (Fig. 9a). This
result confirms that catalytic oxidation is occurring in the
presence of MB, since with an autoxidation pathway both the
alcohol and ketone would be observed. For comparison, a
blank experiment was also conducted using the same
reaction conditions, which showed negligible activity even
after 24 h of reaction (Fig. 9c). After this time both
cyclohexanol and cyclohexanone were formed, which is in
good agreement with the occurrence of autoxidation. Hot
filtration was performed after 12 h reaction of the initial
reaction in order to remove the MB catalyst from the reaction
mixture. Following this, the reaction was continued under
the same reaction conditions. After a further 12 h reaction in
the absence of MB (Fig. 9b), no significant change in
O
evident in presence of radical scavenger only (Table 3).
In fact, it is under these conditions that it is possible to
discriminate between the autoxidation pathway and the
40
activity induced by SiMo12O .
On the other hand, the presence of oxygen in the MB lat-
tice could also play a role in the process to explain this reac-
tivity. In other words, if oxygen is removed from the lattice
V
VI
during the reaction, that in turn disrupts the Mo /Mo
moiety, which could alter the catalytic activity, possibly in
terms of both conversion and selectivity. Therefore, to
demonstrate that the oxygen from the MB structure is playing
a role in the catalytic activity, we have carried out one further
This journal is © The Royal Society of Chemistry 2014
Catal. Sci. Technol.

 Liu, Xi
Liu, Xi
 Conte, Marco
Conte, Marco
 Weng, Weihao
Weng, Weihao
 He, Qian
He, Qian
 Jenkins, Robert L.
Jenkins, Robert L.
 Watanabe, Masashi
Watanabe, Masashi
 Morgan, David J.
Morgan, David J.
 Knight, David W.
Knight, David W.
 Murphy, Damien M.
Murphy, Damien M.
 Whiston, Keith
Whiston, Keith
 Kiely, Christopher J.
Kiely, Christopher J.
 Hutchings, Graham J.
Hutchings, Graham J.