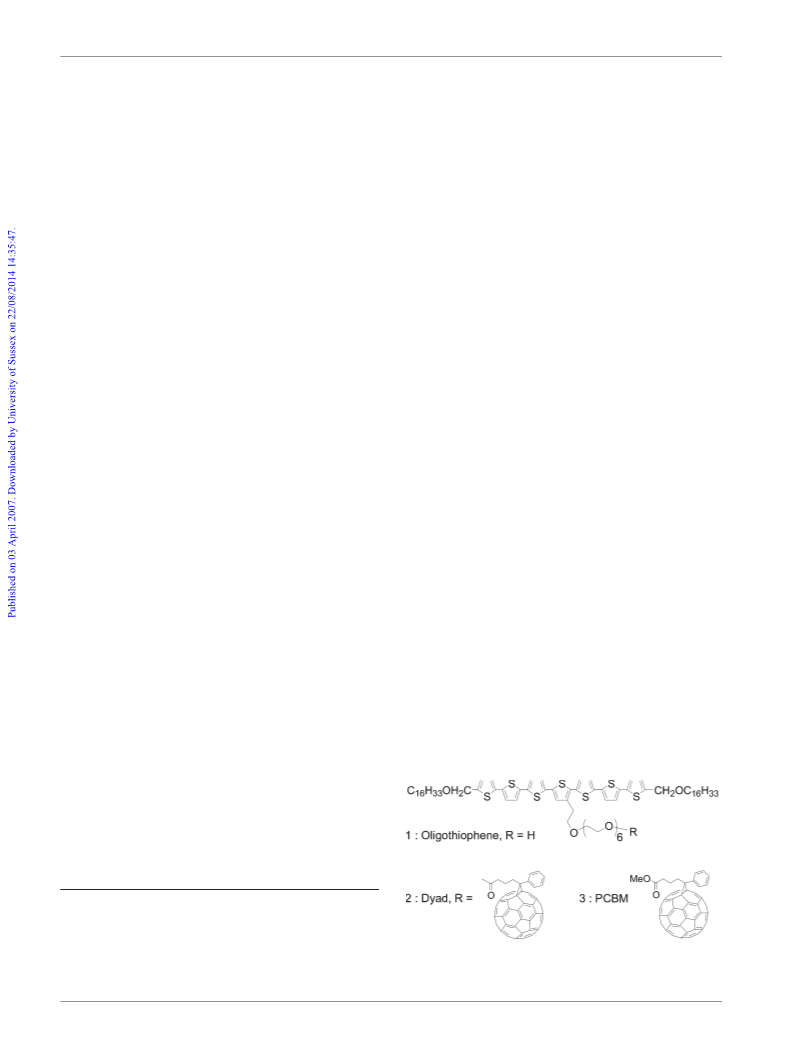
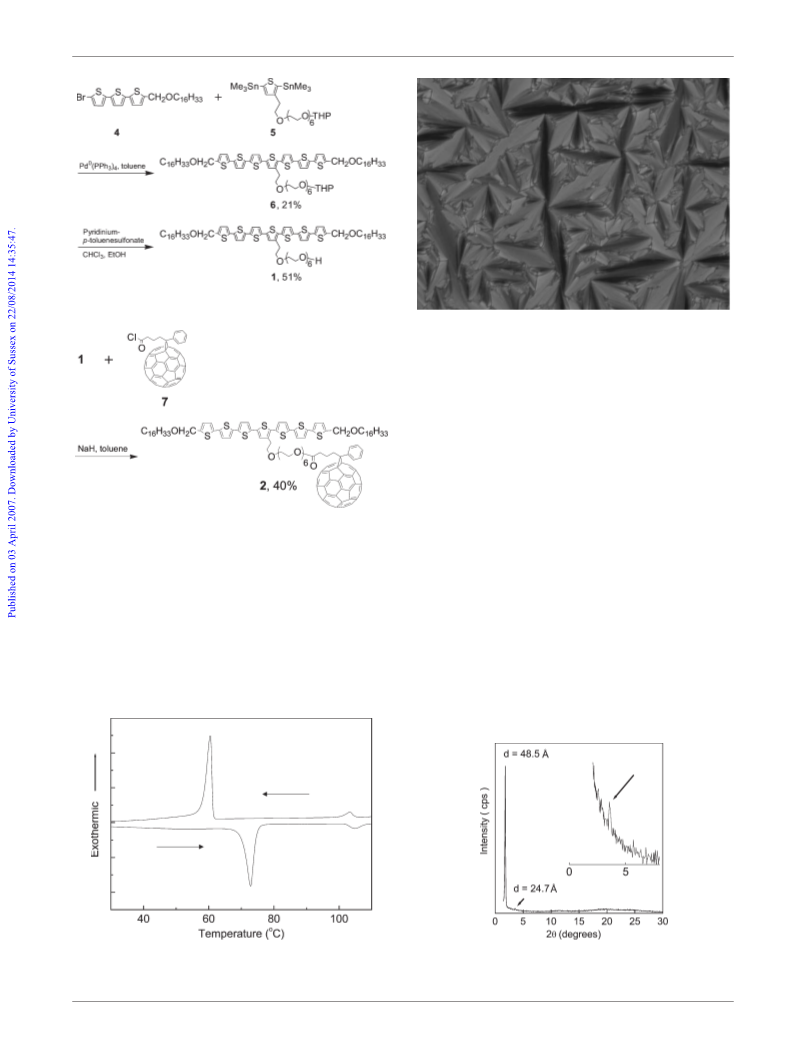
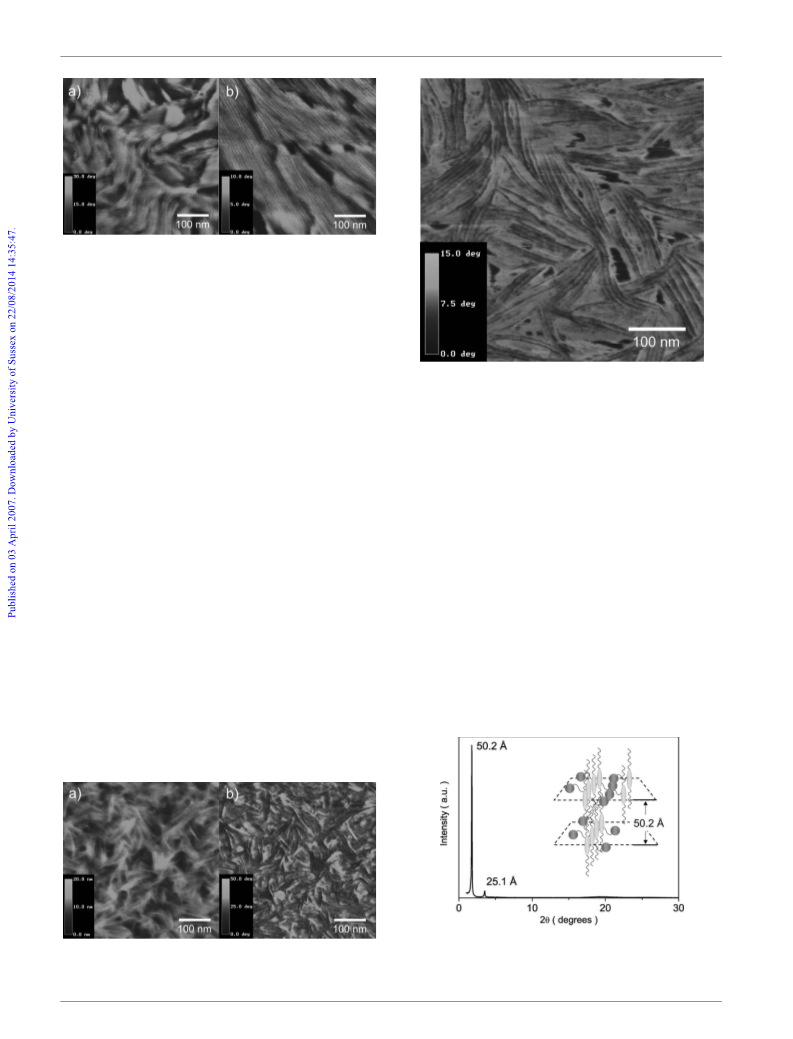
6
7
J. van Herrikhuyzen, A. Syamakumari, A. Schenning and
E. W. Meijer, J. Am. Chem. Soc., 2004, 126, 10021–10027.
A. M. Ramos, S. C. J. Meskers, E. H. A. Beckers, R. B. Prince,
L. Brunsveld and R. A. J. Janssen, J. Am. Chem. Soc., 2004, 126,
J = 7.5 Hz, 2H), 2.20–2.14 (m, 2H), 1.61 (p, J = 6.9 Hz, 4H),
.38–1.23 (m, 52H), 0.88 (t, J = 6.6 Hz, 6H). MALDI TOF-
1
MS m/z: 2270.71 (calc.), 2270.4 (found).
9
630–9644.
8
9
A. Ajayaghosh, R. Varghese, V. K. Praveen and S. Mahesh,
Angew. Chem., Int. Ed., 2006, 45, 3261–3264.
S. J. George and A. Ajayaghosh, Chem.–Eur. J., 2005, 11,
Photovoltaic device preparation and measurement
Photovoltaic devices were prepared with the dyad 2 and the
mixture of oligothiophene 1/PCBM 3 in a structure of
ITO/PEDOT–PSS/2 (or 1/3)/Al. ITO-coated glass substrates
3
217–3227.
1
0 S. J. George, A. Ajayaghosh, P. Jonkheijm, A. Schenning and
E. W. Meijer, Angew. Chem., Int. Ed., 2004, 43, 3422–3425.
1 O. Henze, W. J. Feast, F. Gardebien, P. Jonkheijm, R. Lazzaroni,
P. Leclere, E. W. Meijer and A. Schenning, J. Am. Chem. Soc.,
1
(
R = 10 V per square, Kuramoto Japan) were cleaned by
s
ultra-sonication in detergent, water, acetone, and 2-propanol.
After drying the substrate, PEDOT–PSS (Baytron P) was spin-
coated (4000 rpm) on ITO. The film was dried at 140 uC under
2
006, 128, 5923–5929.
1
2 P. Leclere, M. Surin, P. Viville, R. Lazzaroni, A. F. M. Kilbinger,
O. Henze, W. J. Feast, M. Cavallini, F. Biscarini, A. Schenning and
E. W. Meijer, Chem. Mater., 2004, 16, 4452–4466.
N
2
for 10 min. After cooling the substrate, CHCl
3
solution of
the dyad 2 (16 mg ml ) was spin-coated (2000 rpm). The film
was dried with a N flow for 30 min. For the mixture device,
2
1
13 P. Jonkheijm, N. Stutzmann, Z. J. Chen, D. M. de Leeuw,
E. W. Meijer, A. Schenning and F. Wurthner, J. Am. Chem. Soc.,
2006, 128, 9535–9540.
2
the solution was prepared by dissolving 16 mg of the oligo-
14 D. Hirayama, K. Takimiya, Y. Aso, T. Otsubo, T. Hasobe,
H. Yamada, H. Imahori, S. Fukuzumi and Y. Sakata, J. Am.
Chem. Soc., 2002, 124, 532–533.
thiophene 1 and 1 equivalent (10.5 mg) of PCBM 3 in 1 ml of
CHCl
3
, and the film was fabricated in the same manner.
1
5 H. Imahori and S. Fukuzumi, Adv. Funct. Mater., 2004, 14,
525–536.
Finally, the Al electrode was evaporated on the organic layer
2
4
1
6 E. E. Neuteboom, E. H. A. Beckers, S. C. J. Meskers, E. W. Meijer
and R. A. J. Janssen, Org. Biomol. Chem., 2003, 1, 198–203.
7 C. C. You and F. Wurthner, Org. Lett., 2004, 6, 2401–2404.
8 G. X. Zhang, D. Q. Zhang, X. H. Zhao, X. C. Ai, J. P. Zhang and
D. B. Zhu, Chem.–Eur. J., 2006, 12, 1067–1073.
under high vacuum (6 6 10 Pa). A HAYASHI LA-210UV
xeon lamp and a USHIO SX-UID501CMQ xeon lamp with
an AM 1.5 filter were used for EQE and PCE measure-
ment, respectively. PCE of the devices were estimated at
1
1
2
2
19 F. Langa, M. J. Gomez-Escalonilla, J. M. Rueff, T. M. F. Duarte,
J. F. Nierengarten, V. Palermo, P. Samori, Y. Rio, G. Accorsi and
N. Armaroli, Chem.–Eur. J., 2005, 11, 4405–4415.
1
00 mW cm . The light intensity was adjusted with a silicon
standard solar cell (Bunkou Keiki BS520).
2
0 H. Zhang, F. J. M. Hoeben, M. J. Pouderoijen, A. Schenning,
E. W. Meijer, F. C. Schryver and S. De Feyter, Chem.–Eur. J.,
Acknowledgements
2
1 Y. Morikawa, S. Nagano, K. Watanabe, K. Kamata, T. Iyoda and
T. Seki, Adv. Mater., 2006, 18, 883–+.
22 P. Samori, V. Francke, T. Mangel, K. Mullen and J. P. Rabe, Opt.
Mater., 1998, 9, 390–393.
3 D. M. Guldi, C. P. Luo, A. Swartz, R. Gomez, J. L. Segura,
N. Martin, C. Brabec and N. S. Sariciftci, J. Org. Chem., 2002, 67,
1141–1152.
4 N. Negishi, K. Takimiya, T. Otsubo, Y. Harima and Y. Aso,
Chem. Lett., 2004, 33, 654–655.
25 F. S. Meng, J. L. Hua, K. C. Chen, H. Tian, L. Zuppiroli and
F. Nuesch, J. Mater. Chem., 2005, 15, 979–986.
26 M. A. Loi, P. Denk, H. Hoppe, H. Neugebauer, C. Winder,
D. Meissner, C. Brabec, N. S. Sariciftci, A. Gouloumis, P. Vazquez
and T. Torres, J. Mater. Chem., 2003, 13, 700–704.
006, 12, 9046–9055.
2
We thank Dr K. Hirota for fruitful discussion, Mr Q. S. Wei
for help with AFM measurement, and Rigaku Corporation for
help with the XRD measurement.
2
References
2
1
F. J. M. Hoeben, P. Jonkheijm, E. W. Meijer and A. Schenning,
Chem. Rev., 2005, 105, 1491–1546.
2
3
A. Schenning and E. W. Meijer, Chem. Commun., 2005, 3245–3258.
F. S. Schoonbeek, J. H. van Esch, B. Wegewijs, D. B. A. Rep,
M. P. de Haas, T. M. Klapwijk, R. M. Kellogg and B. L. Feringa,
Angew. Chem., Int. Ed., 1999, 38, 1393–1397.
4
5
V. K. Praveen, S. J. George, R. Varghese, C. Vijayakumar and
A. Ajayaghosh, J. Am. Chem. Soc., 2006, 128, 7542–7550.
X. Q. Li, V. Stepanenko, Z. J. Chen, P. Prins, L. D. A. Siebbeles
and F. Wurthner, Chem. Commun., 2006, 3871–3873.
27 M. Narutaki, K. Takimiya, T. Otsubo, Y. Harima, H. Zhang,
Y. Araki and O. Ito, J. Org. Chem., 2006, 71, 1761–1768.
28 J. C. Hummelen, B. W. Knight, F. LePeq, F. Wudl, J. Yao and
C. L. Wilkins, J. Org. Chem., 1995, 60, 532–538.
This journal is ß The Royal Society of Chemistry 2007
J. Mater. Chem., 2007, 17, 2440–2445 | 2445

 Nishizawa, Takeshi
Nishizawa, Takeshi
 Tajima, Keisuke
Tajima, Keisuke
 Hashimoto, Kazuhito
Hashimoto, Kazuhito