
Phosphorus, Sulfur and Silicon and the Related Elements p. 1886 - 1895 (2010)
Update date:2022-08-18
Topics:
 Bellassoued
Bellassoued
 Aatar
Aatar
 Bouzid
Bouzid
 Damak
Damak
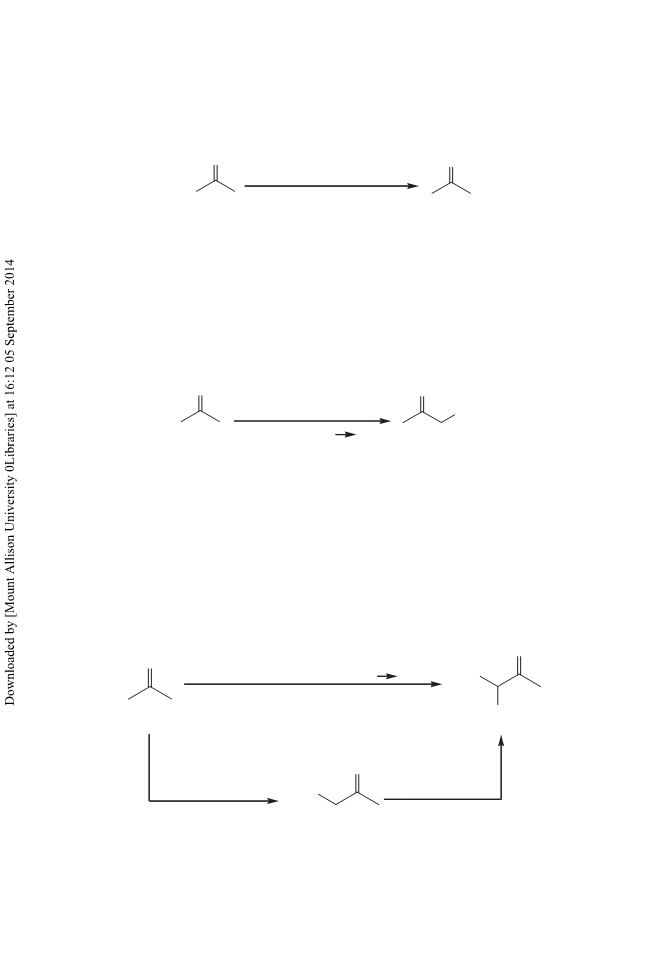
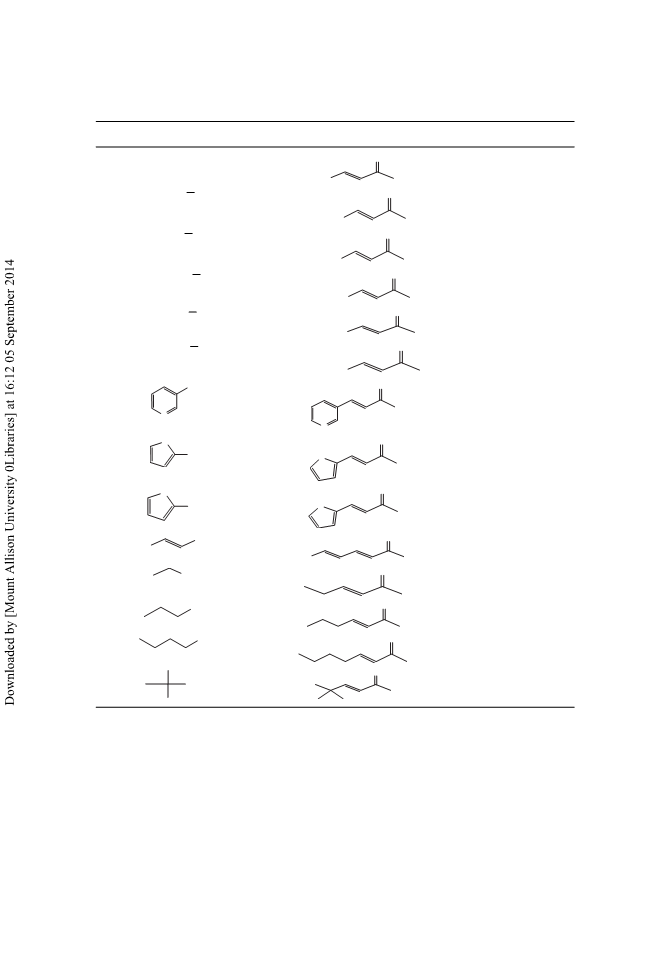
Aldehydes are converted into (E)-α,β-unsaturated methyl ketones in good yield and with a high E stereoselectivity using α,α- bis(trimethylsilyl) N-tert-butyl acetimine 3. The reaction was mediated by a catalytic amount of tetrabutylammonium fluoride (TBAF) under mild conditions. The disilylated reagent 3 is easily generated from N-tert-butylacetimine, lithium diisopropylamide (LDA), and chlorotrimethylsilane. The mechanism of the reaction is discussed. Copyright
View More








Ningbo Tide Imp. & Exp. Co., Ltd.
Contact:+86-571-8993 7933; +86-571-8993 6453
Address:7/F Anno Domini Building, Tower South, 8 Qiu Shi Road,Hangzhou,China.
Shanghai SMEC Trading Co., Ltd.
website:http://www.shanghaismec.com
Contact:+86-21-68811293
Address:ROOM 1101,NO.800 SHANGCHENG RD,SHANGHAI,CHINA
Contact:+86-29-88710656
Address:South Tai bai Road, High Tech Development Zone, Xi'an China
Shandong Loyal Chemical industrial Co.,Ltd
Contact:0533-7451788
Address:Linzi Chemical Industrial Park, Zibo, Shandong Province
Nanjing Capatue Chemical Co., Ltd
Contact:+86-25-86371192 +86-025-85720158
Address:No.20 Jiangjun Avenue, Jiangning Economic & Technical Development Zone
Doi:10.1002/smll.201402472
(2015)Doi:10.1021/jm00128a004
(1989)Doi:10.1021/ic049220u
(2005)Doi:10.1246/bcsj.65.943
(1992)Doi:10.1039/c5ta06548h
(2015)Doi:10.1007/s11743-016-1869-9
(2016)