7
16 Journal of Chemical and Engineering Data, Vol. 51, No. 2, 2006
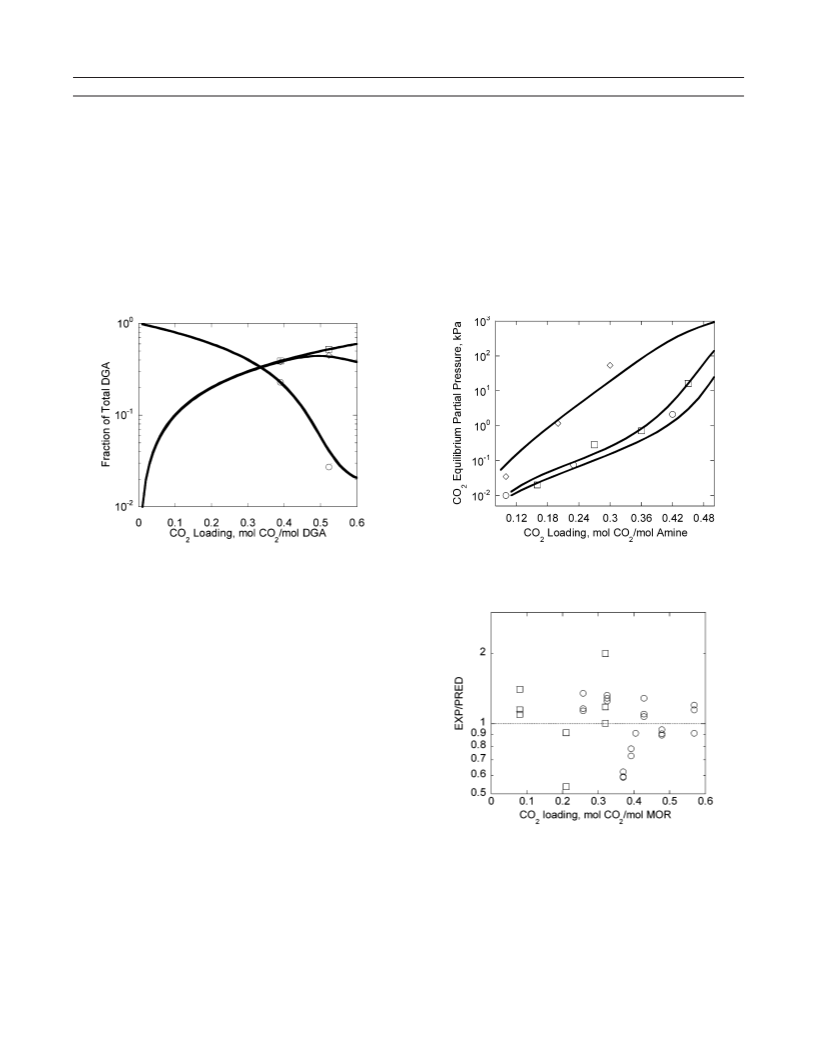
at low loading (< 0.1) in the blend are, however, similar to
those in 17.7 m DGA. This is due to the stability of DGA and
MOR carbamate.
The working capacity of 17.7 m DGA is ∼17 % greater than
the blend. Therefore 17.7 m DGA will require less solvent
circulation than the blend. MOR vapor pressure is ∼100 times
greater than DGA vapor pressure at (313 to 333) K, potentially
resulting in significant losses of MOR by evaporation. The
regeneration energy of 3.6 m MOR + 14.7 m DGA will
probably be less than 17.7 m DGA.
NMR has proven to be a useful technique in quantifying
speciation. The most prevalent reaction product at high loading
(> 0.5 at high loading) is MOR carbamate. The existence of
the protonated DGA has the effect of stabilizing the overall
MOR carbamate formation.
The model presented here is based on experimental results
in 65 wt % amine (17.7 m DGA). Therefore care should be
exercised when extrapolating these results to other amine
concentrations.
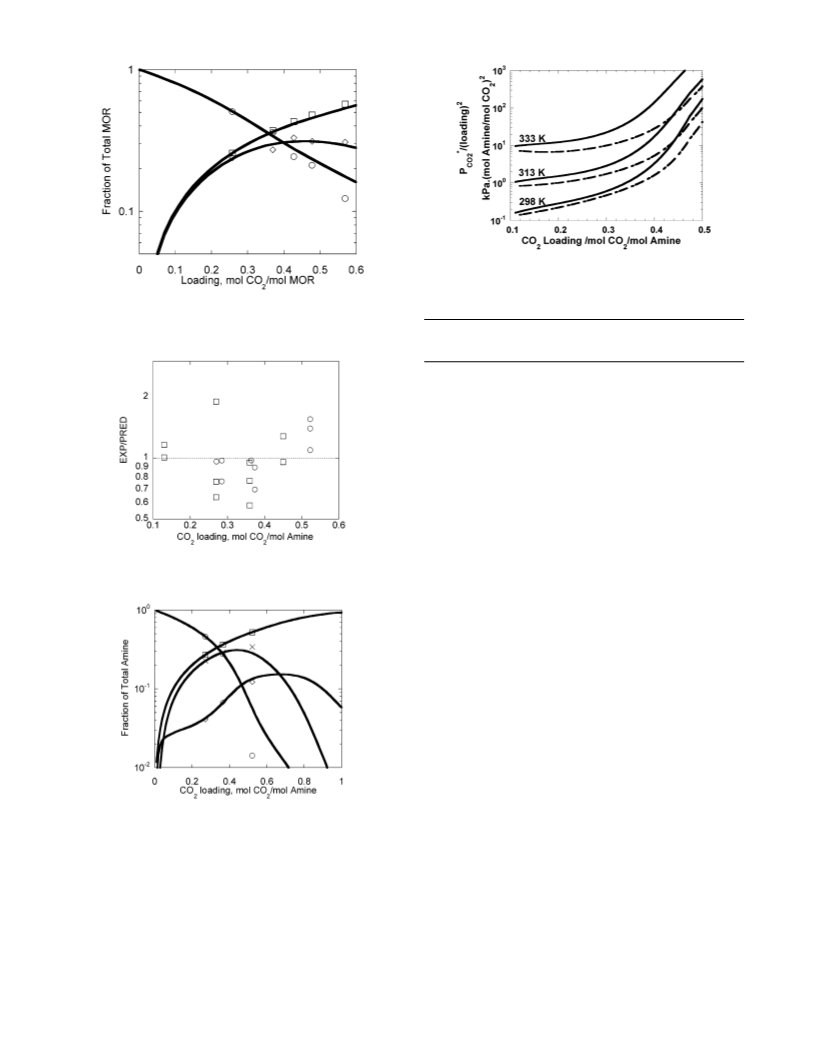
Figure 14. Heat of CO2 absorption at 333 K predicted by the regressed
thermodynamic model. Solid line represents 17.7 m DGA, and dashed line
represents 3.6 m MOR + 14.7 m DGA.
Acknowledgment
Professor Ben Shoulders and the NMR Laboratory at The
University of Texas at Austin Department of Chemistry and
Biochemistry provided support for NMR measurements and
interpretation. Diglycolamine and DGA are both registered trade-
marks.
Literature Cited
(
(
(
1) Harruff, L. G. The origin of morpholine in diglycolamine gas
sweetening plants. First International Conference on Chemistry in
Industry, Manama, Bahrain, 1992.
2) Austgen, D. M. A model for vapor-liquid equilibrium for acid gas-
alkanolamine-water systems. Ph.D. Dissertation, The University of
Texas at Austin, Austin, TX, 1989.
3) Posey, M. L. Thermodynamic model for acid gas loaded aqueous
alkanolamine solutions. Ph.D. Dissertation, The University of Texas
at Austin, Austin, TX, 1996.
Figure 15. Vapor pressure of MOR and DGA over 3.6 m MOR/14.7 m
DGA and CO2 loading of 0.01 mol CO2/mol amine predicted by the
regressed thermodynamic model. Solid line represents MOR, and dashed
line represents DGA.
(4) Pacheco M. A.; Kaganoi, S.; Rochelle, G. T. CO2 absorption into
aqueous mixtures of diglycolamine and methyldiethanolamine. Chem.
Eng. Sci. 2000, 55, 5125-5140.
increasing the loading decreases the heat of absorption. As can
be seen in Figure 14, the heat of absorption of 17.7 m DGA is
slightly higher than that of 3.6 m MOR + 14.7 m DGA. As a
result more energy may be required to regenerate DGA than
the blend solution.
(
5) Glasscock, D. A. Modelling and experimental study of carbon dioxide
absorption into aqueous alkanolamines. Ph.D. Dissertation, The
University of Texas at Austin, Austin, TX, 1990.
(
6) Critchfield, J. E. CO2 absorption/desorption in methyldiethanolamine
solutions promoted with monoethanolamine and diethanolamine: mass
transfer and reaction kinetics. Ph.D. Dissertation, The University of
Texas at Austin, Austin, TX, 1988.
(
7) Bishnoi, S.; Rochelle, G. T. Physical and chemical solubility of carbon
dioxide in aqueous methyldiethanolamine. Fluid Phase Equilib. 2000,
168, 241-258.
Solvent Losses
Vaporization losses are a direct result of alkanolamine vapor
pressure in lean solvent. The predicted vapor pressure of MOR
and DGA over 3.6 m MOR + 14.7 m DGA is presented in
Figure 15. Since total pressures and gas phase compositions
vary widely in industrial absorbers, results are presented as the
vapor side fugacity of MOR. Since the gas leaving the absorber
is in contact with the lean amine solution entering the top of
the column, all calculations are done at a lean loading of 0.01
mol CO2/mol amine. MOR is seen to be very volatile as
compared to DGA.
(
8) Austgen, D. M.; Rochelle, G. T.; Chen, C. C. Model of vapor-liquid
equilibria for aqueous acid gas-alkanolamine systems using the
electrolyte-NRTL equation. Ind. Eng. Chem. Res. 1989, 28, 1060.
(9) Posey, M. L.; Rochelle, G. T. A thermodynamic model of methyldi-
ethanolamine-CO2-H2S-water. Ind. Eng. Chem. Res. 1997, 36,
3944-3958.
(
10) Martin, J. L.; Otto, F. D., Mather, A. E. Solubility of hydrogen sulfide
and carbon dioxide in a diglycolamine solution. J. Chem. Eng. Data
1978, 23, 163.
11) Dingman, J. D.; Jackson, J. L.; Moore, T. F.; Branson, J. A.
Equilibrium data for the H2S-CO2-diglycolamine agent-water
system. Proceedings of the Gas Processors Association Sixty-Second
Annual ConVention, 1983.
(
Conclusions
(12) Versteeg, G. F.; Van Swaaij, W. P. M. Solubility and diffusivity of
acid gases (carbon dioxide, nitrous oxide) in aqueous alkanolamine
solutions. J. Chem. Eng. Data 1988, 33, 29-34.
The carbamate of MOR is less stable than the carbamate of
DGA by a factor of 7 to 10 from (300 to 333) K. The
equilibrium CO2 partial pressure over loaded 3.5 m MOR is
greater that that of 17.7 m DGA and 3.6 m MOR + 14.7 m
DGA. In addition, 3.6 m MOR + 14.7 m DGA has a higher
equilibrium partial pressure of CO2 at high loading by a factor
of 5 to 7 than 17.7 m DGA. This is due to the low carbamate
stability constant of MOR compared to DGA. Partial pressures
(
13) Bishnoi, S. Carbon dioxide absorption and solution equilibrium in
piperazine activated methyldiethanolamine. Ph.D. Dissertation, The
University of Texas at Austin, Austin, TX, 2000.
(14) Smith, W. R.; Missen, R. W. Can. J. Chem. Eng. 1988, 66, 591.
(15) Soave, G. Equilibrium constants from a modified Redlich-Kwong
equation of state. Chem. Eng. Sci. 1972, 27, 1197.
(
16) Chen, C. C.; Evans, L. B. A local composition model for the excess
Gibbs energy of aqueous electrolyte systems. AICHE J. 1986, 32, 444-
454.

 Al-Juaied, Mohammed
Al-Juaied, Mohammed
 Rochelle, Gary T.
Rochelle, Gary T.