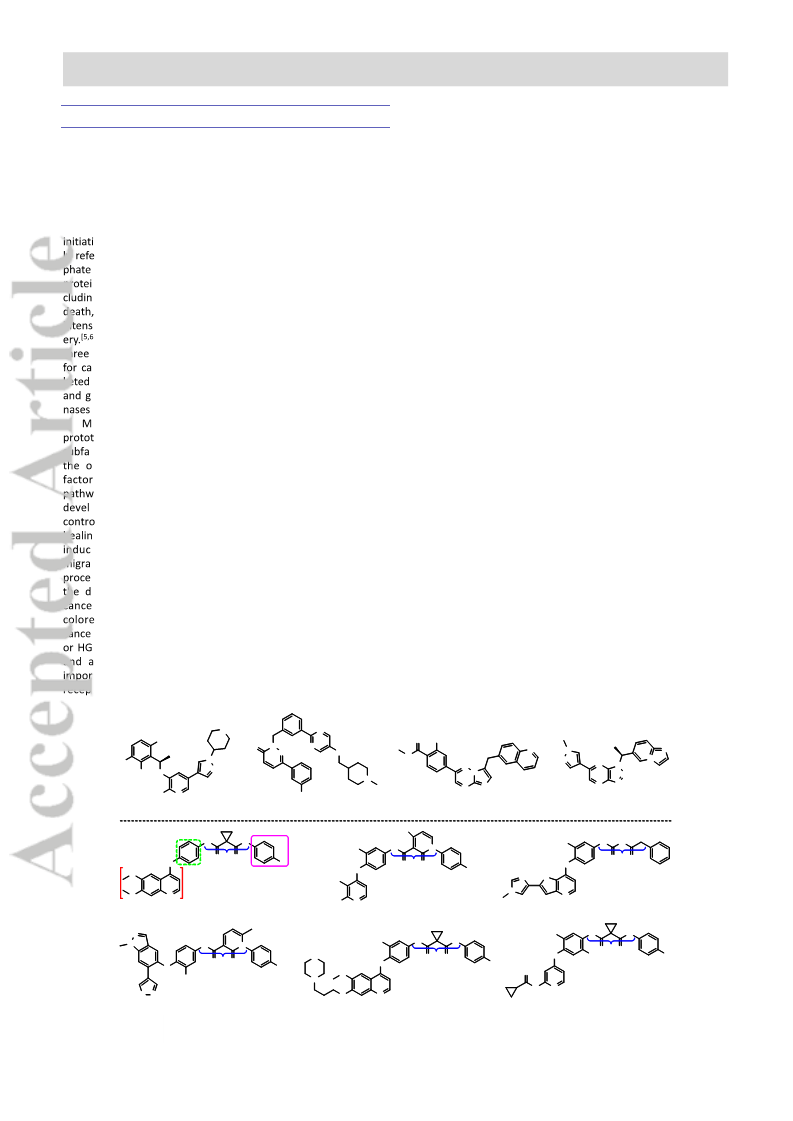
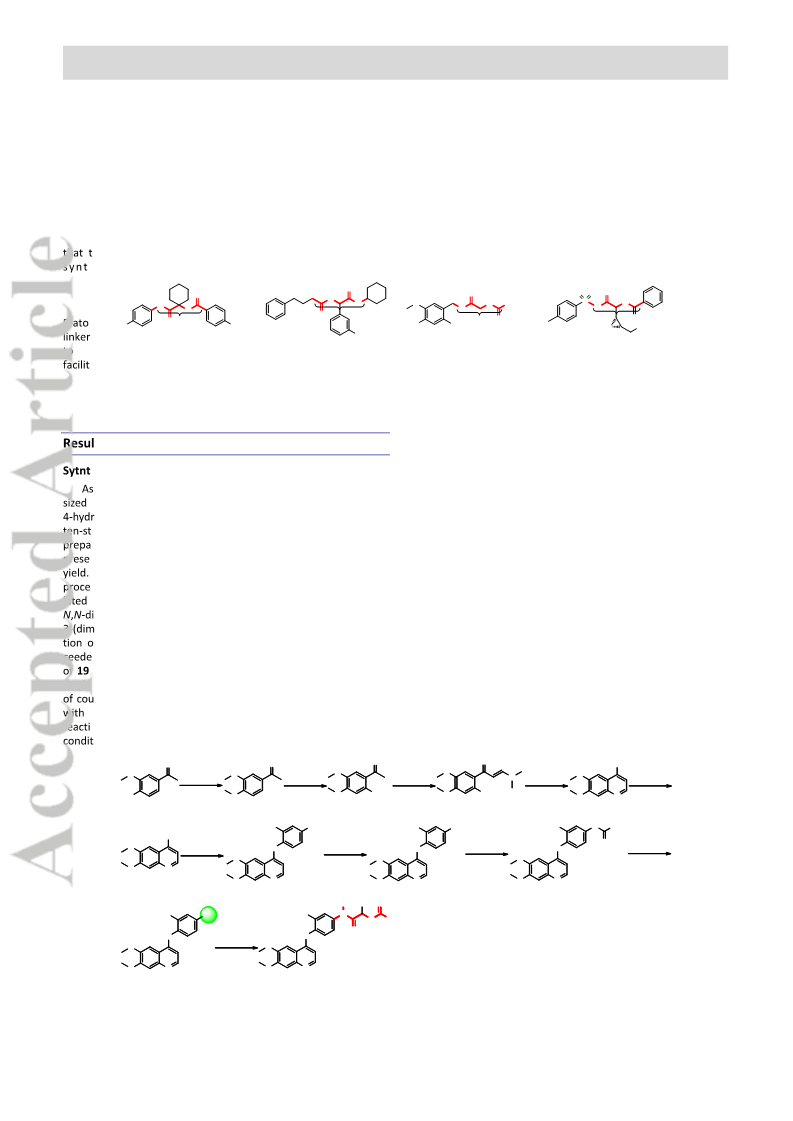
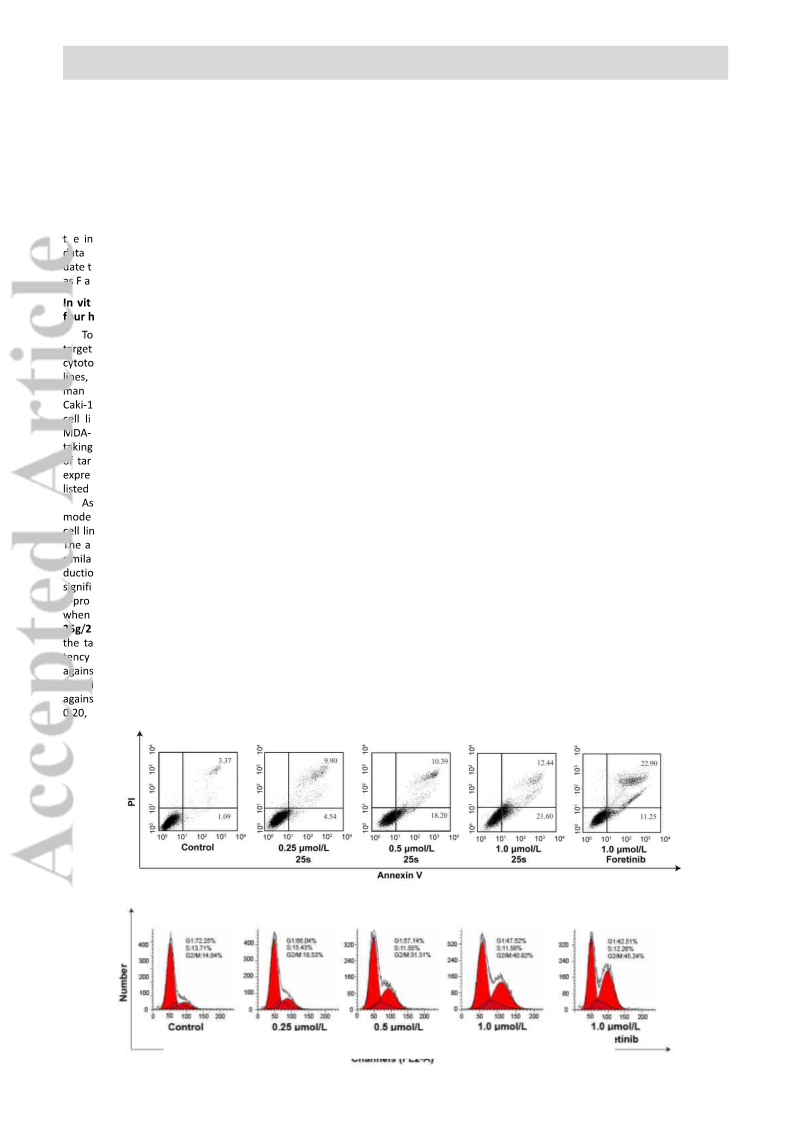
Quinoline derivatives bearing α-acyloxycarboxamide scaffold as potent c-Met kinase inhibitors
Chin. J. Chem.
[20] D’Angelo, N. D.; Bellon, S. F.; Booker, S. K.; Cheng, Y.; Coxon, A.;
Dominguez, C.; Fellows, I.; Hoffman, D.; Hungate, R.; Kaplan-Lefko, P.;
Lee, M. R.; Li, C.; Liu, L.; Rainbeau, E.; Reider, P. J.; Rex, K.; Siegmund,
A.; Sun, Y.; Tasker, A. S.; Xi, N.; Xu, S.; Yang, Y.; Zhang, Y.; Burgess, T.
L.; Dussault, I.; Kim, T. -S. Design, Synthesis, and Biological Evaluation
of Potent c-Met Inhibitors. J. Med. Chem. 2008, 51, 5766–5779.
[21] Wang, W.; Xu, S.; Du, Y.; Liu, X.; Li, X.; Wang, C.; Zhao, B.; Zheng, P.;
Zhu, W. Synthesis and bioevaluation and doking study of
1H-pyrrolo[2,3-b] pyridine derivatives bearing aromatic hydrazone
moiety as c-Met inhibitors. Eur. J. Med. Chem. 2018, 145, 315–327.
[22] Liu, J.; Gong, Y.; Shi, J.; Hao, X.; Wang, Y.; Zhou, Y.; Hou, Y.; Liu, Y.;
Ding, S.; Chen, Y. Design, synthesis and biological evaluation of novel
N-[4-(2-fluorophenoxy)pyridin-2-yl]cyclopropanecarboxamide
derivatives as potential c-Met kinase inhibitors. Eur. J. Med. Chem.
2020, 194, 112244.
[23] Parikh, P. K.; Ghate, M. D. Recent advances in the discovery of small
molecule c-Met Kinase inhibitors. Eur. J. Med. Chem. 2018, 143,
1103–1138.
[24] Yuan, H.; Liu, Q.; Zhang, L.; Hu, S.; Chen, T.; Li, H.; Chen, Y.; Xu, Y.; Lu,
T. Discovery, optimization and biological evaluation for novel c-Met
kinase inhibitors. Eur. J. Med. Chem. 2018, 143, 491–502.
[25] Nan, X.; Li, H. -J.; Fang, S. -B.; Li, Q. -Y.; Wu, Y. -C. Structure-based
discovery of novel 4-(2-fluorophenoxy)quinoline derivatives as c-Met
inhibitors using isocyanide-involved multicomponent reactions. Eur.
J. Med. Chem. 2020, 193, 112241.
[26] Zhuo, L. -S.; Xu, H. -C.; Wang, M. -S.; Zhao, X. -E.; Ming, Z. -H.; Zhu, X.
-L.; Huang, W.; Yang, G. -F. 2,7-Naphthyridinone-based MET kinase
Acknowledgement
This work was supported by the Fundamental Research Funds
for the Central Universities (31920170193, 31920200026).
References
[2] Torre, L. A.; Bray, F.; Siegel, R. L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal,
A. Global cancer statistics, CA Cancer J. Clin. 2015, 65, 87–108.
[3] Bray,F.; Jemal, A.; Grey, N.; Ferlay, L.; Forman, D. Global cancer
transitions according to the Human Development Index (2008–2030):
a population-based study. Lancet Oncol. 2012, 13, 790–801.
[4] Manning, G.; Whyte, D. B.; Martinez, R.; Hunter, T.; Sudarsanam, S.
The protein kinase complement of the human genome. Science 2002,
298, 1912–1934.
[5] Wang, Z.; Cole, P. A. Catalytic mechanisms and regulation of protein
kinases. Methods Enzymol. 2014, 548, 1–21.
[6] Lu, X; Smaill, J. B.; Ding, K. Medicinal chemistry strategies for the
development of kinase inhibitors targeting point mutations. J. Med.
Chem. 2020, 63, 10726–10741.
[7] Wu, P.; Nielsen, T. E.; Clausen, M. H. Small molecule kinase inhibitors:
an analysis of FDA-approved drugs. Drug Discovery Today 2016, 21,
5–10.
[8] Wu, P.; Nielsen, T. E.; Clausen, M. H. FDA-approved small molecule
kinase inhibitors. Trends Pharmacol. Sci. 2015, 36, 422–439.
[9] Cui, J. J. Targeting receptor tyrosine kinase MET in cancer: small
molecule inhibitors and clinical progress. J. Med. Chem. 2014, 57,
4427–4453.
[10] Dean, M.; Park, M.; Le Beau, M. M.; Robins, T. S.; Diaz, M. O.; Rowley,
J. D.; Blair, D. G.; Vande Woude, G. F. The human met oncogene is
related to the tyrosine kinase oncogenes. Nature 1985, 318,
385–388.
[11] Bottaro, D.P.; Rubin, J. S.; Faletto, D. L.; Chan, A. M. -L.; Kmiecik, T. E.;
Vande Woude, G. F.; Aaronson, S. A. Identification of the hepatocyte
growth factor receptor as the c-met proto-oncogene product. Science
1991, 251, 802–804.
[12] Robinson, D. R.; Wu, Y. -M.; Lin, S. -F. The protein tyrosine kinase
family of the human genome. Oncogene 2000, 19, 5548–5557.
[13] Trusolino, L.; Comoglio, P. M. Scatter-factor and semaphorin
receptors: cell signaling for invasive growth. Nat. Rev. Canc. 2002, 2,
289–300.
[14] Birchmeier, C.; Birchmeier, W.; Gherardi, E.; Vande Woude, G. F. Met,
metastasis, motility and more. Nat. Rev. Mol. Cell Biol. 2003, 4,
915–925.
[15] Gao, W.; Han, J. Overexpression of ING5 inhibits HGF-induced
proliferation, invasion and EMT in thyroid cancer cells via regulation
of the c-Met/PI3K/Akt signaling pathway. Biomed. Pharmacother.
2018, 98, 265–270.
[16] Takayama, H.; LaRochelle, W. J.; Sharp, R.; Otsuka, T.; Kriebel, P.;
Anver, M.; Aaronson, S. A.; Merlino, G. Diverse tumorigenesis
associated with aberrant development in mice overexpressing
hepatocyte growth factor/scatter factor. Proc. Natl. Acad. Sci. USA
1997, 94, 701–706.
[17] Baldacci, S.; Kherrouche, Z.; Cockenpot, V.; Stoven, L.; Copin, M. C.;
Werkmeister, E.; Marchand, N.; Kyheng, M.; Tulasne, D.; Cortot, A. B.
MET amplification increases the metastatic spread of EGFR-mutated
NSCLC. Lung Cancer 2018, 125, 57–67.
[18] Tsakonas, G.; Botling, J.; Micke, P.; Rivard, C.; LaFleur, L.; Mattsson, J.;
Boyle, T.; Hirsch, F. R.; Ekman, S. c-MET as a biomarker in patients
with surgically resected non-small cell lung cancer. Lung Cancer 2019,
133, 69–74.
[19] Tao, L.; Qiu, J.; Slavin, S.; Ou, Z.; Liu, Z.; Ge, J.; Zuo, L.; Guancial, E. A.;
Messing, E. M.; Chang, C.; Yeh, S. Recruited T cells promote the
bladder cancer metastasis via up-regulation of the estrogen receptor
β/IL-1/c-MET signals. Cancer Lett. 2018, 430, 215–223.
inhibitors:
a
promising novel scaffold for antitumor drug
development. Eur. J. Med. Chem. 2019, 178, 705–714.
[27] Liu, J.; Yang, D.; Yang, X.; Nie, M.; Wu, G.; Wang, Z.; Li, W.; Liu, Y.;
Gong, P. Design, synthesis and biological evaluation of novel
4-phenoxyquinoline
derivatives
containing
3-oxo-3,4-dihydroquinoxaline moiety as c-Met kinase inhibitors.
Bioorg. Med. Chem. 2017, 25, 4475–4486.
[28] Liao, W.; Xu, C.; Ji, X.; Hu, G.; Ren, L.; Liu, Y.; Li, R.; Gong, P.; Sun, T.
Design and optimization of novel 4-(2-fluorophenoxy)quinoline
derivatives bearing a hydrazone moiety as c-Met kinase inhibitors.
Eur. J. Med. Chem. 2014, 87, 508–518.
[29] Zhou, S.; Ren, J.; Liu, M.; Ren, L.; Liu, Y.; Gong, P. Design, synthesis
and
pharmacological
evaluation
of
6,7-disubstituted-4-phenoxyquinoline derivatives as potential
antitumor agents. Bioorg. Chem. 2014, 57, 30–42.
[30] Zhou, S.; Liao, H.; He, C.; Dou, Y.; Jiang, M.; Ren, L.; Zhao, Y.; Gong, P.
Design, synthesis and structure-activity relationships of novel
4-phenoxyquinoline derivatives containing pyridazinone moiety as
potential antitumor agents. Eur. J. Med. Chem. 2014, 83, 581–593.
[31] Dömling, A. Recent developments in isocyanide based
multicomponent reactions in applied chemistry. Chem. Rev. 2006,
106, 17–89.
[32] Dömling, A.; Wang, W.; Wang, K. Chemistry and biology of
multicomponent reactions. Chem. Rev. 2012, 112, 3083–3135.
[33] Kou, L.; Wang, M. -J.; Wang, L. -T.; Zhao, X. -B.; Nan, X.; Yang, L.; Liu, Y.
third-generation spin-labeled podophyllotoxin derivatives using
isocyanide multicomponent reactions. Eur. J. Med. Chem. 2014, 75,
282–288.
[34] Zhao, X. -B.; Wu, D.; Wang, M. -J.; Goto, M.; Morris-Natschke, S. L.;
Liu, Y. -Q.; Wu, X. -B.; Song, Z. -L.; Zhu, G. -X.; Lee, K. -H. Design and
synthesis of novel spin-labeled camptothecin derivatives as potent
cytotoxic agents. Bioorg. Med. Chem. 2014, 22, 6453–6458.
[35] Zhang, J.; Lin, S. -X.; Cheng, D. -J.; Liu, X. -Y.; Tan, B. Phosphoric
acid-catalyzed asymmetric classic passerini reaction. J. Am. Chem.
Soc. 2015, 137, 14039–14042.
[36] Serafini, M.; Griglio, A.; Aprile, S.; Seiti, F.; Travelli, C.; Pattarino, F.;
Grosa, G.; Sorba, G.; Genazzani, A. A.; Gonzalez-Rodriguez, S.; Butron,
L.; Devesa, I.; Fernandez-Carvajal, A.; Pirali, T.; Ferrer-Montiel, A.
Targeting transient receptor potential vanilloid 1 (TRPV1) channel
Chin. J. Chem. 2021,39, XXX-XXX
© 2021 SIOC, CAS, Shanghai, & WILEY-VCH GmbH

 Feng, Yu-juan
Feng, Yu-juan
 Ren, Yu-Lin
Ren, Yu-Lin
 Zhao, Li-Ming
Zhao, Li-Ming
 Xue, Guo-Qiang
Xue, Guo-Qiang
 Yu, Wen-Hao
Yu, Wen-Hao
 Yang, Jia-Qi
Yang, Jia-Qi
 Liu, Jun-Wei
Liu, Jun-Wei