Russian Journal of Organic Chemistry, Vol. 38, No. 9, 2002, pp. 1384 1385. From Zhurnal Organicheskoi Khimii, Vol. 38, No. 9, 2002, pp. 1435 1436.
Original English Text Copyright
2002 by Tajik, Shirini, Farahkah, Lakouraj, Baltork.
SHORT
COMMUNICATIONS
Coupling of Thiols Catalyzed by Trinitratocerium(IV) Bromate*
1
1
1
2
3
H. Tajik , F. Shirini , O. R. Farahkah , M. M. Lakouraj , and I. M. Baltork
1
Department of Chemistry, College of Science, Guilan University, Rasht, Iran
2
Department of Chemistry, College of Science, Mazandaran University, Babolsar, Iran
3
Department of Chemistry, College of Science, Isfahan University, Iran
Received October 25, 2001
Oxidative coupling of thiols to disulfides under
neutral and mild conditions is important from both
biological and practical viewpoints [1]. Thiols are
compounds which can be overoxidized; therefore,
extensive studies have been carried out on their con-
trolled oxidation [2 16]. All reagents presently used
for this purpose are characterized by one or more of
the following disadvantages: relatively long reaction
time [8, 16], low accessibility [15], difficult prepara-
tion [14], laborious isolation procedure [14]. We
have already described the use of trinitratocerium(IV)
bromate (NO ) CeBrO (TNCB) as a safe, easily
obtainable, and stable oxidant with respect to benzyl
alcohols and acyloins in boiling acetonitrile [17].
We now report that TNCB can act as a very efficient
reagent for oxidation of thiols to disulfides in boiling
acetonitrile, which ensures high yields of the products
The authors are thankful to the Research Council
(Guilan University) for partial support of this work.
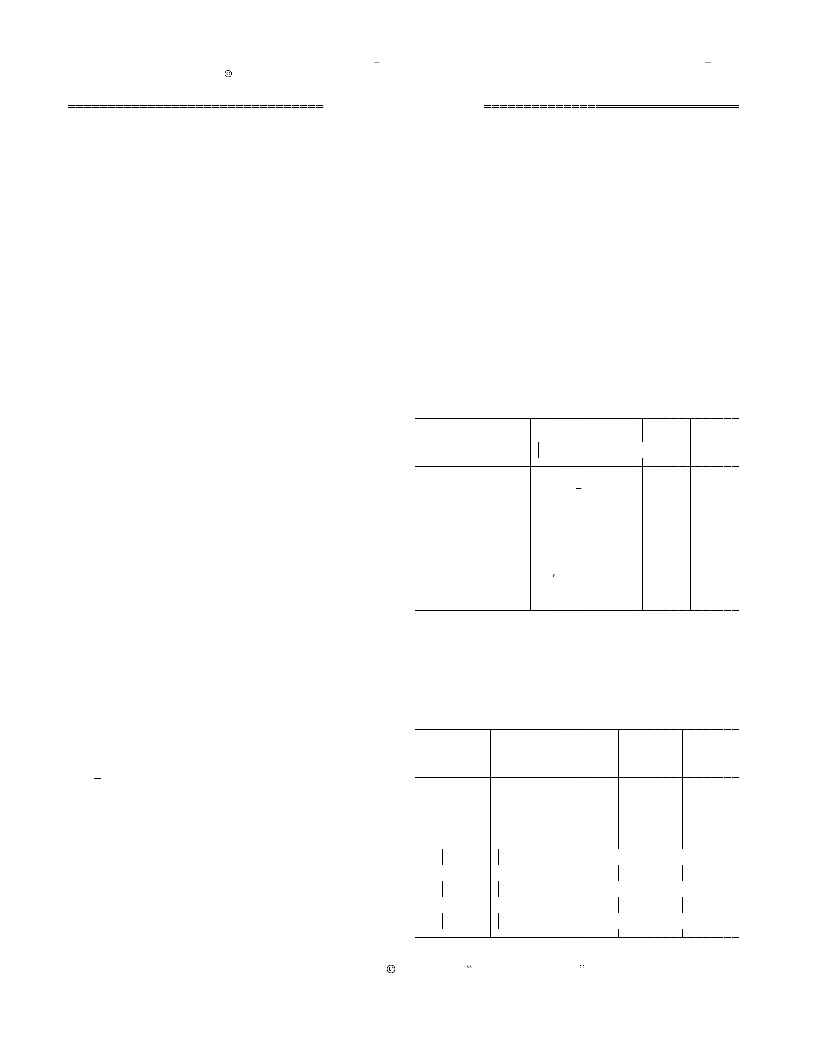
Table 1. Oxidation of thiols with trinitratocerium(IV)
bromate in boiling acetonitrile
Time, Yield,a
Substrate
C H SH
Product
(C H S )
h
%
0.25
0.3
0.16
4.5
92
95
90
0
6
4
6
5
2
p-ClC H SH
(p-ClC H S)
6
4
6 4 2
3
3
3
p-MeC H SH
(p-MeC H S)
2
6
4
6
4
o-HSC H COOH
(o-HOCOC H S)
6
4
6
4
2
C H CH SH
(C H CH S)
0.25
0.42
90
95
89
6
5
2
6
5
2
2
cyclo-C H SH
(C H S)
6
11
6 11
2
2
-Mercaptopyridine 2,2 -Dithiopyridine 0.75
b
Ethane-1,2-dithiol
0.42
(Table 1).
To illustrate the efficiency of the proposed proce-
a
Yield of the isolated product.
Polymerization occurred.
dure, Table 2 compares some our results with pub-
lished data [8]. The easy experimental and isolation
procedure, short reaction time, and excellent yields
of the products make the proposed procedure a useful
supplement to the existing methods.
Typical procedure for oxidation of thiols with
trinitratocerium(IV) bromate. A solution of 1 mmol
of thiol in 3 ml of acetonitrile was treated with
b
Table 2. Oxidation of thiols with trinitratocerium(IV)
bromate (A), bis[trinitratocerium(IV) bromate] (B) [8], and
pyridinium chlorochromate (C) [8]
Oxidant (oxidant-to- Time,
Yield,
%
Substrate
C H SH
0
0
.2 equiv of TNCB, and the mixture was heated for
.16 4.5 h under reflux. The progress of the reaction
substrate ratio)
h
was monitored by TLC and GLC. When the reaction
was complete, the mixture was filtered, and the solid
precipitate was filtered off and washed with 10 ml
of acetonitrile. The filtrate was evaporated, and the
residue was subjected to column chromatography on
silica gel. The products were identified by the melting
points and IR and NMR spectra, which were com-
pared with published data for authentic samples.
A (0.2)
B (2)
0.25
4
0.03
0.42
92
90
85
95
6
5
C H SH
6
5
C H SH
C (1)
6
5
cyclo-
A (0.2)
C H SH
6
11
cyclo-
B (2)
C (1)
6
2
80
0
C H SH
6
11
cyclo-
_
*
___________
C H SH
6
11
The original article was submitted in English.
1
070-4280/02/3809-1384$27.00 2002 MAIK Nauka/Interperiodica

 Tajik
Tajik
 Shirini
Shirini
 Farahkah
Farahkah
 Lakouraj
Lakouraj
 Baltork
Baltork