1
236
M. A. REZVANI ET AL.
11. Chung, K.Y.; Lee, S.J.; Chung, S.M.; Lee, M.Y.; Bae, O.N.; Chung, J.H.
Effect of the Solvent
Thromb. Res. 2005, 116, 157.
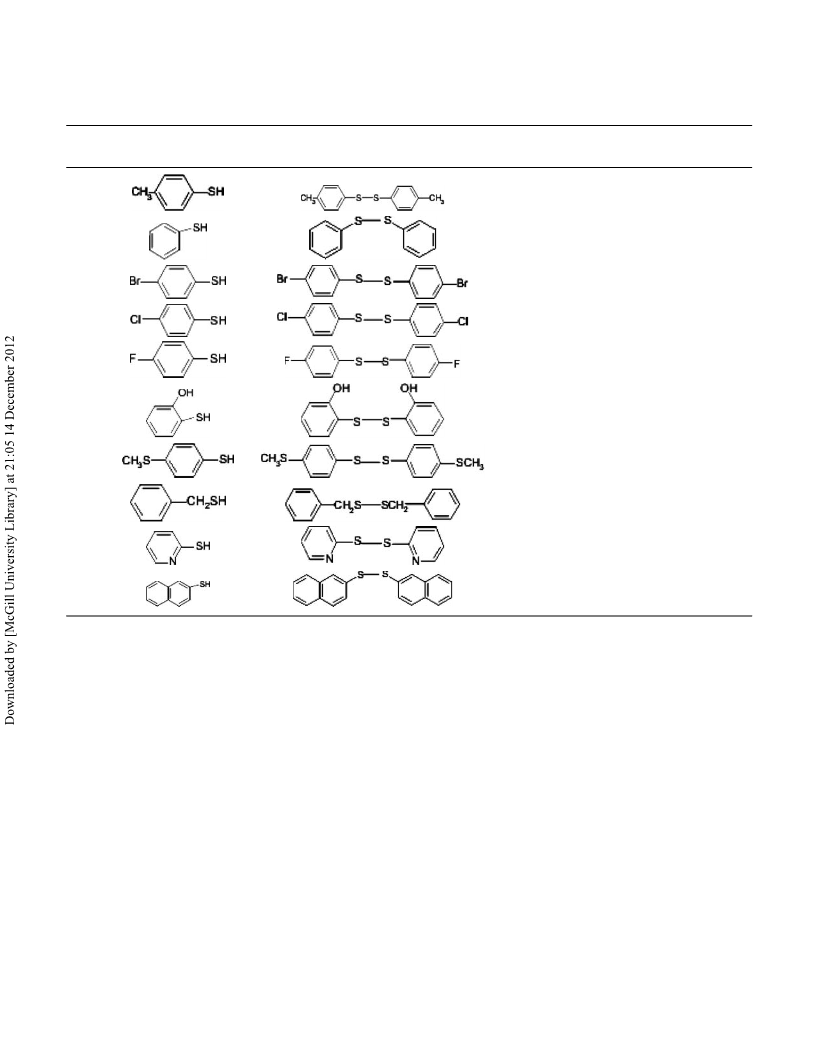
Table 3 shows solvent effect on oxidation of solid thiols.
12. Bagiyan, G.A.; Koroleva, I.K.; Soroka, N.V.; Umtsev, A.V. Russian Chem.
4-Chlorothiophenol was taken as a model compound and the
Bull. 2003, 52, 1135.
reaction conditions were optimized by varying the solvent. We
examined various solvents to facilitate this transformation and
13. Kumar, B.; Parmar, A.; Rajpal, A.; Kumar, H. Ind. J. Chem. Sect. B 1998,
37B, 593.
determined that (EtOH + H2O) was the most effective (Table 3 14. Oae, S. (Ed.): Organic Sulfur Chemistry: Structure and Mechanism; Boca
Raton, FL: CRC Press, 1991.
5. Cremlyn, R.J. An Introduction to Organosulfur Chemistry; New York: Wi-
ley, 1996.
entries 1–13).
1
Effect of the Substituent
16. Dhar, D.N.; Bag, A.K. Ind. J. Chem. 1984, 23B, 974.
1
1
7. Firouzbadi, H.; Iranpoor, N.; Parham, H.A. Synth. Commun. 1984, 14, 717.
8. Noureldin, N.A.; Caldwell, M.; Hendry, J.; Lee, D.G. Synthesis 1998,
The effects of various substituents on the yields of pro-
duced disulfides have been examined in the presence of
H5[PMo10V2O40] catalyst. As shown in Table 4, not only the
nature of the substituent is important, but so is its position.
1
587.
9. Firouzabadi, H.; Naderi, M.; Sardarian, A.; Vessal, M. Synth. Commun.
983, 13, 611.
1
1
As examples of electron-withdrawing groups, bromo-, chloro-, 20. Wallace, T.J. J. Org. Chem. 1966, 31, 1217.
2
2
2
2
2
1. Liu, K.T.; Tong, Y.C. Synthesis 1978, 669.
and nitro-substituted thiophen were chosen and were converted
to their corresponding disulfides. Methyl and hydroxy groups
were chosen as electron-donating groups. Thiols with electron-
donation substituents were oxidized easily respect to thiols with
electron-drawing substituents.
2. McKillop, A.; D. Koyuncu, Tetrahedron Lett. 1990, 31, 5007.
3. Ramesha, A.R.; Chandrasekaran, S. J. Org. Chem. 1994, 59, 1354.
4. Ramadas, K.; Srinivasan, N. Synth. Commun. 1995, 25, 227.
5. Pryor, W.A.; Church, D.F.; Govindan, C.K.; Crank, G. J. Org. Chem. 1982,
47, 156.
26. Heravi, M.M.; Kh. Bakhtiari; Bamoharram, F.F. Catal. Commun. 2006, 7,
3
73.
CONCLUSION
27. Bamoharram, F.F.; Heravi, M.M.; Roshani, M.; Gharib, A.; Jahangir, M. J.
Mol. Catal. A: Chem. 2006, 252, 90.
The H5[PMo10V2O40] HPA was the most successful HPA
in this reaction. Although it is difficult to offer an explanation
for the different activity between these HPAs, certainly there
is a complex relationship between the activity and structure of
2
8. Heravi, M.M.; Bakhtiari, Kh.; Bamoharram, F.F. Catal. Commun. 2006, 7,
99.
4
29. Heravi, M.M.; Motamedi, R.; Seifi, N.; Bamoharram, F.F. J. Mol. Catal. A:
Chem. 2006, 249, 1.
polyanion. By changing the constituent elements of polyanion 30. Bamoharram, F.F.; Heravi, M.M.; Roshani, M.; Jahangir, M.; Gharib, A. J.
Appl. Catal. A: Gen. 2006, 302, 42.
31. Heravi, M.M.; Ranjbar, L.; Derikvand, F.; Bamoharram, F.F. Catal. Com-
mun. 2007, 8, 289.
2. Heravi, M.M.; Khorasani, M.; Derikvand, F.; Oskooie, H.A.; Bamoharram,
F.F. Catal. Commun. 2007, 8, 1886.
(
both hetero and addenda atoms), the acid strength of HPAs as
[8]
well as its catalytic activity is able to vary in a wide range. For
the first time, by an inexpensive solid acid catalyst and an easily
prepared H5[PMo10V2O40] oxidation of thiols with electron-
3
withdrawing and electron-donating groups to the corresponding 33. Heravi, M.M.; Behbahani, F.K.; Bamoharram, F.F. J. Mol. Catal. A: Chem.
2
006, 253, 16.
disulfides have been studied. The results show that the catalyst
type, the nature and positions of substituents, and temperature
are important factors. The Keggin-type polyoxometalates lead to
effective reaction in comparison with Well-Dawson type poly-
oxometalates.
3
4. Heravi, M.M.; Behbahani, F.K.; Hekmatshoar, R.; Oskooie, H.A. Catal.
Commun. 2006, 7, 136.
5. Heravi, M.M.; Derikvand, F.; Ranjbar, L.; Bamoharram, F.F. J. Mol. Catal.
A: Chem. 2007, 261, 156–159.
36. Alizadeh, M.H.; Razavi, H.; Bamoharram, F.F.; Daneshvar, K. J. Mol. Catal.
A: Chem. 2003, 206, 89–93.
3
37. Heravi, M.M.; Derikvand, F.; Bamoharram, F.F. J. Mol. Catal. A: Chem.
REFERENCES
2007, 263, 112–114.
1
. Izumi, Y.; Urabe, K.; Onaka, M. Zeolites Clay and Heteropolyacid in Or-
38. Heravi, M.M.; Bakhtiari, Kh.; Javadi, N.M.; Bamoharram, F.F.; Saeedi, M.;
Oskooie, H.A. J. Mol. Catal. A: Chem. 2007, 264, 50–52.
39. Heravi, M.M.; Benmord, T.; Bakhtiari, K.; Bamoharram, F.F.; Oskooie,
H.A. J. Mol. Catal. A: Chem. 2006, 264, 318–321.
40. Heravi, M.M.; Derikvand, F.; Ranjbar, L.; Oskooie, H.A.; Bamoharram,
F.F. J. Mol. Catal. A: Chem. 2007, 261, 156.
ganic Reactions; Tokyo: Kodansha, 1992; Vol. 99.
2
3
4
. Kesavan, V.; Bonnet-Delpon, D.; Begue, J.P. Synthesis 2000, 223.
. Kozhevnikov, I.V. Chem. Rev. 1998, 98, 171.
. Kozhevnikov, I.V. Catalysis for Fine Chemical Synthesis, Catalysis by Poly-
oxometalates; New York: Wiley, 2002; Vol. 2.
5
. L o´ pez, X. Theoretical Study of the Basicity and the Redox Properties of
Heteropolyanions; Ph.D. Dissertation: Universitat Rovira i Virgili, 2003.
. Langpape, M.; Millet, J.C.M. Appl. Catal. A: Gen. 2000, 89, 200.
. Rong, C.; Anson, F.C. Inorg. Chem. 1994, 33, 1064.
. Cavani, F.; Comuzzi, C. J. Catal. 1996, 160, 317.
. Pope, M.T.; Muller, A. Polyoxometalates: From Platonic Solid to Anti-
41. Heravi, M.M.; Derikvand, F.; Bamoharram, F.F. J. Mol. Catal. A: Chem.
2007, 263, 112.
42. Shikata, S.; Nakata, S.; Okuhara, T.; Misono, M. J. Catal. 1997, 166, 263.
43. Keggin, J.F. Proc. R. Soc. London, Ser. A 1934, 144, 75.
44. Khenkin, A.M.; Weiner, L.; Wang, Y.; Neumann, R. J. Am. Chem. Soc.
2001, 123, 8531.
6
7
8
9
Retroviral Activity; Dorderecht, the Netherlands: Kluwer Academic, 1994. 45. Perrin, D.D.; Armarego, W.L.F. Purification of Laboratory Chemicals, 3rd
10. Rao, T.V.; Sain, B.; Murthy, P.S.; Prasada Rao, T.S. R.; Jain, A.K.; Joshi,
Edn.; New York: Pergamon Press, 1988.
G.C. J. Chem. Res. Synop. 1997, 8, 300.
46. Tsigdinos, G.A.; Hallada, C. J. Inorg. Chem. 1968, 7, 437.

 Rezvani, M. A.
Rezvani, M. A.
 Harutyunyan, R.
Harutyunyan, R.
 Heravi, Majid M.
Heravi, Majid M.