PleaseC dh oe mn oi ct a al dS cj ui es nt cme argins
Page 8 of 9
ARTICLE
Journal Name
K. Murakami, K. Itami, Science 2018, 359, 435.
T. Friščić, D. G. Reid, I. Halasz, R. S. Stein, R. E. Dinnebier, M. J.
DOI: 10.1039/C9SC02185J
Duer, Angew. Chem. Int. Ed. 2010, 49, 712.
5
For examples of solvent-free solid-state Suzuki–Miyaura cross-
coupling reactions using mechanochemistry, see: (a) G. 15 The selected example of vapour-assisted-grinding, see: C. Jia, J.
Cravotto, D. Garella, S. Tagliapietra, A. Stolle, S. Schüßler, S. E.
Wang, X. Feng, Q. Lin, W. Tuan, CrystEngComm. 2014, 16, 6552.
S. Leonhardt, B. Ondruschka, New J. Chem. 2012, 36, 1304. (b) 16 The selected examples of polymer-assisted-grinding, see: (a) D.
F. Schneider, B. Ondruschka, ChemSusChem, 2008, 1, 622. (c) S.
F. Nielsen, D. Peters, O. Axelsson, Synth. Commun. 2000, 30,
Hasa, G. S. Rauber, D. Voinovich, W. Jones, Angew. Chem., Int.
Ed. 2015, 54, 7371. (b) D. Hasa, E. Carlino, W. Jones, Cryst.
Growth Des. 2016, 16, 1772.
3
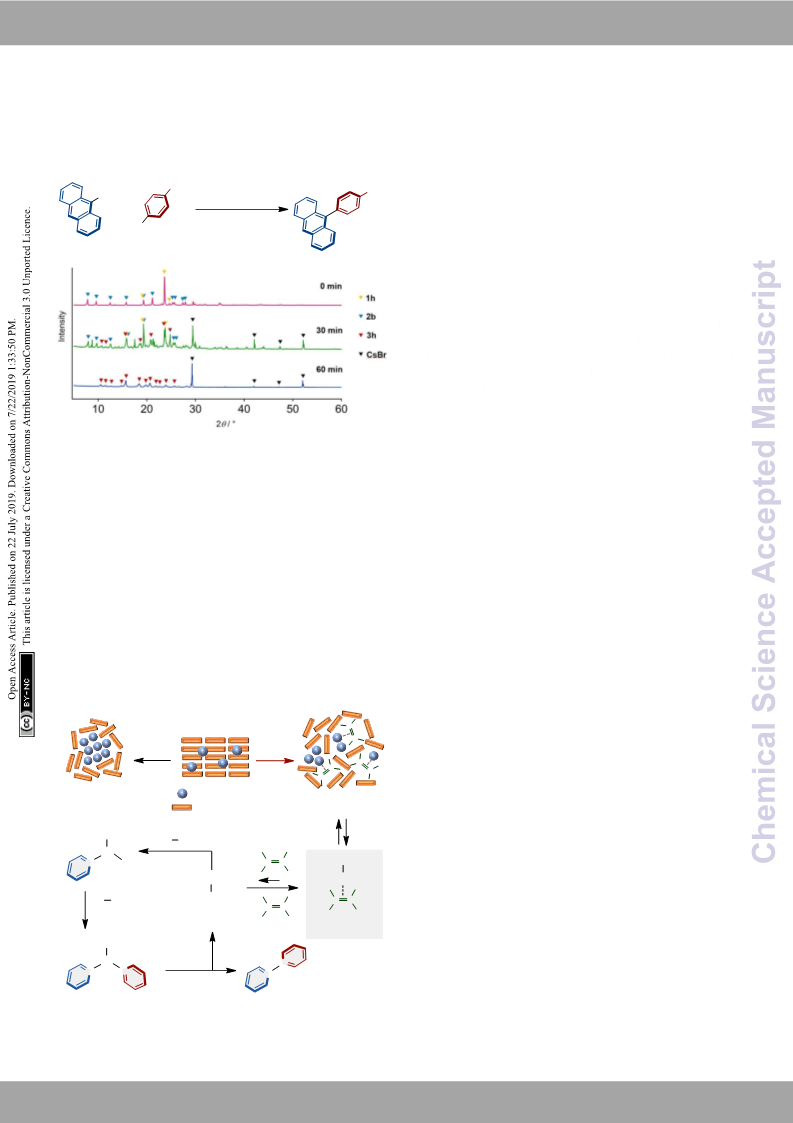
2
501. (d) E. N. Leadbeater, M. L. Klingensmith, Tetrahedron Lett.
003, 44, 765.
17 (a) J. Hou, H.–Y. Chen, S. Zhang, G. Li, Y. Yang, J. Am. Chem. Soc.
2008, 130, 16144. (b) Z. Yao, M. Zhang, H. Wu, L. Yang, R. Ki, P.
Wang, J. Am. Chem. Soc. 2015, 130, 16144. (c) J. Zhang, W.
Chen, A. J. Rojas, E. V. Jucov, T. V. Timofeeva, T. C. Parker, S.
Barlow, S. R. Marder, J. Am. Chem. Soc. 2013, 135, 16376.
18 When the solution-based reaction was carried out at room
temperature, the product yield was significantly lower (33%
NMR yield).
6
7
For examples of solvent-free Suzuki–Miyaura cross-coupling
reactions of liquid substrates using mechanochemistry, see: (a)
F. Schneider, A. Stolle, B. Ondruschka, H. Hopf, Org. Process Res.
Dev. 2009, 13, 44. (b) Z.–J. Jiang, Z.–H. Li, J.–B. Yu, W.–K. Su. J.
Org. Chem. 2016, 81, 10049.
For recent reviews on organic syntheses using
mechanochemistry, see: (a) S. L. James, C. J. Adams, C. Bolm, D.
Braga, P. Collier, T. Friščić, F. Grepioni, K. D. M. Harris, G. Hyett, 19 The reaction under an atmosphere of Ar provided a similar
W. Jones, A. Krebs, J. Mack, L. Maini, A. G. Orpen, I. P. Parkin,
W. C. Shearouse, J. W. Steed, D. C. Waddell, Chem. Soc. Rev.
result as the reaction conducted in air, suggesting that the
presence/absence of oxygen is not crucial for the reaction,
which is another advantage of this method.
2
012, 41, 413. (b) G.-W. Wang, Chem. Soc. Rev. 2013, 42, 7668.
(
c) T. Friščić, I. Halasz, V. Štrukil, M. Eckert-Maksić, R. E. 20 For structure and reactivity of [(LPd)n(1,5-cod)] complexes, see:
Dinnebier, ACS Cent. Sci. 2017, 3, 13. (d) J. G. Hernández, C.
Bolm, J. Org. Chem. 2017, 82, 4007. (e) J. G. Hernández, Chem.-
Eur. J. 2017, 23, 17157. (f) T.-X. Métro, J. Martinez, F. Lamaty, 21 We have also attempted to prepare the DavePhos-Pd(0)-(1,5-
H. G. Lee, P. J. Milner, M. T. Colvin, L. Andreas, S. L. Buchwald,
Inorganic Chimica Acta 2014, 422, 188.
ACS Sustainable Chem. Eng. 2017, 5, 9599. (g) T. K. Achar, A.
Bose, P. Mal, Beilstein J. Org. Chem. 2017, 13, 1907. (h) J.-L. Do,
T. Friščić, Synlett 2017, 28, 2066. (i) D. Tan, T. Friščić, Eur. J. Org.
Chem. 2018, 18. (j) O. Eguaogie, J. S. Vyle, P. F. Conlon, M. A.
Gîlea, Y. Liang, Beilstein J. Org. Chem. 2018, 14, 955. (k) J. L.
Howard, Q. Cao, D. L. Browne, Chem. Sci. 2018, 9, 3080. (l) J.
Andersen, J. Mack, Green Chem. 2018, 20, 1435. (m) N. R.
Rightmire, T. P. Hanusa, Dalton Trans. 2016, 45, 2352. (n) M.
Leonardi, M. Villacampa, J. C. Menéndez, Chem. Sci. 2018, 9,
2 2
cod) complex via the reaction of (1,5-cod)Pd(CH TMS) and
DavePhos in pentane, but the complex was probably unstable
in solution and decomposed during the reaction. However, the
3
1
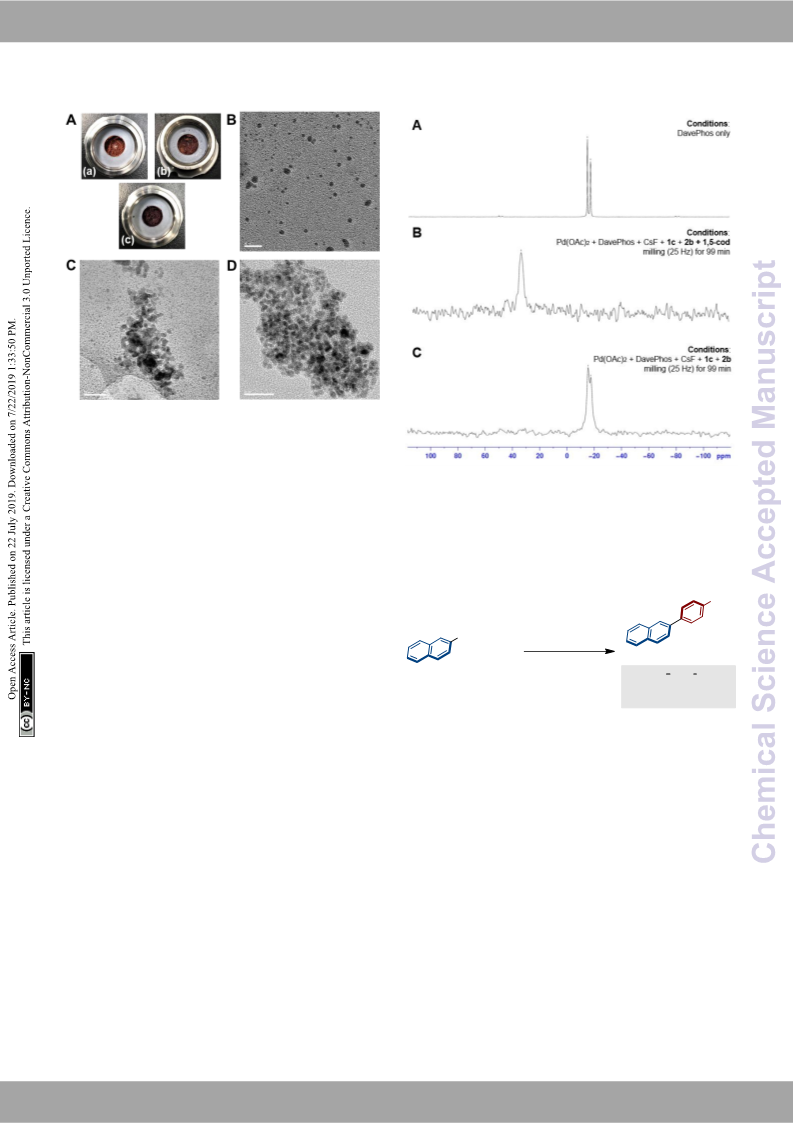
8
in situ P NMR analysis of the reaction in d -toluene showed a
signal around 35 ppm, which suggests the formation of the
DavePhos-Pd(0)-(1,5-cod) complex. The chemical shift is
3
1
consistent with the SS P NMR shown in Figure 3B, suggesting
3
1
that the observed signal (37.6 ppm) in the SS P NMR would
be derived from the DavePhos-Pd(0)-(1,5-cod) complex.
2
042. (o) C. Xu, S. De, A. M. Balu, M. Ojeda, R. Luque, Chem. 22 (a) J. Hu, Y. Liu, Langmuir 2005, 21, 2121. (b) T. Yurino, Y. Ueda,
Commun. 2015, 51, 6698. (p) D. Tan, T. Friščić, Chem. Commun.
Y. Shimizu, S. Tanaka, H. Nishiyama, H. Tsurugi, K. Sato, K.
Mashima, Angew. Chem., Int. Ed. 2015, 54, 14437.
2
016, 52, 7760. (q) A. A. Gečiauskaité, F. García, Beilstein J. Org.
Chem. 2017, 13, 2068. (r) M. J. Muñoz-Bastista, D. Rodriguez-
Padron, A. R. Puente-Santiago, R. Luque, ACS Sustainable Chem.
Eng. 2018, 9, 9530. (s) J. Andersen, J. Mack, Green Chem. 2018,
2
0, 1435. (t) E. Colacino, M. Carta, G. Pia, A. Porcheddu, P. C.
Ricci, F. Delogu, ACS Omega, 2018, 3, 9196. (u) D. Tan, T. Friščić,
Eur. J. Org. Chem. 2018, 1, 18. (v) D. Tan, F. García, 2019, DOI:
1
0.1039/C7CS00813A. (w) C. Bolm, J. D. Hernández, Angew.
Chem., Int Ed. 2019, 58, 3285.
8
K. Kubota, T. Seo, K. Koide, Y. Hasegawa, H. Ito, Nat. Commun.
2
019, 10, 111.
9
1
I. J. S. Fairlamb, Org. Biomol. Chem. 2008, 6, 3645.
0
For selected examples of Suzuki–Miyaura cross-coupling
reactions for the synthesis of polyaromatic hydrocarbons
under solvent-based conditions, see: (a) J. Huang, B. Xu, J.–H.
Su, H. C. Chen, H. Tian, Tetrahedron 2010, 66, 7577. (b) B. Kobin,
S. Behren, B. Braun–Cula, S. Hecht, J. Phys. Chem. A. 2016, 120,
5
474. (c) S.–K. Kim, B. Yang, Y. Ma, J.–H. Lee, J.–W. Park, J.
Mater. Chem. 2008, 18, 3376. (d) S. Jeelani Basha, R.
Venkatachalam, V. Babu, S. Sethuraman, Beilstein J. Org. Chem.
2
013, 9, 698. (e) H. Lee, B. Kim, S. Kim, J. Kim, J. Lee, H. Shin, J.–
H. Lee, J. Park, J. Mater. Chem. C. 2014, 2, 4737.
1
1
1
2
J.–P. Corbet, G. Mignani, Chem. Rev. 2006, 106, 2651.
(a) D. W. Old, J. P. Wolfe, S. L. Buchwald, J. Am. Chem. Soc. 1998,
1
4
4
20, 9722. (b) R. Martin, S. L. Buchwald, Acc. Chem. Res. 2008,
1, 1461. (c) A. C. Sather, S. L. Buchwald, Acc. Chem. Res. 2016,
9, 2146.
1
3
The selected example of liquid-assisted grinding, see: T. Friščić,
S. L. Childs, S. A. A. Rizvi, W. Jones, CrystEngComm. 2009, 11,
4
18.
8
| J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Seo, Tamae
Seo, Tamae
 Ishiyama, Tatsuo
Ishiyama, Tatsuo
 Kubota, Koji
Kubota, Koji
 Ito, Hajime
Ito, Hajime