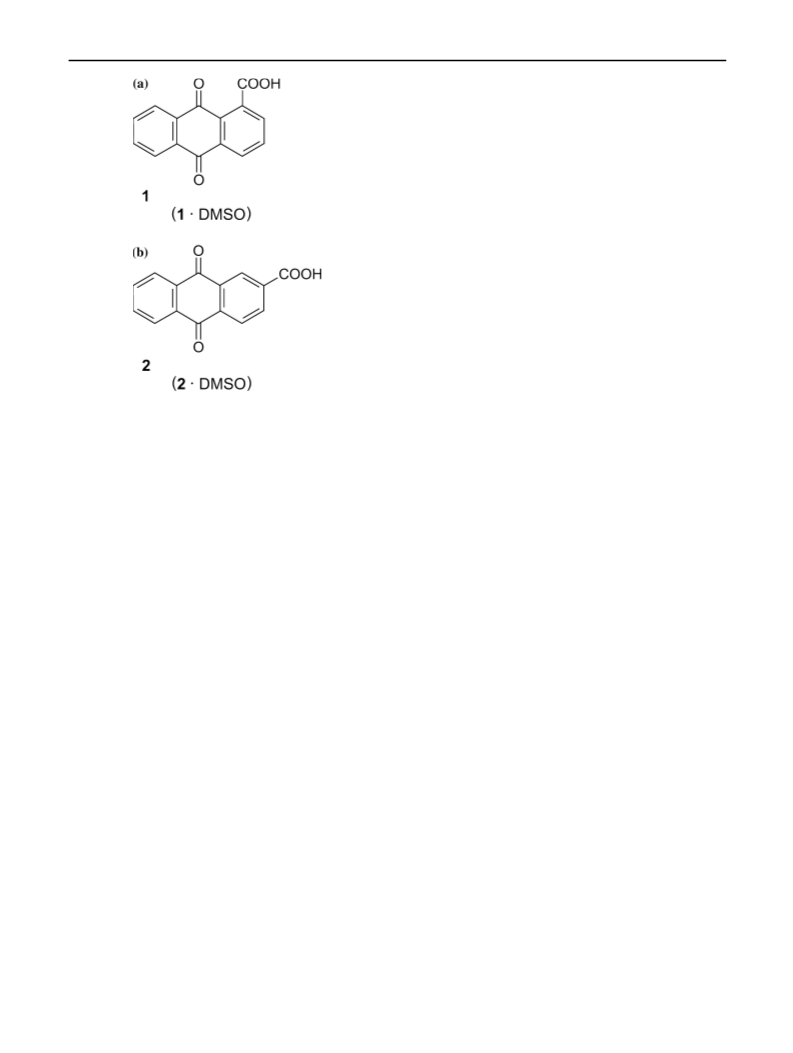
Struct Chem (2010) 21:1079–1083
1083
which are associated amongst one another using weak
˚
Supplementary data
hydrogen bonding [C(3)–H(3A)ÁÁÁO(3) 2.50 A, 138.7°].
CCDC 777783 (1a) and 777784 (2a) contain the supple-
mentary crystallographic data for this article. These data
Data Centre (CCDC), 12 Union Road, Cambridge CB2
1EZ, UK; fax: ?44(0)1223-336033); e-mail: deposit@ccdc.
cam.ac.uk].
Conclusion
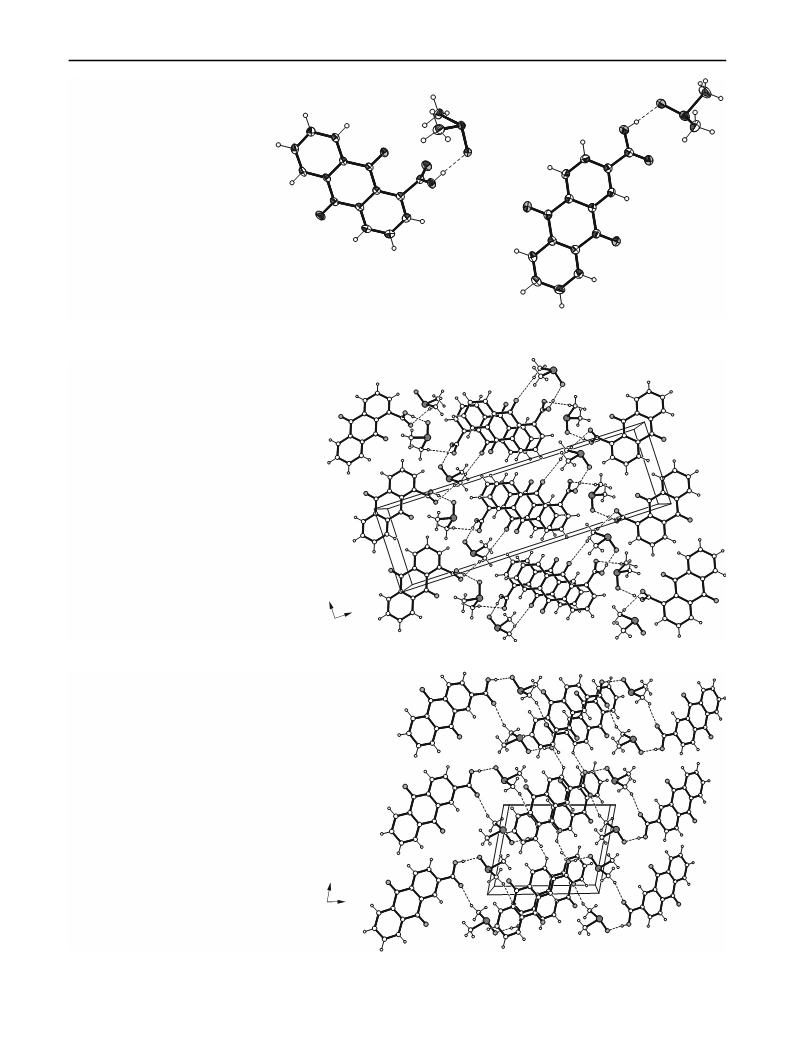
Irrespective of the constitutional isomerism of 1 and 2,
both the compounds form crystalline complexes (1a and
2a) with DMSO of 1:1 stoichiometry that show in some
respect structural correspondence but also distinct differ-
ences in their structures. Owing to the strong acceptor
character of the solvent component, O–HÁÁÁO-bonded 1:1
carboxylic acid–DMSO aggregates represent the basic
supramolecular entities in 1a and 2a which, however, are
interconnected in different ways. The position and orien-
tation of the carboxy substituent in 1a obviously favours
extensive carboxylic acid–DMSO and DMSO–DMSO
association comprising O–HÁÁÁO and C–HÁÁÁO hydrogen
bonding. Another important parameter stabilizing the lat-
tice structure of 1a is certainly p-stacking interaction. The
changed position of the carboxyl group of 2 markedly
diminishes the extent of molecular cross-linking in the
complex 2a, the crystal structure of which is characteristic
of cyclic supramolecular aggregates each containing two
carboxylic acid and two DMSO molecules. Interaction
between these aggregates is confined to fewer contacts.
This is in particular visible from the behaviour of the
anthraquinone oxygens, being involved in intermolecular
interactions with DMSO in 1a but totally excluded from
interaction in 2a, though sterically less obstructed by the
neighbouring carboxyl group compared with 1a.
References
1. Weber E (1989) J Mol Graphics 7:12
2. Desiraju GR (1996) In: MacNicol DD, Toda F, Bishop R (eds)
Comprehensive supramolecular chemistry, vol 6. Elsevier,
Oxford, pp 1–22
3. Weber E (1996) In: MacNicol DD, Toda F, Bishop R (eds) Com-
prehensive supramolecular chemistry, vol 6. Elsevier, Oxford,
pp 535–592
4. Weber E, Czugler M (1988) In: Weber E (ed) Molecular inclu-
sion and molecular recognition–clathrates II. Topics in current
chemisty, vol 149. Springer, Berlin-Heidelberg, pp 45–135
5. Weber E (2004) In: Atwood JL, Steed JW (eds) Encyclopedia of
supramolecular chemistry. Marcel Dekker, New York, pp 261–273
6. Weber E (1991) In: Atwood JL, Davies JED, MacNicol DD (eds)
Inclusion compounds, vol 4. Oxford University Press, Oxford,
pp 188–262
¨
7. Csoregh I, Czugler M, Ertan A, Weber E, Ahrendt J (1990) J Incl
Phenom 8:275
8. Makhkamov KK, Ibragimov BT, Weber E, Beketov KM (1999)
J Phys Org Chem 12:157
9. Kobayashi Y, Kodama SK, Saigo K (2008) Tetrahedron Asym-
metr 19:295
10. Golden R, Stock LM (1972) J Am Chem Soc 94:3080
11. Arjunan P, Berlin KD (1981) Org Prep Proced Int 13:368
12. Sheldrick GM (2008) Acta Crytsallogr A64:112
13. Desiraju GR, Steiner T (1999) The weak hydrogen bond in struc-
tural chemistry and biology. Oxford University Press, Oxford,
pp 29–121
14. Dance I (2004) In: Atwood JL, Steed JW (eds) Encyclope-
dia of supramolecular chemistry. Marcel Dekker, New York,
pp 1076–1092
15. Burrows AD (2004) In: Mingos DMP (ed) Supramolecular
assembly via hydrogen bonds I. Structure and bonding, vol 108.
Springer, Berlin-Heidelberg, pp 55–95
16. Fitzgerald LJ, Gerkin RE (1997) Acta Crystallogr C53:1080
17. Desiraju GR (1995) Angew Chem Int Ed 34:2311
18. Eißmann F, Schindler D, Weber E (2010) Struct Chem 21:245
In summary, on the basis of the two simple compounds
1 and 2, it is shown that carboxylic acids are prevented
from creation of the typical carboxylic dimer synthon [15]
in the crystalline state (e.g. existent in the crystal of
anthracene-1-carboxylic acid [16]) if a strong hydrogen
bond acceptor molecule is present such as DMSO acting as
successful competitor. Under these circumstances, changed
structures are formed whilst maintaining, for example,
p-stacking as a subordinate synthon mode [17]. A similar
situation has recently been described with the DMSO
complexes of certain nucleobase derivatives [18].
123

 Gruber, Tobias
Gruber, Tobias
 Helas, Stefanie F.
Helas, Stefanie F.
 Seichter, Wilhelm
Seichter, Wilhelm
 Weber, Edwin
Weber, Edwin