
Journal of Physical Organic Chemistry p. 585 - 592 (2009)
Update date:2022-08-10
Topics:
 Jarmoumi, Chakir
Jarmoumi, Chakir
 Lakhrissi, Brahim
Lakhrissi, Brahim
 Mondieig, Denise
Mondieig, Denise
 Negrier, Philippe
Negrier, Philippe
 Leger
Leger
 Massip
Massip
 Lazard, Zhor
Lazard, Zhor
 Benali, Bouziane
Benali, Bouziane
 Massoui, Mohamed
Massoui, Mohamed
 Essassi, El Mokhtar
Essassi, El Mokhtar
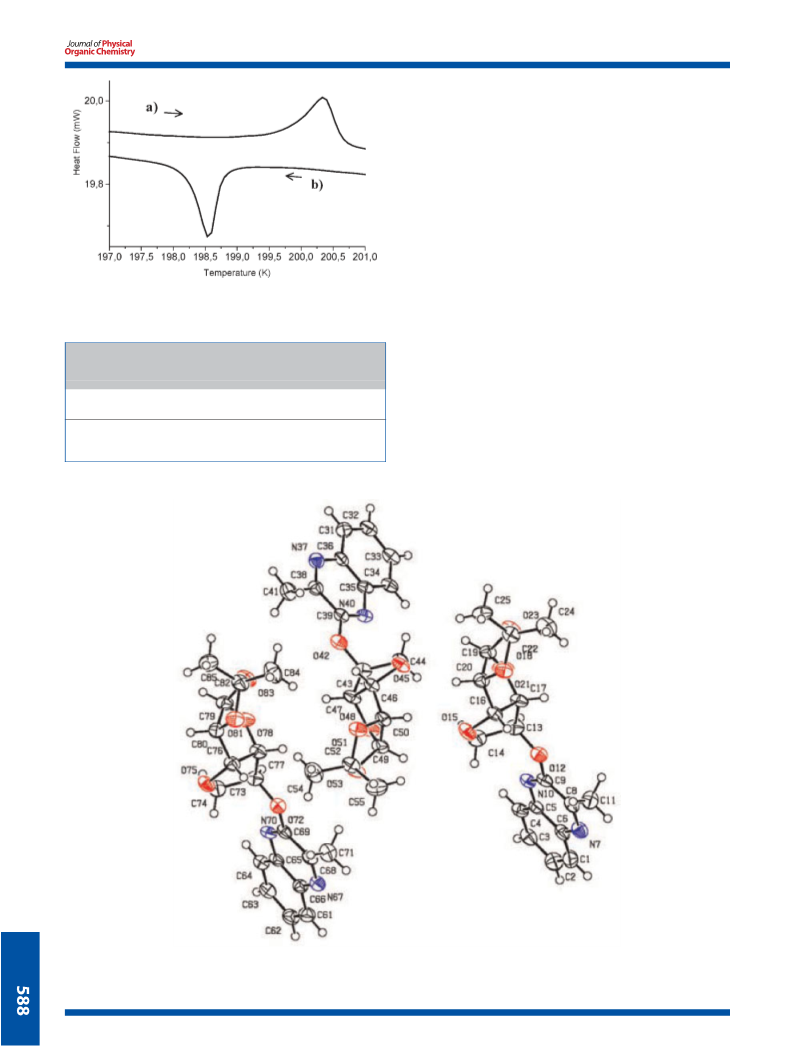
Reaction of 3-methyl-2(1H)-quinoxalinone (4) and 2(1H)-quinoxalinone (5) with 5,6-anhydro-1,2-O-isopropylidene-α-D-glucofuranose 6 gives the unexpected O-glucoquinoxalines derivatives by the intermediary novel intramolecular rearrangement of 5,6-anhydro-1,2-O-isopropylidene-α-D- glucofuranose to the corresponding 3,6-anhydro form. The obtained O-glucoquinoxalines 7,8 were identified by NMR spectroscopy. The X-ray crystal structures have been determined at room temperature. Moreover, a solid-solid phase transition has been detected at 198.9 K for O-glucoquinoxalines 7 and the structure of the low-temperature phase has been solved at 188K.
View More






Hangzhou Think Chemical Co. Ltd
website:http://www.thinkchem.com/
Contact:86-571-89986307/81956191/81956084/81956192
Address:Room 501-502, Tower E, Yuanjian Building, Yuanyang International Center, Hangzhou-310011, China
Tianjin Anda North Industrial & Business Co.Ltd.
Contact:86-22-24999306
Address:No.11 Erwei Road,Dongli Development Area,Tianjin,China
Luojiang Chenming Biological Products Co
Contact:+86 15000297032
Address:GROUP NO.4, HE SHENG VILLAGE, PANLONG TOWN,
Suzhou Chiral Pharmaceuticals Co., Ltd.
Contact:86-0512-63197058
Address:Building A, No. 2358, Chang'an Road, Wujiang Science Park
Yancheng Creator Chemical Co., Ltd
Contact:0086-515-88710008 88710068 88710858 88710868
Address:No.21 Renming Road, Longgang Town, Yandu County,Yancheng City
Doi:10.1016/j.poly.2014.06.002
(2014)Doi:10.1016/S0040-4039(00)00165-9
(2000)Doi:10.1016/j.chemphyslip.2016.08.004
(2016)Doi:10.1002/1521-3773(20020301)41:5<777::AID-ANIE777>3.0.CO;2-7
(2002)Doi:10.1021/ja01107a038
(1953)Doi:10.1016/0957-4166(94)80074-X
(1994)