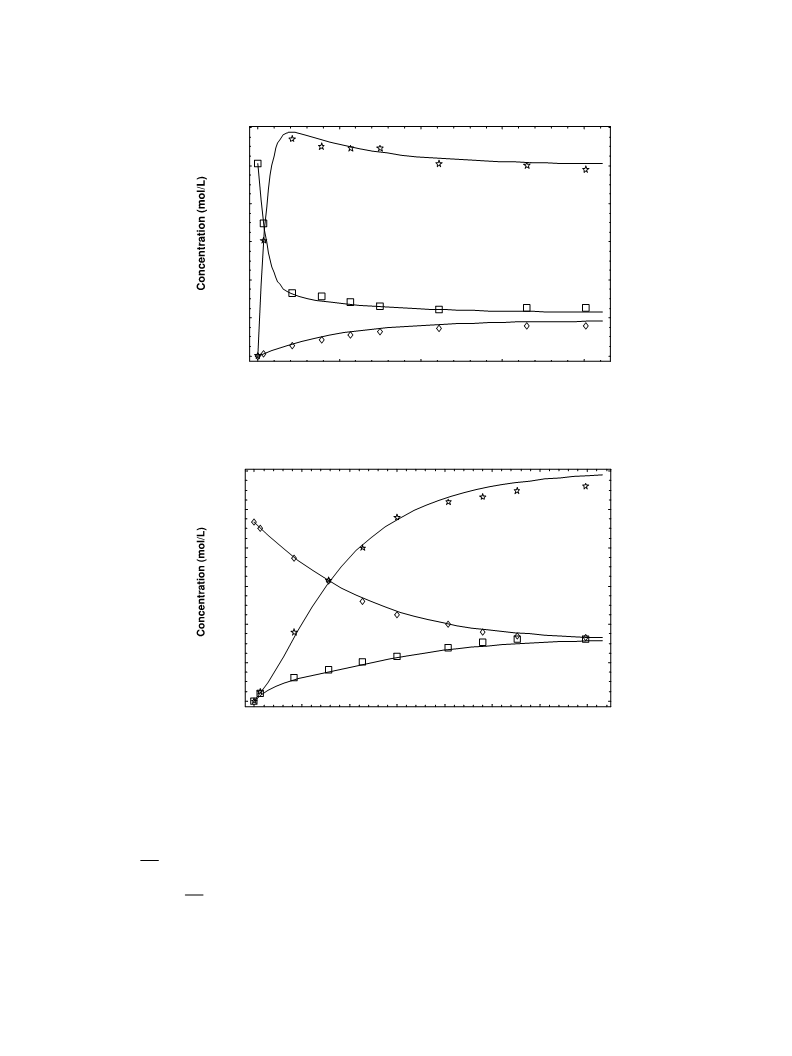
112
QUARTARONE ET AL.
Table III Kinetic Constants Calculated by Nonlinear Fitting of the Experimental Data
103 Initial Concentration
102 kD
(L mol−1 s−1
10−3 kDR
(s−1
10−2 kT
(L mol−1 s−1
10−3 kTR
(s−1
(mol L−1
)
)
)
)
)
ε kDa
104 ε kTa
108 σ2
1
1
1
3
4
3.0
5.0
6.3
1.01
0.94
2.65
2.59
2.26
3.09
1.71
1.79
1.75
1.53
2.08
1.15
9.11
9.72
8.63
8.41
8.63
9.26
9.87
8.75
8.55
8.75
1.1
1.6
1.5
1.0
4.3
9.8
12
19
6.6
9.1
2.8
3.2
8.5
0.36
0.6
aThe correlation matrix values of the regression variables (kD and kT) are comprised between 0.1 and 0.4.
activity coefficient factor is practically constant in the
range investigated.
6. Quartarone, G.; Bellomi, T.; Zingales, A. Corros Sci
2003, 45, 715.
7. Quartarone, G.; Bonaldo, L.; Tortato, C. Appl Surf Sci
2006, 252, 8251.
8. Du¨du¨kcu¨, M.; Koleli, F. Prog Org Coatings 2006, 55,
324.
9. Popova, A.; Christov, M.; Zwetanova, A. Corros Sci
2007, 49, 2131.
10. Ishii, H.; Murakami, K.; Sakurada, E.; Hosoya, K.;
Murakami, Y. J Chem Soc, Perkin Trans I 1988, 2377.
11. Wu, Y.; Lobeck, W. G., Jr.; Ryan, R. P. J Med Chem
1972, 15, 529.
12. Oddo, B. Gazz Chim Ital 1913, 43, 385.
13. Schmitz-Dumont, O. Ber Deut Chem Ges 1930, 63, 323.
14. Noland, W. E.; Hammer, C. F. J Org Chem 1960, 25,
1525.
Except indole protonation, which is in stationary
state, the faster step is the formation of 3 that is the
direct consequence of the high reactivity of the carbo-
cation (2). The values of the kinetic constant (kD) are
comparable with those measured by other authors for
the additions of benzhydrylium ions to indoles [17]. As
expected, the nucleophilic attack of indole to the proto-
nated indole dimer (3) is much slower because the pos-
itive charge is mainly localized on the nitrogen atom,
which is, in this case, a secondary ammonium ion.
Furthermore, the protonated 2-alkyl-aniline (4) does
not react with (1), since the positive charge is on the
nitrogen atom and the anilinium ion is ineffective as
electrophile. Nevertheless, under different experimen-
tal conditions, Plancher rearrangement, additions and
aromatization reactions occur [3,4,10,18].
15. Pal, B.; Giri, V. S.; Jaisankar, P. Catal Commun 2005, 6,
711.
16. Terrier, F.; Pouet, M. J.; Halle, J. C.; Hunt, S.; Jones,
J. R. J Chem Soc, Perkin Trans 2 1993, 1665.
17. Lakhdar, S.; Westermayer, M.; Terrier, F.; Goumont, R.;
Boubaker, T.; Ofial, A. R.; Mayr, H. J Org Chem 2006,
71, 9088.
The complete thermodynamic and kinetics analysis
of the reactions are in course of study.
18. Bocchi, V.; Palla, G. Tetrahedron 1986, 42, 5019.
19. Bates, D. M.; Watts, D. G. Nonlinear Regression Anal-
ysis & Its Applications; Wiley: New York, 1988.
20. Mathews, J. H. Numerical Methods for Computer Sci-
ence, Engineering and Mathematics; Prentice-Hall In-
ternational: Englewood Cliffs, NJ, 1987.
21. Hinman, R. L.; Lang, J. J Am Chem Soc 1964, 86, 3796.
22. Marziano, N. C.; Cimino, G.; Passerini, R. J Chem Soc,
Perkin Trans 2 1973, 1915.
We thank Dr. Laura Sperni for mass spectra, Sig. Augusto
Tassan for NMR spectra and for elemental analysis, and
Dr. Marco Bortoluzzi for the helpful suggestions and dis-
cussions.
BIBLIOGRAPHY
23. Hoyuelos, F. J.; Garc´ıa, B.; Ibeas, S.; Mun˜oz, M. S.;
Navarro, A. M.; Pen˜acoba, I.; Leal, J. M. Eur J Org
Chem 2005, 1161.
24. Andonowski, B. S.; Stojkovich, G. M. Acta Chim Slov
2000, 47, 349.
25. Marziano, N. C.; Tommasin, A.; Tortato, C.; Isandelli,
P. J Chem Soc, Perkin Trans 2 1998, 2535.
26. Robertson, E. B.; Dunford, H. B. J Am Chem Soc 1964,
86, 5080.
27. Chen, H.; Irish, D. E. J Phys Chem 1971, 75, 2672.
28. Sykes, P. A Guidebook to Mechanism in Organic Chem-
istry, 6th ed.; Longman: London, 1985; p. 66.
1. Penieres-Carrill, G.; Garcıa-Estrada, J. G.; Gutierrez-
Ramırez, J. L.; lvarez-Toledano, C. Green Chem 2003,
5, 337.
2. Restivo, A. M.; Arslan, A. A.; Goldberg, G. L. Obstet
Gynecol 2005, 105, 45.
3. Sundberg, R. J. Indoles; Academic Press: San Diego,
CA, 1996.
4. Robertson, N.; Parsons, S.; MacLean, E. J.; Coxall,
R. A.; Mount, A. R. J Mater Chem 2000, 10, 2043.
5. Otero, N.; Mandado, M.; Mosquera, R. A. J Phys Chem
A 2007, 111, 5557.
International Journal of Chemical Kinetics DOI 10.1002/kin

 Quartarone
Quartarone
 Ronchin
Ronchin
 Tortato
Tortato
 Vavasori
Vavasori