upon the identity of 1 and the solvent composition as found in
the hydrogen peroxide reaction (see later). It may thus be
regulated by varying the identity of the selenoxide and/or
changing the composition of the medium to suit the hydro-
genation of alkenes with varying reactivities.
quently falls off to about one third. At the latter stage 77Se NMR
shows 8 and 1 in the approximate ratio 2+1 thus indicating a
slow reoxidation. The reaction of 1b in methanol is acid
catalysed while that of 1a is not. This indicates a rate
determining addition step for 1b, at least in the absence of acid,
in accord with the report that the racemization of optically
active 1b, assumed to occur by reversible selenurane formation,
requires acid catalysis6 while 1a is racemized by water
alone.11,12
In THF the reactions of both 1a and 1b are acid catalysed.
This is hardly surprising. The formation of 7 is not an
elementary process but also involves a proton transfer step
which requires a proton donating component in the medium.
Two observations suggest that the elimination step may be rate
determining at high reaction rates. The reactions of 1a in neutral
and 1a and 1b in acidic methanol are more strongly inhibited by
water than the reactions of 1b in neutral methanol and 1a in
THF. Moreover, the order of reactivities 1a+1b is reversed in
acidic 94% methanol.
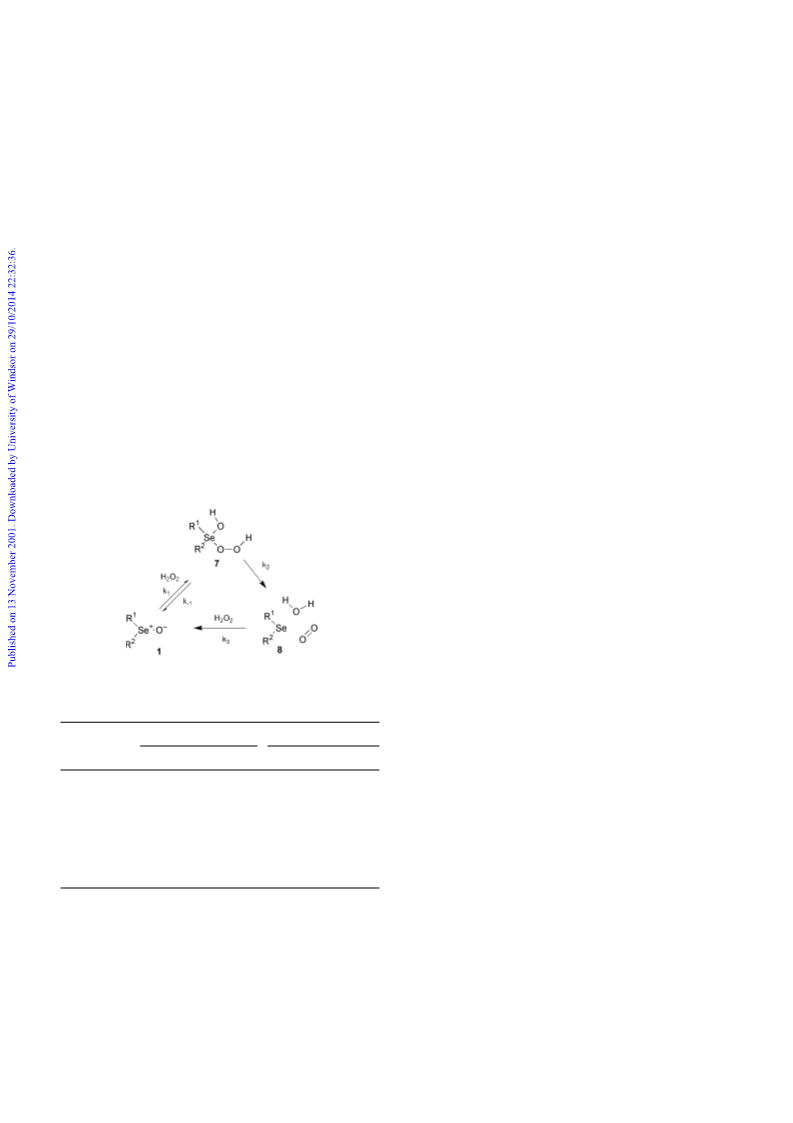
While the three-stage mechanism of hydrogen peroxide
decomposition seems established it cannot in the absence of
stereochemical information be finally shown that the elimina-
tion step is pericyclic. However, the observation that triethyl-
amine does not increase the initial rate in the reaction of 1a is a
strong argument against an acyclic, base or solvent assisted
reaction. The dissociative cycloelimination step calls for the
oxygen molecule to be produced in the singlet state. We were
unable to detect oxygen consumption by singlet oxygen
acceptors such as furanes. However, this cannot be taken as an
argument against the pericyclic reaction. The oxygen molecule
is generated in a solvent cage together with a molecule of 8
which, in analogy with diethyl sulfide13 is expected to be a
powerful quenching agent for singlet oxygen.
Since the kinetics of the hydrazine oxidation are not simple
we turned to the reaction of 1 with hydrogen peroxide in order
to obtain additional information on the medium dependence of
the addition–cycloelimination sequence. It has been reported9
that hydrogen peroxide is disproportionated into water and
dioxygen with selenoxides as catalysts. We suggest that the
catalytic cycle involves a reversible addition of hydrogen
peroxide to 1 with formation of selenurane (7), a cycloelimina-
tion giving dioxygen and selenide (8) and a reoxidation step
(Scheme 5). This sequence is supported by the observation10
that tert-butyl hydroperoxide lacking the vicinal hydrogen atom
necessary for the elimination does not give a similar reaction
and by our observation that 8 is present in the reaction mixtures
at steady-state concentrations depending on the identity of the
medium. The rate of oxygen evolution is first order in hydrogen
peroxide with an apparent rate constant proportional to the
concentration of the selenoxide. Some representative second
order rate constants are given in Table 2. This type of kinetics
can be reconciled with either of three situations: (i) rate
determining addition to give 7 (k1); (ii) rate determining
elimination to give 8 (k2) preceded by the formation of 7 in a
rapid equilibrium, (iii) rate determining reoxidation to 1 (k3).
The three steps appear to have comparable rates and depending
upon the medium either one may be rate determining. The
concentration of 8 is very low in neutral or acidic methanol
showing that k3 cannot be rate determining. Addition of
triethylamine gives an unchanged initial rate which subse-
In conclusion, a dissociative cycloelimination reaction is
indicated by the stereospecific oxidation of hydrazine with 1 to
give cis-2. The same mechanism is compatible with the features
of the selenoxide (or selenide) catalysed disproportionation of
hydrogen peroxide in which case the disproportionation
releases no reactive intermediates into the reaction medium, a
feature of possible interest in selenium biochemistry.
Notes and references
1 K. B. Sharpless and R. F. Lauer, J. Am. Chem. Soc., 1973, 94, 7154.
2 K. B. Sharpless and R. F. Lauer, J. Am. Chem. Soc., 1974, 95, 2697.
3 D. Neville Jones, D. Mundy and R. D. Whitehouse, J. Chem. Soc.,
Chem. Commun., 1972, 86.
4 L. Henriksen, Tetrahedron Lett., 1994, 35, 7057.
5 L. Henriksen and N. Stuhr-Hansen, Phosphorus, Sulfur, 1998, 136–138,
175.
6 N. Stuhr-Hansen, H. O. Sørensen, L. Henriksen and S. Larsen, Acta
Chem. Scand., 1997, 51, 1186.
7 D. J. Pasto and R. T. Taylor, Org. React., 1991, 40, 91.
8 K. Kondo, S. Murai and N. Sonoda, Tetrahedron Lett., 1977, 42,
3727.
9 H. J. Reich, S. Wollowitz, J. E. Trend, F. Chow and D. F. Wendelborn,
J. Org. Chem., 1978, 43, 1697.
Scheme 5
Table 2 Second order rate constantsa (102 k/s21 M21 = 102 kobs [1]21) for
the selenoxide catalyzed decomposition of hydrogen peroxide at 300 K
Selenoxide
Conditions
1a
1b
Neutral
4M AcOH
Neutral
4M AcOH
Solvent:
94% MeOH
80% MeOH
60% MeOH
3.7
1.5
0.5
3.7
1.7
0.5
0.60
0.43
0.18
5.2
1.5
0.55
10 T. Hori and K. B. Sharpless, J. Org. Chem., 1978, 43, 1689.
11 F. Davis, J. Billmers and O. Stringer, Tetrahedron Lett., 1983, 24,
3191.
96% THF
90% THF
84% THF
a Reproducibility 20%.
2.0
2.0
2.0
5.8
4.2
3.2
0.36
0.95
0.50
12 F. Davis, O. Stringer and J. McCauley, Jr, Tetrahedron, 1985, 41,
4747.
13 R. W. Denny and A. Nickon, Org. React., 1973, 20, 133.
Chem. Commun., 2001, 2448–2449
2449

 Henriksen
Henriksen
 Jakobsen
Jakobsen