3 8
H
in diluted feed mixtures.11 The observed
and 773 K, with propene yields < 10% and traces of
propionaldehyde. The relatively low yield of these products
over steamed H-ZSM-5 strongly supports that iron is predom-
inantly responsible for the observed performance, as concluded
CO and C
behaviour could be attributed to the different intrinsic mecha-
nism of these processes: in direct N O decomposition or SCR of
O, removal of atomic oxygen from the catalyst surface is rate
determining, while propane activation determines the rate of
the ODP reaction. What is clear from our results is that
extraframework iron species in FeMFI catalysts are active and
2
N
2
12
in other N
position or selective oxidation of benzene to phenol.
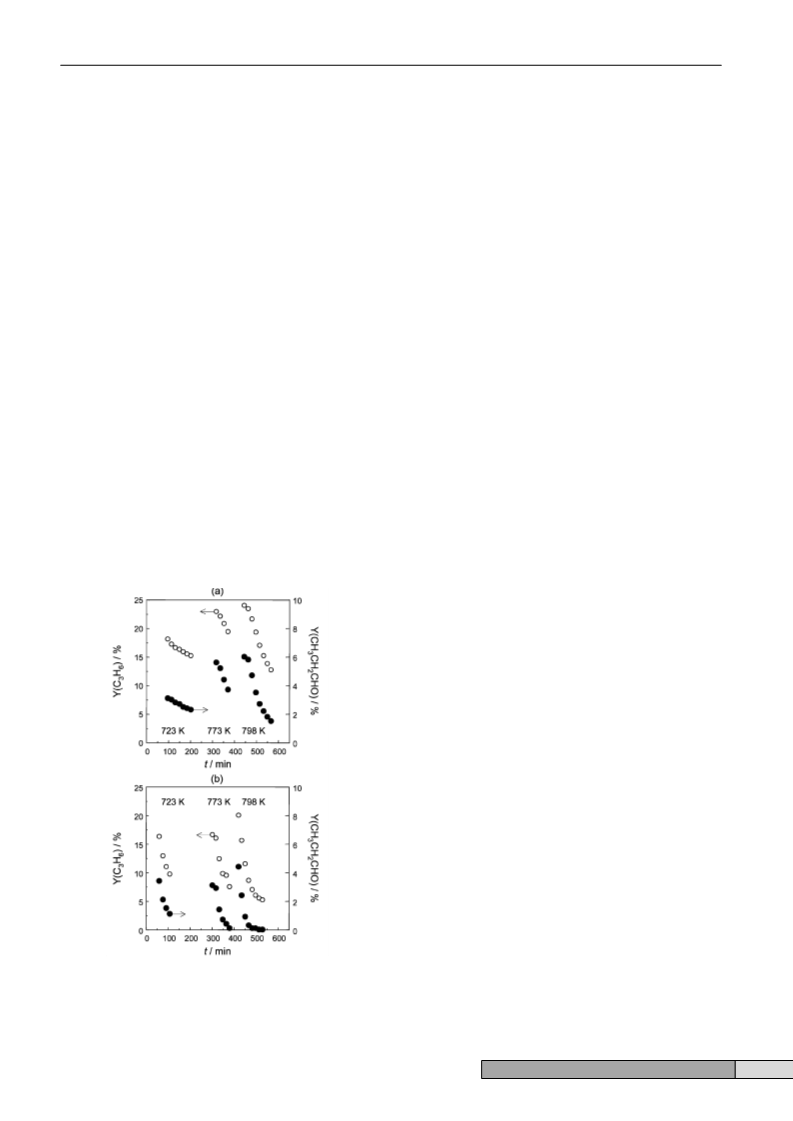
Fig. 1 shows the yield of the iron zeolites vs. time-on-stream
at different temperatures. The conversion of C and N
2
O conversions over Fe-zeolites, e.g. direct decom-
5,9,10
3
H
8
2
O
selective towards propene in ODP with N
2
O. In contrast, Uddin
et al. claimed that only framework iron of silicalite is selective
towards the olefin in the ODP with O . The similar initial
6
continuously declines in a period of 100 min, due to coke
formation. As a consequence, the high initial yield towards
propene and propionaldehyde progressively decreases. The rate
of deactivation over FeZSM-5 is significantly lower at 723 K
than at higher temperatures, in agreement with the decreased
carbon balance upon increasing temperature in Table 1. Fe-
silicalite is very sensitive to deactivation, even at 723 K. Both
catalysts completely recovered the initial activity at all the
temperatures after pretreatment in pure oxygen at 773 K for 30
min. This was checked upon by repetition of five successive
2
performance of the Fe-zeolites also indicates that Brønsted and
Lewis acidity in steam-activated FeZSM-5 plays a minor role in
the N O-mediated ODP reaction. The faster deactivation over
2
Fe-silicalite compared to FeZSM-5 is remarkable, especially at
723 K (see Fig. 1). The acidity in FeZSM-5 should in principle
accelerate the deactivation process due to coke formation, while
the opposite was observed. This is tentatively attributed to the
presence of mesopores in FeZSM-5, due to dealumination upon
steam treatment, which are absent in Fe-silicalite.8 The more
open structure of FeZSM-5 may reduce pore blocking by coke
and also improve transport of reactants and products, retarding
deactivation.
,9
2
ODP-reaction and O -regeneration cycles.
From the results above, it can be concluded FeZSM-5 is more
active and selective than Fe-silicalite. However, the difference
in performance between both catalysts is marginal if their
constitution with respect to iron is taken into account. Steam-
activated Fe-silicalite does not show any sign of iron associa-
tion, and the majority of extraframework iron species are
uniform and well isolated, with a small fraction of iron in
framework positions.11 In contrast, significant iron clustering
was observed in steam-activated FeZSM-5, where extraframe-
work isolated iron ions and oligonuclear species in the zeolite
channels coexist with iron oxide nanoparticles of 1–2 nm.8
Our results suggest that the markedly different nature and
distribution of extraframework iron species in the steam-
activated catalysts does not have a strong influence on the initial
ODP performance. This observation contrasts with the active
site structure sensitivity exhibited by these catalysts in other
Our results show that steam-activated FeMFI zeolites are
highly efficient catalysts for oxidative dehydrogenation of C
with N O using high partial reactant pressures. Initial propene
yields of 21–24% at 773–798 K are very similar to state-of-the-
art catalysts based on V and Mo oxides with O . Over vanadia-
based catalysts, N O is a less effective oxidant than O to obtain
high propene yields,13 corroborating the superior performance
of FeMFI with N O. Another feature of FeMFI relates to the
3 8
H
2
2
2
2
,9
2
high propionaldehyde yield, since this product has been never
observed in ODP over typical oxide systems.
This novel application of FeMFI leads to the simultaneous
functionalisation of propane, a relatively cheap and abundant
feedstock, and utilisation of N
gas. This concept can be economically applied in tail-gases of
specific chemical processes, e.g. those using HNO as oxidizing
agent, where N O is produced in high concentration (typically
5–40 vol.%). This approach has been proposed for the
selective oxidation of benzene to phenol with N O in tail-gases
2
O, an environmentally harmful
2
N O conversions, including direct decomposition and SCR with
3
2
2
2
from adipic acid plants.5 An apparent drawback of steam-
activated FeMFI catalysts in ODP is the rapid deactivation by
coke. This issue can be easily overcome, since the initial
performance is completely recovered after oxygen pretreatment
at 773 K. The practical realisation of this process in continuous
mode can be accomplished using a configuration of two reactors
in parallel with alternating reaction–regeneration. Investiga-
tions in such a system are on the way.
Notes and references
1
2
3
4
O. Buyevskaya and M. Baerns, Catalysis, 2002, 155, 16.
C. Pak, A. T. Bell and T. Don Tilley, J. Catal., 2002, 206, 49.
R. B. Watson and U. S. Ozkan, J. Catal., 2000, 191, 12.
Y. Liu, J. Wang, G. Zhou, M. Xian, Y. Bi and K. Zhen, React. Kinet.
Catal. Lett., 2001, 73, 199.
5
6
G. I. Panov, CATTECH, 2000, 4, 18.
Md. A. Uddin, T. Komatsu and T. Yashima, J. Catal., 1994, 150,
4
39.
7
8
9
S. N. Vereshchagin, N. P. Kirik, N. N. Shishkina and A. G. Anshits,
Catal. Lett., 1998, 56, 145.
J. Pérez-Ramírez, G. Mul, F. Kapteijn, J. A. Moulijn, A. R. Overweg, A.
Ribera and I. W. C. E. Arends, J. Catal., 2002, 207, 113.
J. Pérez-Ramírez, F. Kapteijn, J. C. Groen, A. Doménech, G. Mul and
J. A. Moulijn, J. Catal., 2003, 214, 33.
1
1
0 J. Pérez-Ramírez, F. Kapteijn, G. Mul and J. A. Moulijn, Catal.
Commun., 2002, 3, 19.
1 J. Pérez-Ramírez, F. Kapteijn and A. Brückner, J. Catal., 2003, 218,
Fig. 1 Yield of (2) propene and (5) propionaldehyde vs. time-on-stream
234.
over steam-activated (a) FeZSM-5 and (b) Fe-silicalite at different
12 J. Pérez-Ramírez, F. Kapteijn, G. Mul and J. A. Moulijn, J. Catal., 2002,
208, 211.
13 E. V. Kondratenko and M. Baerns, Appl. Catal. A, 2001, 222, 133.
3 8 2
temperatures. Conditions: feed mixture of C H /N O/Ne = 10/10/80 and t
1
=
0.043 s gcat ml2
.
CHEM. COMMUN., 2003, 2152–2153
2153

 Perez-Ramirez, Javier
Perez-Ramirez, Javier
 Kondratenko, Evgueni V.
Kondratenko, Evgueni V.