Page 5 of 6
Green Chemistry
Please do not adjust margins
Journal Name
COMMUNICATION
DOI: 10.1039/D0GC00633E
Benignity and Efficient Resource Utilization, and National
Department of Education Engineering Research Centre for
Chemical Process Simulation and Optimization.
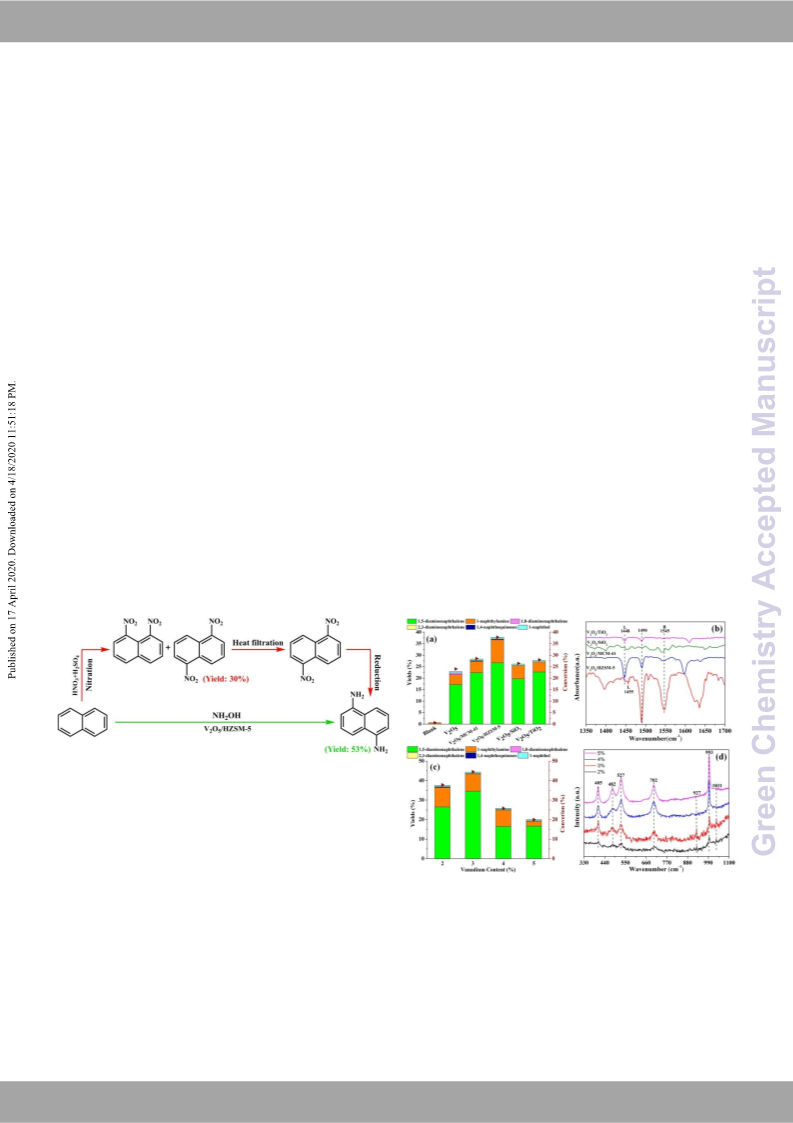
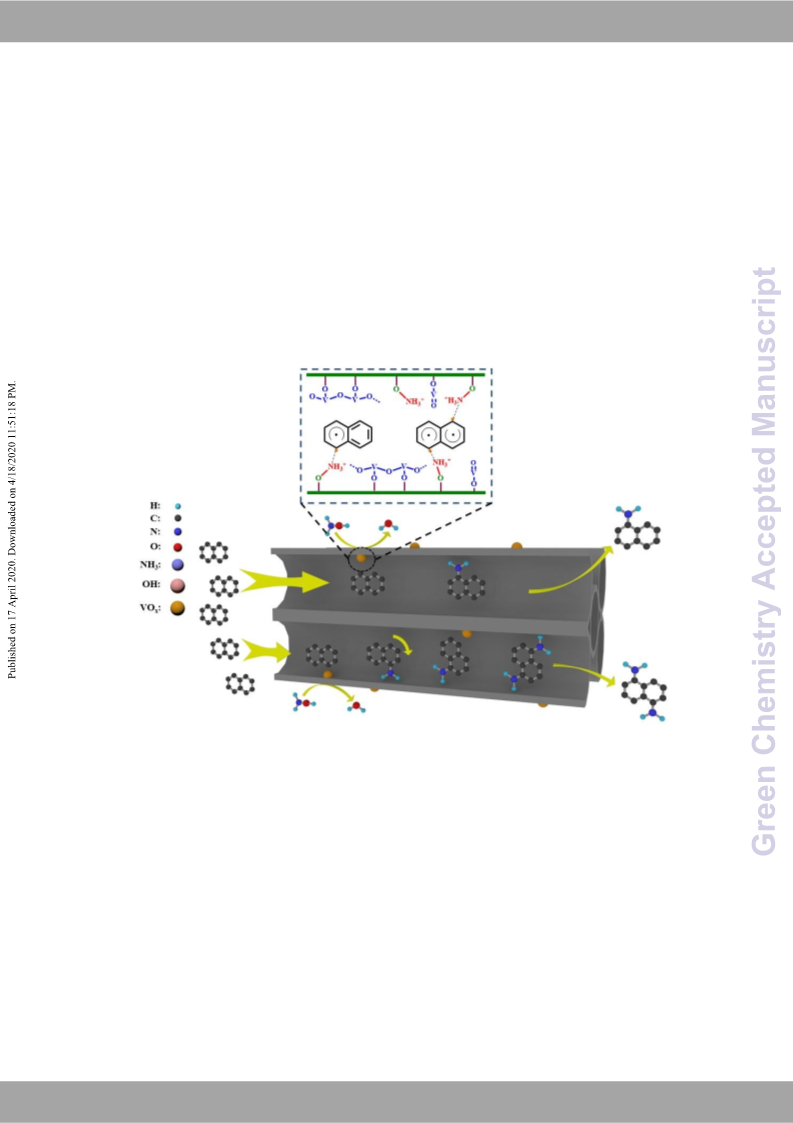
amino groups combine with different kinds of naphthalene free
radicals to form corresponding naphthylamine and other oxidative
products. Affected by steric hindrance, the heterocyclic activation
and homocyclic passivate effector by the protonated amino radical
+
ion (-NH3 ), the formations of 1-naphthylamine and 1,5-
diaminonaphthalene are relatively easier than other positions of
naphthalene ring. It can also explain why there are few amounts of
2,3-diaminonaphthalene and 1,8-diaminonaphthalene formed
Notes and references
during the reaction.
Additionally, considered the control
1
a) W. J. Chen, Y. K. Kang, G. W. Richard and S. Daniel, Angew.
Chem. Int. Ed., 2014, 53, 5179-5182; b) G. Dyker, Angew.
Chem. Int. Ed., 1999, 38, 1698-1712; c) H. Kogelnik, N. Adam
and E. Prolingheuer, CeIlarPolymers 1992, 38, 45-54.
H. U. Blank, F. Durholz and G. Skipka, US 4053526, 1977.
G. Zhu, Y. Yi, Z. Han, K. Wang and X, Wu, Anal. Chim. Acta,
2014, 845, 30-37.
experiment in Table S3, it shows that 1-naphthylamine could be
converted to 1,5-diaminonaphthalene during the one-step
naphthalene amination reaction. Comparison with the results when
naphthalene is used as the starting material, it also indicates that
1,5-diaminonaphthalene formation is mainly from the one-step
amination of naphthalene.
2
3
4
5
6
7
M. Xiong, H. Hu, X. Hu and Y. Liu, Org. Lett., 2018, 20, 3661–
3665.
O. Sepai, G. Sabbioni, Int. J. Occuo. Med. Env., 2017, 30, 579-
591.
J. A. Bikker, N. Brooijmans and A Wissner, J. Med. Chem.,
2009, 52, 1493-1509.
a) S. Sauer and W. Frank, Angew. Chem. Int. Ed., 2014, 53,
7428-7448; b) E.N. Squire and G. Mills, US 3919155,1975; c)
E.N. Squire, G. Mills, US 3929889,1975.
8
9
S. Christian and W. Gerhard, WO 9419310, 1994.
K. L. Paik, N. S. Baek, H. K. Kim, J. H. Lee and Y. Lee, Opt.
Mater., 2003, 21, 135-142.
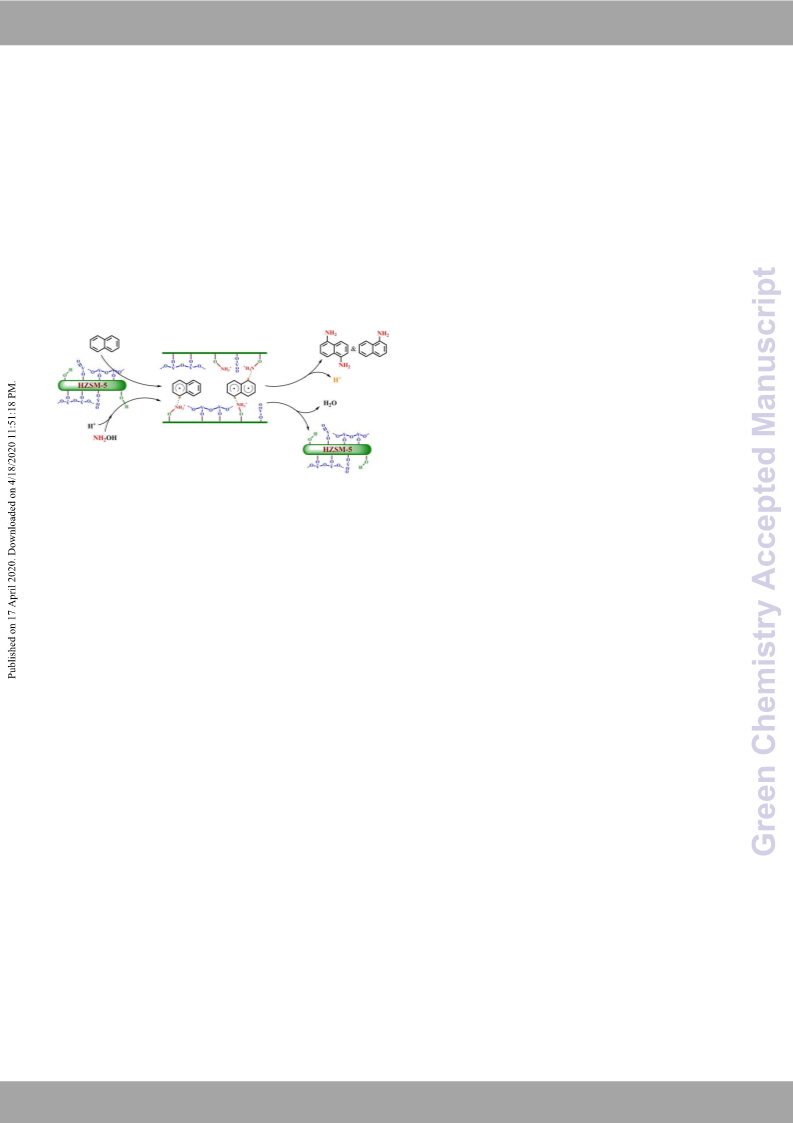
Scheme
2 The proposed reaction mechanism of naphthalene
10 H. G. Gilch, W. L. Wheelwright, J. Polym. Sci. Part A-1: Polym.
Chem., 1966, 4, 1337-1349.
amination over VOx/HZSM-5
11 J. P.Wibant, German Institute for Stardization, 1917, 50, 541-
546
12 P. Desrosiers, S. Guan and A. Hagemeyer, Appl. Catal., A,
Conclusions
2002, 227, 43-61.
13 N. Hoffmann and M. Muhler, Catal. Lett., 2005, 103, 155-
In conclusion, this work propose a promising naphthylamine
synthesis process by one-step amination of naphthalene with
hydroxylamine hydrochloride and vanadium oxide supported
catalysts under very mild conditions. The results show that 1,5-
159.
14 Y. A. Lisitsyn and A. V. Sukhov, Russ. J. Gen. Chem., 2017, 87,
929-933.
15 J. Becker and W. F. Hölderich, Catal. Lett., 1998, 54, 125-128.
diaminonaphthalene is the main naphthylamine product which is 16 M. A. Mantegazza, G. Leofanti, G. Petrini, M. Padovan and S.
Bordiga, Stud. Surf. Sci. Catal., 1994, 82, 541-550.
17 M. Nan, Y. Luo, G. Li and C. Hu, RSC Adv., 2017, 7, 21974-
21981.
18 N. I. Kuznetsova, L. I. Kuznetsova, L. G. Detusheva, V. A.
Likholobov and H. Cheng, Mol. Catal., 2000, 161, 1-9.
much valuable for producing its derivative of superior
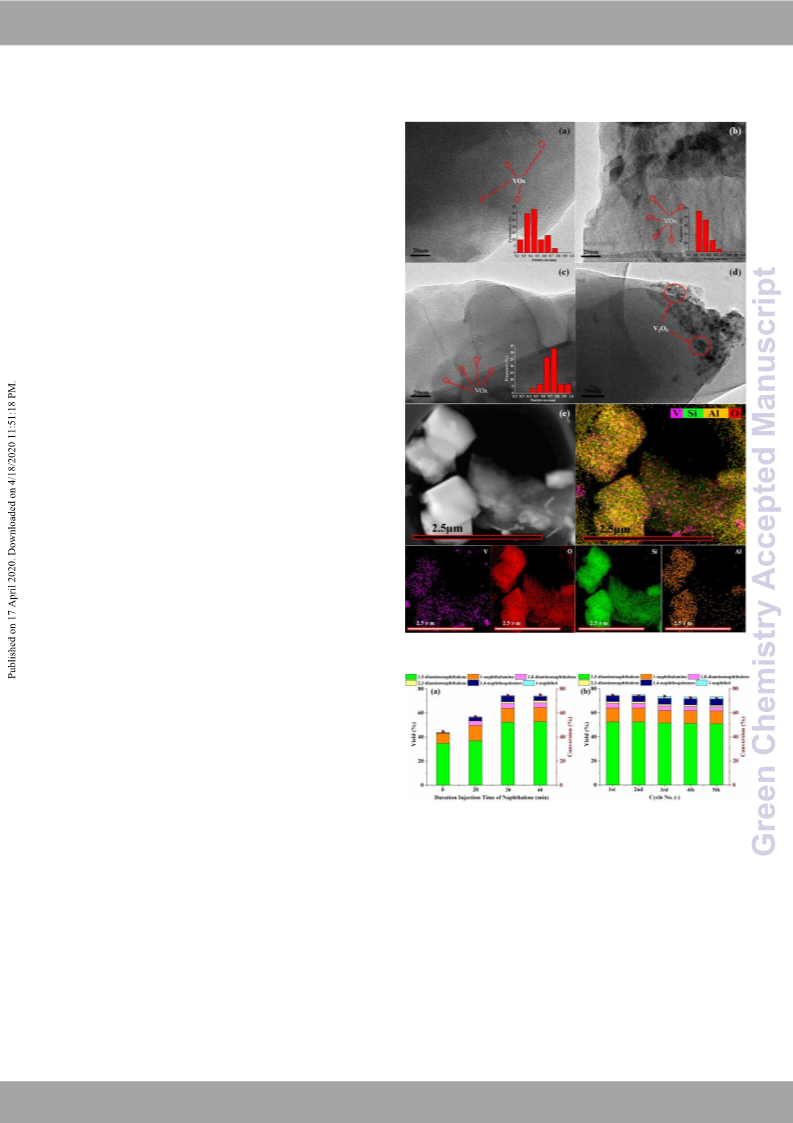
polyurethane. V2O5/HZSM-5 catalysts with high dispersion of
vanadium oxide nanoparticles exhibit high catalytic performance
+
due to the activation of hydroxylamine to form -NH3 by their
enriched Brönsted acid sites. Meanwhile, the polymerized V-O-V 19 L. F. Zhu, B. Guo, D. Y. Tang, X. K. Hu, G. Y. Li and C. W. Hu, J.
Catal., 2007, 245, 446-455.
20 K. M. Parida, S. S. Dash and S. Singha, Appl. Catal., A, 2008,
351, 59-67.
21 S. R. Waldvogel, S. MöHle, S. Herold, F. Richte and H.
Nefzger, ChemElectroChem., 2017, 4, 2196-2210.
structure and monovanadate V=O band in the catalyst can activate
the C-H bonds of naphthalene. The synergic effect of these specific
vanadium species and Brönsted acid of 3%V2O5/HZSM-5 obtains a
~70% yield of naphthylamine, in which 1,5-diaminonaphthalene and
1-naphthylamine yields are 52.3 % and 11.6 %, respectively. A 22 F. Zhang, X. Ren, H. Huang, J. Huang, M. Sudhakar and L. Liu,
Chin. J. Chem. Eng., 2018, 26, 1031-1040.
23 A. Alhanash, E. F. Kozhevnikova and I. V. Kozhevnikov, Appl.
Catal., A 2012, 378, 11-18.
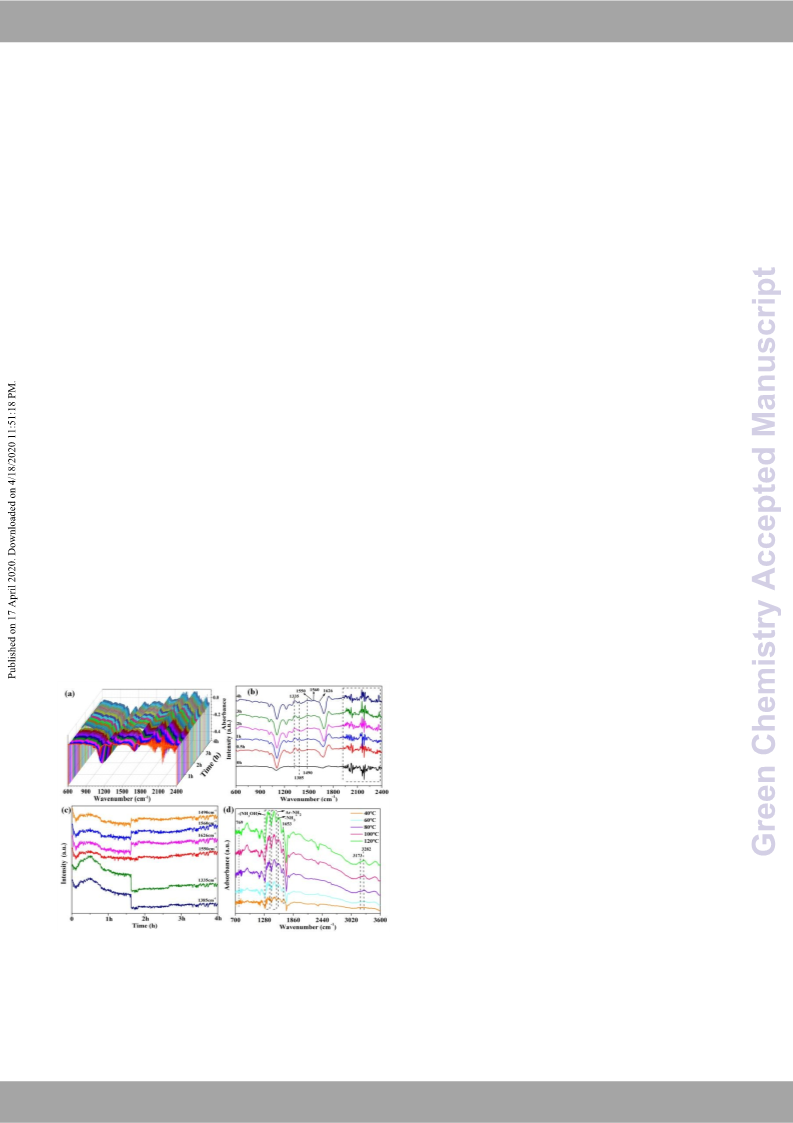
possible reaction mechanism is also proposed to combine with the
reaction results, real-time IR, and in-situ DRIFTs.
24 G. S. Foo, D. Wei, D. S. Sholl and C. Sievers, ACS Catal., 2014,
4, 3180-3192.
25 H. Kosslick, G. Lischke, G. Walther, W. Storek, R. Fricke,
Microporous Mater., 1997, 9, 13–33.
26 S. Kayal, J. Kikuchi, M. Shimizu and M. Terada, ACS Catal.,
2019, 9, 6846-6850.
Conflicts of interest
There are no conflicts to declare.
27 J. L. Zhang, L. Zhu, K. Shen, H. M. Yang, X. C. Hang and G. X.
Jiang, Chem. Sci., 2019, 10, 1070-1074.
28 S. A. Alghamdi and H. I. D. Lasa, Fuel, 2014, 128, 120-140.
29 a) H. Berndt, A. Martin, A. Brückner, E. Schreier, D. Müller
and H. Kosslick, J. Catal., 2000, 191, 384-400; b) Y. M. Liu, Y.
Acknowledgments
This work was supported by NSFC (21908185), Project of
Hunan Provincial Natural Science Foundation of China
(2016JJ2123, 2018JJ3497), Project of Hunan Provincial
This journal is © The Royal Society of Chemistry 20xx
J. Name., 2013, 00, 1-3 | 5
Please do not adjust margins

 Hao, Fang
Hao, Fang
 Wang, Xin
Wang, Xin
 Huang, Linfang
Huang, Linfang
 Xiong, Wei
Xiong, Wei
 Liu, Pingle
Liu, Pingle
 Luo, Hean
Luo, Hean