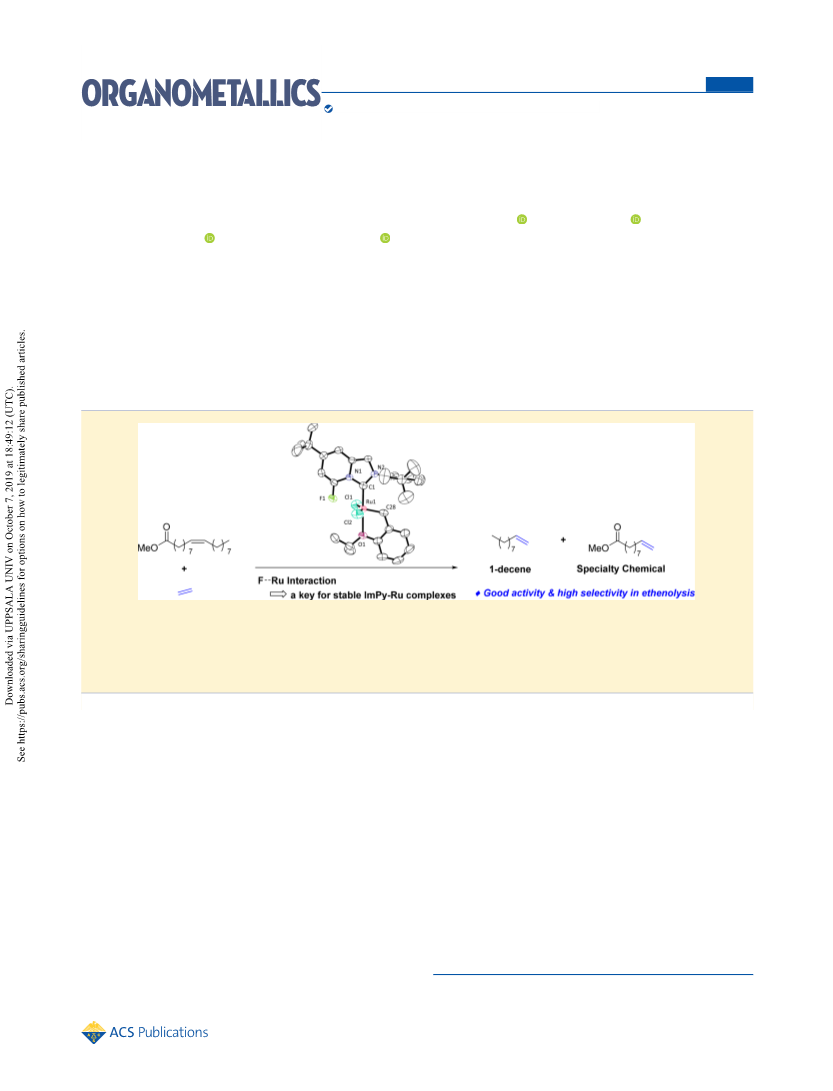
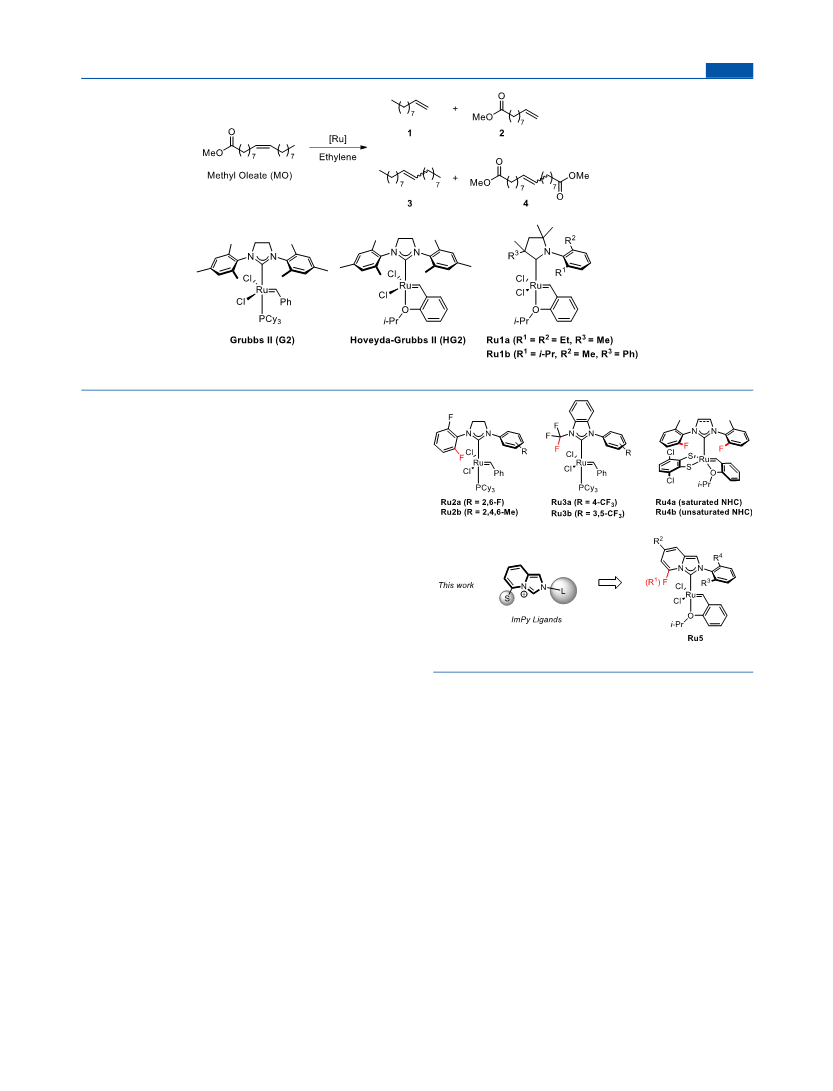
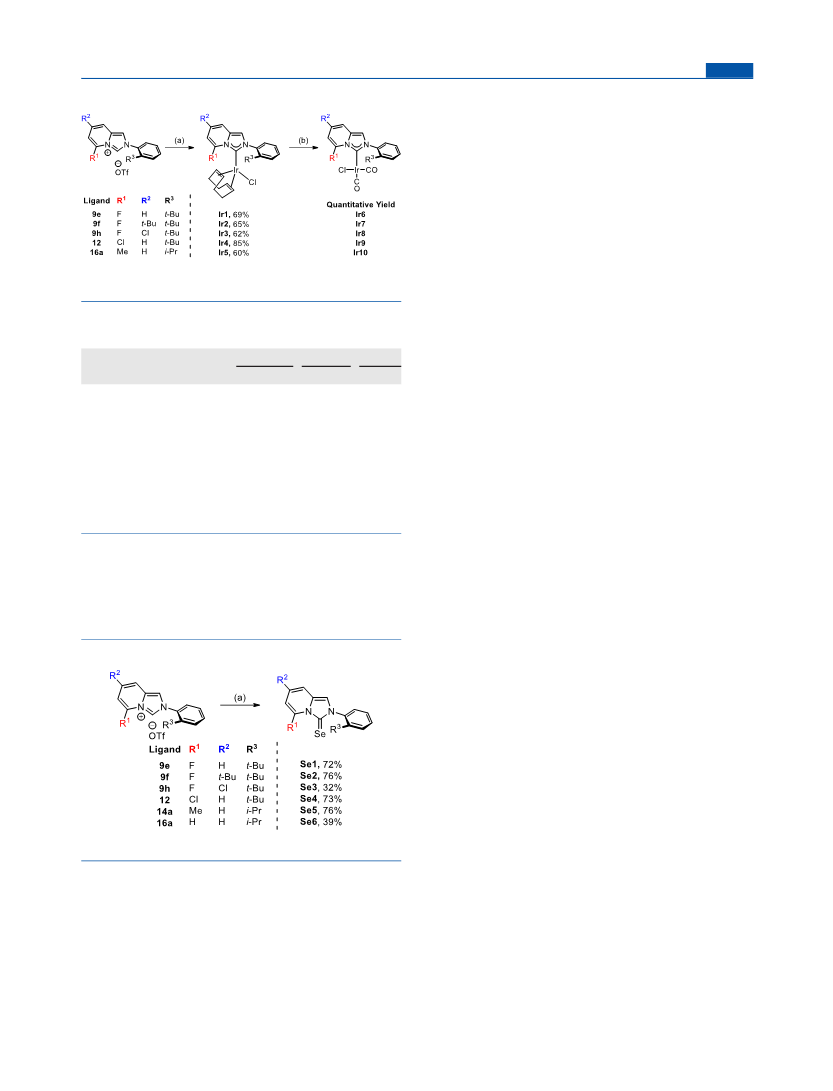
Organometallics
Article
A Highly Efficient Olefin Metathesis Process for the Synthesis of
(12) (a) Kingsbury, J. S.; Harrity, J. P. A.; Bonitatebus, P. J.;
Hoveyda, A. H. A Recyclable Ru-Based Metathesis Catalyst. J. Am.
Chem. Soc. 1999, 121, 791−799. (b) Garber, S. B.; Kingsbury, J. S.;
Gray, B. L.; Hoveyda, A. H. Efficient and Recyclable Monomeric and
Dendritic Ru-Based Metathesis Catalysts. J. Am. Chem. Soc. 2000, 122,
8168−8179. (c) Gessler, S.; Randl, S.; Blechert, S. Synthesis and
metathesis reactions of a phosphine-free dihydroimidazole carbene
ruthenium complex. Tetrahedron Lett. 2000, 41, 9973−9976.
Terminal Alkenes from Fatty Acid Esters. Top. Catal. 2012, 55, 518−
523. (e) Chikkali, S.; Mecking, S. Refining of Plant Oils to Chemicals
by Olefin Metathesis. Angew. Chem., Int. Ed. 2012, 51, 5802−5808.
(f) Mol, J. C. Catalytic Metathesis of Unsaturated Fatty Acid Esters
and Oils. Top. Catal. 2004, 27, 97−104. (g) Burdett, K. A.; Harris, L.
D.; Margl, P.; Maughon, B. R.; Mokhtar-Zadeh, T.; Saucier, P. C.;
Wasserman, E. P. Renewable Monomer Feedstocks via Olefin
Metathesis: Fundamental Mechanistic Studies of Methyl Oleate
Ethenolysis with the First-Generation Grubbs Catalyst. Organo-
metallics 2004, 23, 2027−2047.
(13) For recent reviews see: (a) Janssen-Muller, D.; Schlepphorst,
̈
C.; Glorius, F. Privileged chiral N-heterocyclic carbene ligands for
asymmetric transition-metal catalysis. Chem. Soc. Rev. 2017, 46,
4845−4854. (b) Peris, E. Smart N-Heterocyclic Carbene Ligands in
Catalysis. Chem. Rev. 2018, 118, 9988−10031. (c) Wang, M. H.;
Scheidt, K. A. Cooperative Catalysis and Activation with N-
Heterocyclic Carbenes. Angew. Chem., Int. Ed. 2016, 55, 14912−
14922. (d) Teator, A. J.; Lastovickova, D. N.; Bielawski, C. W.
Switchable Polymerization Catalysts. Chem. Rev. 2016, 116 (4),
1969−1992. (e) Lazreg, F.; Nahra, F.; Cazin, C. S. J. Copper−NHC
complexes in catalysis. Coord. Chem. Rev. 2015, 293−294, 48−79.
(f) Flanigan, D. M.; Romanov-Michailidis, F.; White, N. A.; Rovis, T.
Organocatalytic Reactions Enabled by N-Heterocyclic Carbenes.
Chem. Rev. 2015, 115, 9307−9387. (g) N-Heterocyclic Carbenes:
Effective Tools for Organometallic Synthesis; Nolan, S. P., Ed.; Wiley-
VCH: Weinheim, Germany, 2014. (h) Bellemin-Laponnaz, S.;
Dagorne, S. Group 1 and 2 and Early Transition Metal Complexes
Bearing N-Heterocyclic Carbene Ligands: Coordination Chemistry,
Reactivity, and Applications. Chem. Rev. 2014, 114, 8747−8774.
(i) Hopkinson, M. N.; Richter, C.; Schedler, M.; Glorius, F. An
overview of N-heterocyclic carbenes. Nature 2014, 510, 485−496.
(
7) (a) Olefin metathesis of renewable platform chemicals; de Espinosa,
L. M., Meier, M. A. R., Eds.; Springer: Berlin, Heidelberg, 2012.
b) Biermann, U.; Bornscheuer, U.; Meier, M. A. R.; Metzger, J. O.;
Schafer, H. J. Oils and Fats as Renewable Raw Materials in Chemistry.
(
̈
Angew. Chem., Int. Ed. 2011, 50, 3854−3871. (c) Behr, A.; Gomes, J.
P. The Refinement of Renewable Resources: New Important
Derivatives of Fatty Acids and Glycerol. Eur. J. Lipid Sci. Technol.
2010, 112, 31−50. (d) Marshall, A. L.; Alaimo, P. J. Useful Products
from Complex Starting Materials: Common Chemicals from Biomass
Feedstocks. Chem. - Eur. J. 2010, 16, 4970−4980. (e) The Biorefinery
Introduction to Chemicals from Biomass; Clark, J. H., Deswarte, F. E. I.,
Eds.; Wiley-VCH: Weinheim, Germany, 2008. (f) Behr, A.;
Westfechtel, A.; Gomes, J. P. Catalytic Processes for the Technical
Use of Natural Fats and Oils. Chem. Eng. Technol. 2008, 31, 700−714.
(
g) Clavier, H.; Grela, K.; Kirschning, A.; Mauduit, M.; Nolan, S. P.
Sustainable Concepts in Olefin Metathesis. Angew. Chem., Int. Ed.
007, 46, 6786−6801. (h) Corma Canos, A.; Iborra, S.; Velty, A.
2
Chemical Routes for the Transformation of Biomass into Chemicals.
Chem. Rev. 2007, 107, 2411−2502. (i) Mol, J. C. Application of
Olefin Metathesis in Oleochemistry: An Example of Green Chemistry.
Green Chem. 2002, 4, 5−13.
(j) Dro
Carbenes. Angew. Chem., Int. Ed. 2010, 49, 6940−6952. (k) Nolan, S.
P.; Marion, N.; Díez-Gonzalez, S. N-Heterocyclic Carbenes in Late
̈
ge, T.; Glorius, F. The Measure of All RingsN-Heterocyclic
́
(
8) (a) Biobased Lubricants and Greases: Technology and Products;
Honary, L. A. T., Richter, E., Eds.; Wiley-VCH: Weinheim, Germany,
011. (b) Lubricants and Lubrication; Mang, T., Drese, W., Eds.;
Transition Metal Catalysis. Chem. Rev. 2009, 109, 3612−3676.
(l) Kantchev, E. A. B.; O’Brien, C. J.; Organ, M. G. Palladium
Complexes of N-Heterocyclic Carbenes as Catalysts for Cross-
Coupling ReactionsA Synthetic Chemist-s Perspective. Angew.
Chem., Int. Ed. 2007, 46, 2768−2813.
2
Wiley-VCH: Weinheim, Germany, 2005. (c) Chum, P. S.; Swogger,
K. W. Olefin Polymer Technologies-History and Recent Progress at
The Dow Chemical Company. Prog. Polym. Sci. 2008, 33, 797−819.
(14) (a) Vougioukalakis, G. C.; Grubbs, R. H. Ruthenium-Based
Heterocyclic Carbene-Coordinated Olefin Metathesis Catalysts.
Chem. Rev. 2010, 110, 1746−1787. (b) Samojłowicz, C.; Bieniek,
M.; Grela, K. Ruthenium-Based Olefin Metathesis Catalysts Bearing
N-Heterocyclic Carbene Ligands. Chem. Rev. 2009, 109, 3708−3742.
(9) Ruthenium examples: (a) Engl, P. S.; Santiago, C. B.; Gordon, C.
P.; Liao, W. C.; Fedorov, A.; Coperet, C.; Sigman, M. S.; Togni, A.
́
Exploiting and Understanding the Selectivity of Ru-N-Heterocyclic
Carbene Metathesis Catalysts for the Ethenolysis of Cyclic Olefins to
α,ω-Dienes. J. Am. Chem. Soc. 2017, 139, 13117−13125. (b) Engl, P.
(15) (a) Małecki, P.; Gajda, K.; Gajda, R.; Wozniak, K.;
́
S.; Fedorov, A.; Coper
́
et, C.; Togni, A. N-Trifluoromethyl NHC
Trzaskowski, B.; Kajetanowicz, A.; Grela, K. Specialized Ruthenium
Olefin Metathesis Catalysts Bearing Bulky Unsymmetrical NHC
Ligands: Computations, Synthesis, and Application. ACS Catal. 2019,
Ligands Provide Selective Ruthenium Metathesis Catalysts. Organo-
metallics 2016, 35, 887−893. (c) Miyazaki, H.; Herbert, M. B.; Liu, P.;
Dong, X.; Xu, X.; Keitz, B. K.; Ung, T.; Mkrtumyan, G.; Houk, K. N.;
Grubbs, R. H. Z - Selective Ethenolysis with a Ruthenium Metathesis
Catalyst: Experiment and Theory. J. Am. Chem. Soc. 2013, 135, 5848−
9, 587−598. (b) Wyrebek, P.; Maecki, P.; Sytniczuk, A.; Kosnik, W.;
́
Gawin, A.; Kostrzewa, J.; Kajetanowicz, A.; Grela, K. Looking for the
Noncyclic(Amino)(Alkyl)Carbene Ruthenium Catalyst for Ethenol-
ysis of Ethyl Oleate: Selectivity Is on Target. ACS Omega 2018, 3,
18481−18488. (c) Paradiso, V.; Bertolasi, V.; Costabile, C.; Caruso,
5858. (d) Thomas, R. M.; Keitz, B. K.; Champagne, T. M.; Grubbs, R.
H. Highly Selective Ruthenium Metathesis Catalysts for Ethenolysis.
J. Am. Chem. Soc. 2011, 133, 7490−7496. (e) Schrodi, Y.; Ung, T.;
Vargas, A.; Mkrtumyan, G.; Lee, C. W.; Champagne, T. M.; Pederson,
R. L.; Hong, S. H. Ruthenium Olefin Metathesis Catalysts for the
Ethenolysis of Renewable Feedstocks. Clean: Soil, Air, Water 2008, 36,
T.; Da b̧ rowski, M.; Grela, K.; Grisi, F. Expanding the Family of
Hoveyda−Grubbs Catalysts Containing Unsymmetrical NHC
Ligands. Organometallics 2017, 36, 3692−3708. (d) Małecki, P.;
Gajda, K.; Ablialimov, O.; Malinska, M.; Gajda, R.; Wozniak, K.;
́
́
6
(
69−673.
Kajetanowicz, A.; Grela, K. Hoveyda−Grubbs-Type Precatalysts with
Unsymmetrical N-Heterocyclic Carbenes as Effective Catalysts in
Olefin Metathesis. Organometallics 2017, 36, 2153−2166. (e) Hamad,
F. B.; Sun, T.; Xiao, S.; Verpoort, F. Olefin metathesis ruthenium
catalysts bearing unsymmetrical heterocylic carbenes. Coord. Chem.
Rev. 2013, 257, 2274−2292. (f) Vougioukalakis, G. C.; Grubbs, R. H.
Ruthenium Olefin Metathesis Catalysts Bearing an N-Fluorophenyl-
N-Mesityl-Substituted Unsymmetrical N-Heterocyclic Carbene. Orga-
nometallics 2007, 26, 2469−2472. (g) Ledoux, N.; Allaert, B.; Pattyn,
S.; Mierde, H. V.; Vercaemst, C.; Verpoort, F. N,N’-Dialkyl- and N-
Alkyl-N-mesityl-Substituted N-Heterocyclic Carbenes as Ligands in
Grubbs Catalysts. Chem. - Eur. J. 2006, 12, 4654−4661. (h) Vehlow,
K.; Maechling, S.; Blechert, S. Ruthenium Metathesis Catalysts with
10) Molybdenum and tungsten examples: (a) Marinescu, S. C.;
Levine, D. S.; Zhao, Y.; Schrock, R. R.; Hoveyda, A. H. Isolation of
Pure Disubstituted E Olefins through Mo-Catalyzed Z-Selective
Ethenolysis of Stereoisomeric Mixtures. J. Am. Chem. Soc. 2011, 133,
11512−11514. (b) Marinescu, S. C.; Schrock, R. R.; Muller, P.;
̈
Hoveyda, A. H. Ethenolysis Reactions Catalyzed by Imido Alkylidene
Monoaryloxide Monopyrrolide (MAP) Complexes of Molybdenum. J.
Am. Chem. Soc. 2009, 131, 10840−10841.
(11) Scholl, M.; Ding, S.; Lee, C. W.; Grubbs, R. H. Synthesis and
Activity of a New Generation of Ruthenium-Based Olefin Metathesis
Catalysts Coordinated with 1,3-Dimesityl-4,5-dihydroimidazol-2-
ylidene Ligands. Org. Lett. 1999, 1, 953−956.
J
Organometallics XXXX, XXX, XXX−XXX

 Byun, Seunghwan
Byun, Seunghwan
 Seo, Huiyeong
Seo, Huiyeong
 Choi, Jun-Ho
Choi, Jun-Ho
 Ryu, Ji Yeon
Ryu, Ji Yeon
 Lee, Junseong
Lee, Junseong
 Chung, Won-Jin
Chung, Won-Jin
 Hong, Sukwon
Hong, Sukwon