1
34 Michael A. Daniele et al.
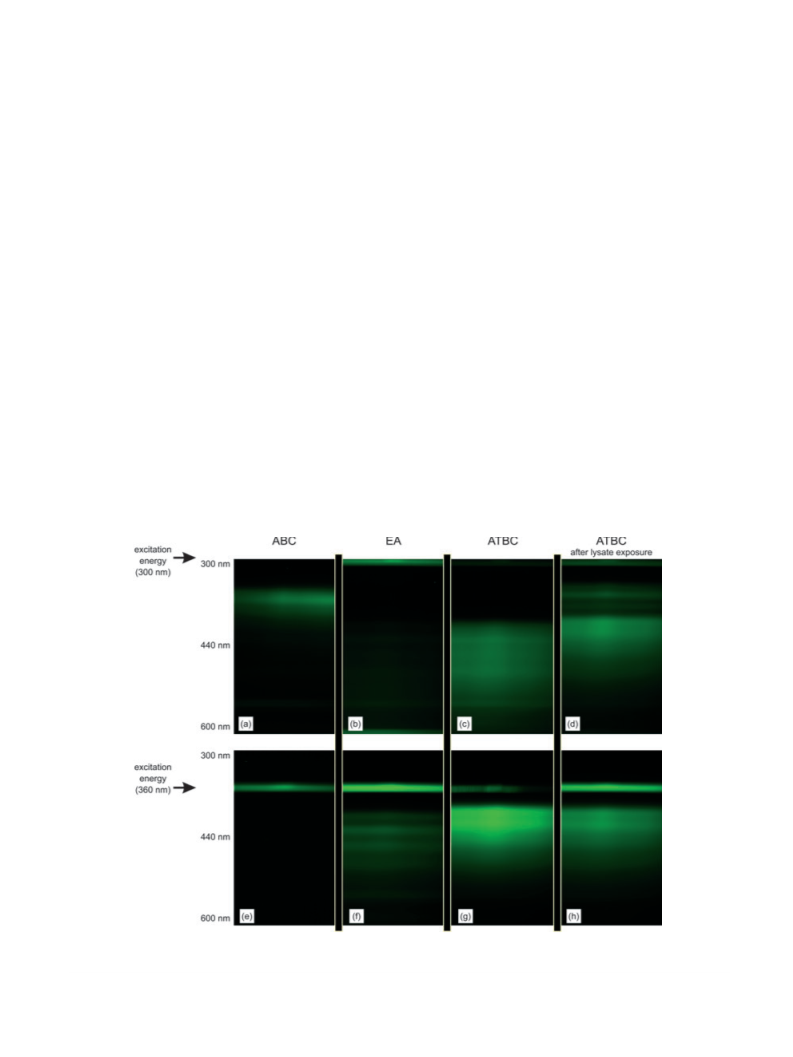
the carbazole moiety, as well as residues that incorporate an
anthracene fluorophore. The anthracene signature, as indicated
by the two peaks at 381 and 403 nm, is significantly less than the
contribution from carbazole-containing residues, which occurs
at wavelengths under 360 nm. This indicates that the emission
at 410 nm can be efficiently achieved by exciting the carbazole
moiety and transferring energy to the anthracene moiety; it also
suggests that the supernatant has a mixture of residues that
incorporate ABC and EA, as well as ATBC. Increasing the
emission wavelength to 450 nm is sufficient to remove contri-
butions that are directly attributed to the carbazole moiety,
though this wavelength is in the middle of the emission
spectrum for both EA and ATBC. Nonetheless, the PLE
spectrum at this emission wavelength indicates a contribution
from the carbazole fluorophore, suggesting that either the
supernatant contains (1) ATBC and ⁄ or (2) carbazole and
anthracene flourophores that are not chemically linked, but
¨
2. Forster, T. (1946) Energiewanderung and Fluoreszenz. Naturwis-
senschaften 6, 166–175.
3
. Prasuhn, D. E., A. Feltz, J. B. Blanco-Canosa, K. Susumu, M. H.
Stewart, B. C. Mei, A. V. Yakovlev, C. Loukov, J. M. Mallet,
M. Oheim, P. E. Dawson and I. L. Medintz (2010) Quantum dot
peptide biosensors for monitoring caspase-3 proteolysis and cal-
cium ions. ACS Nano. 4, 5487–5497.
4
. Amantonico, A., P. L. Urban and R. Zenobi (2010) Analytical
techniques for single-cell metabolomics: state of the art and trends.
Anal. Bioanal. Chem. 398, 2493–2504.
5. Patterson, A. D., F. J. Gonzalez and J. R. Idle (2010) Xenobiotic
metabolism: a view through the metabolometer. Chem. Res.
Toxicol. 23, 851–860.
6
. Ouchiyama, N., Y. Zhang, T. Omori and T. Kodama (1993)
Biodegradation of carbazole by Pseudomonas spp. CA06 and
CA10. Biosci. Biotechnol. Biochem. 57, 455–460.
7. Ahuja, S. K., G. M. Ferreira and A. R. Moreira (2004) Utilization
of enzymes for environmental applications. Crit. Rev. Biotechnol.
2
4, 125–154.
8
. Seo, J. S., Y. S. Keum and Q. X. Li (2009) Bacterial degradation
of aromatic compounds. Int. J. Environ. Res. Public Health, 6,
278–309.
¨
spatially within the Forster radius for energy transfer. Extend-
ing the emission wavelength to 495 nm verifies that the
supernatant must contain ATBC as this fluorophore would be
the dominating emitter at this wavelength.
9. Wang, X., Z. H. Gai, B. Yu, J. H. Feng, C. Y. Xu, Y. Yuan, Z. X.
Lin and P. Xu (2007) Degradation of carbazole by microbial cells
immobilized in magnetic gellan-gum gel beads. Appl. Environ.
Microbiol. 73, 6421–6428.
1
0. Larentis, A. L., H. C. C. Sampaio, C. C. Carneiro, O. B. Martins
and T. L. M. Alves (2011) Evaluation of growth of carbazole
biodegradation and anthranilic acid production. Braz. J. Chem.
Eng. 28, 37–44.
CONCLUSION
In summary, a simple diagnostic tool was presented that
employed a coupled donor ⁄ acceptor pair that was formed
through a click transformation. The FRET pair exhibited a
significant variation in PL response with exposure to the
P. resinovorans CA10 lysate, an organism that can degrade
variants of both the donor and acceptor fluorophores. This
general approach can be tailored for a range of metabolic
processes and be employed as a method for single cell meta-
bolomic studies.
1
1. Habe, H., K. Ide, M. Yotsumoto, H. Tsuji, H. Hirano, J. Widada,
T. Yoshida, H. Nojiri and T. Omori (2001) Preliminary examin-
ations for applying a carbazole-degrader, Pseudomonas spp. strain
CA10, to dioxin-contaminated soil remediation. Appl. Microbiol.
Biotechnol. 56, 788–795.
1
2. Waldau, D., K. Methling, A. Mikolasch and F. Schauer (2009)
Characterization of new oxidation products of 9H-carbazole and
structure related compounds by biphenyl-utilizing bacteria. Appl.
Microbiol. Biotechnol. 81, 1023–1031.
13. Nojiri, H., H. Habe and T. Omori (2001) Bacterial degradation of
aromatic compounds via angular dioxygenation. J. Gen. Appl.
Microbiol. 47, 279–305.
Acknowledgements—The authors thank DARPA (grant number:
N66001-04-1-8933), the State of South Carolina, and the Gregg-
Graniteville Foundation for financial support.
1
1
1
4. Nojiri, H., J. W. Nam, M. Kosaka, K. I. Morii, T. Takemura, K.
Furihata, H. Yamane and T. Omori (1999) Diverse oxygenations
catalyzed by carbazole 1,9a-dioxygenase from Pseudomonas sp.
strain CA10. J. Bacteriol. 181, 3105–3113.
5. Guan, J., D. E. Kyle, L. Gerena, Q. A. Zhang, W. K. Milhous and
A. J. Lin (2002) Design, synthesis, and evaluation of new
chemosensitizers in multi-drug-resistant Plasmodium falciparum.
J. Med. Chem. 45, 2741–2748.
SUPPORTING INFORMATION
Additional Supporting Information may be found in the online
version of this article:
Figure S1. Photoluminescence spectrum for the serial
dilution of ATBC normalized to the mass of chromophore at
6. Xiao, Q., R. T. Ranasinghe, A. M. P. Tang and T. Brown (2007)
Naphthalenyl- and anthracenyl-ethynyl dT analogues as base
discriminating fluorescent nucleosides and intramolecular energy
transfer donors in oligonucleotide probes. Tetrahedron 63, 3483–
3490.
kex = 295 nm.
1
7. Jin, T., S. Kamijo and Y. Yamamoto (2004) Copper-
catalyzed synthesis of N-unsubstituted 1,2,3-triazoles from
non-activated terminal alkynes. Eur. J. Org. Chem. 18, 3789–
Figure S2. Photoluminescence spectrum for the serial
dilution of ATBC normalized to the mass of chromophore at
kex = 360 nm.
3
791.
Figure S3. Mass spectroscopy comparison for donor–
18. Bradford, M. M. (1976) Rapid and sensitive method for quanti-
tation of microgram quantities of protein utilizing principle of
protein-dye binding. Anal. Biochem. 72, 248–254.
acceptor system (ATBC), paired moieties (ABC, EA) and
biotransform residues of ATBC (CA10 biotransformation).
Please note: Wiley-Blackwell is not responsible for the
content or functionality of any supporting information sup-
plied by the authors. Any queries (other than missing material)
should be directed to the corresponding author for the article.
1
9. Kolb, H. C., M. G. Finn and K. B. Sharpless (2001) Diverse
chemical function from a few good reactions. Angew. Chem. Int.
Ed. Engl. 40, 2004–2021.
20. Johnson, G. E. (1974) Spectroscopic study of carbazole by
photoselection. J. Phys. Chem. 78, 1512–1521.
2
1. Ray, K., D. Bhattacharjee and T. N. Misra (1997) Photophysical
characteristics of 9-cyanoanthracene molecules organized in
Langmuir-Blodgett films. J. Chem. Soc. Faraday. Trans. 93, 4041–
REFERENCES
4
045.
1
. Giepmans, B. N. G., S. R. Adams, M. H. Ellisman and R. Y.
Tsien (2006) The fluorescent toolbox for assessing protein location
and function. Science 312, 217–224.
22. Ray, K. and T. N. Mishra (1999) Energy transfer between car-
bazole and anthracene moieties organized in Langmuir-Blodgett
films. J. Phys. Chem. Solids 60, 401–405.

 Daniele, Michael A.
Daniele, Michael A.
 Bandera, Yuriy P.
Bandera, Yuriy P.
 Foulger, Stephen H.
Foulger, Stephen H.