
Carbohydrate Research p. 480 - 487 (2011)
Update date:2022-08-17
Topics:
 Shi, Xuejun
Shi, Xuejun
 Wu, Yulong
Wu, Yulong
 Li, Panpan
Li, Panpan
 Yi, Huaifeng
Yi, Huaifeng
 Yang, Mingde
Yang, Mingde
 Wang, Gehua
Wang, Gehua
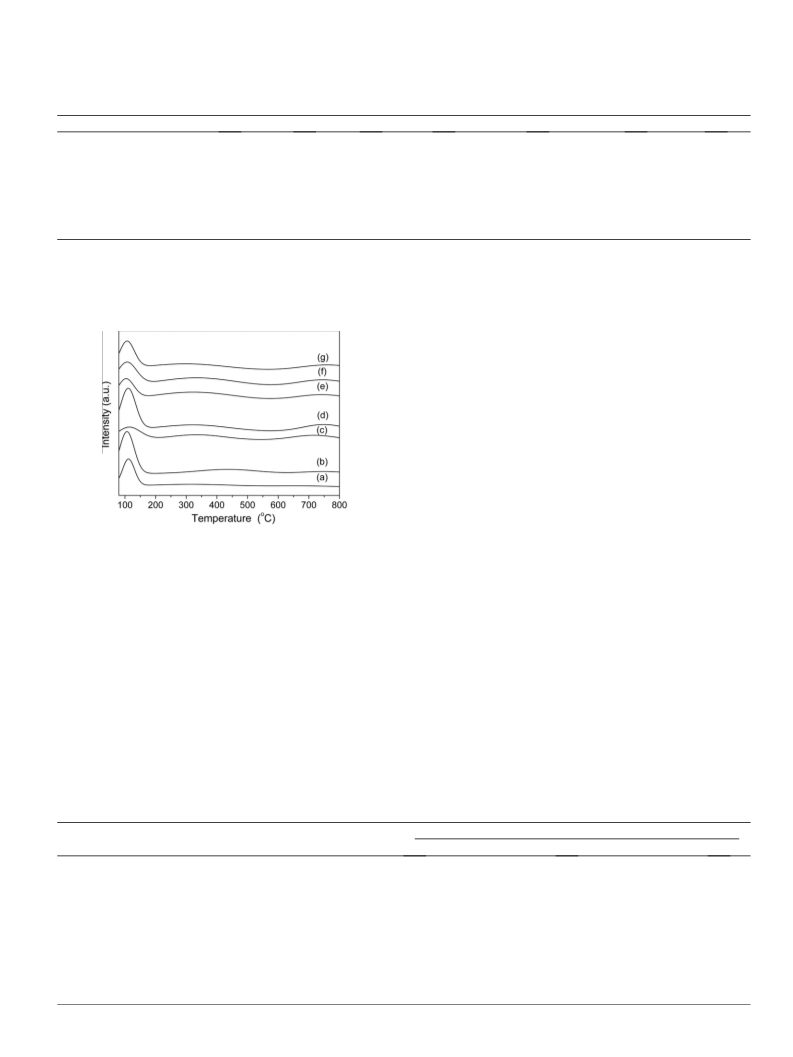
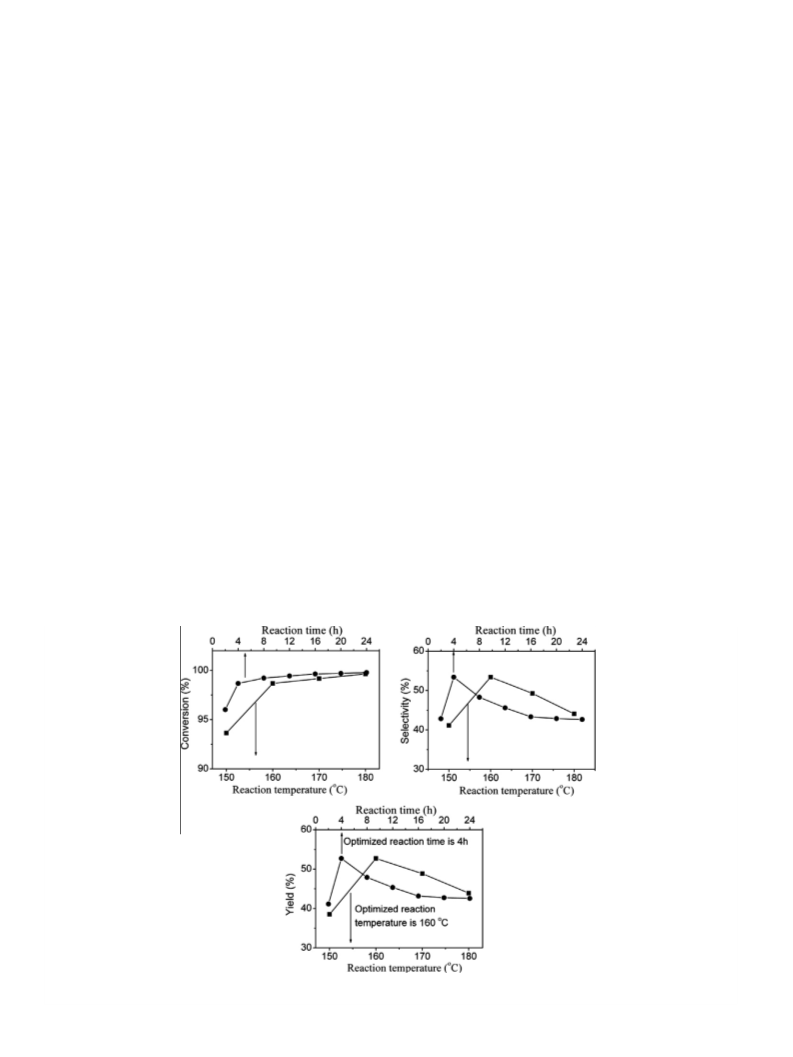
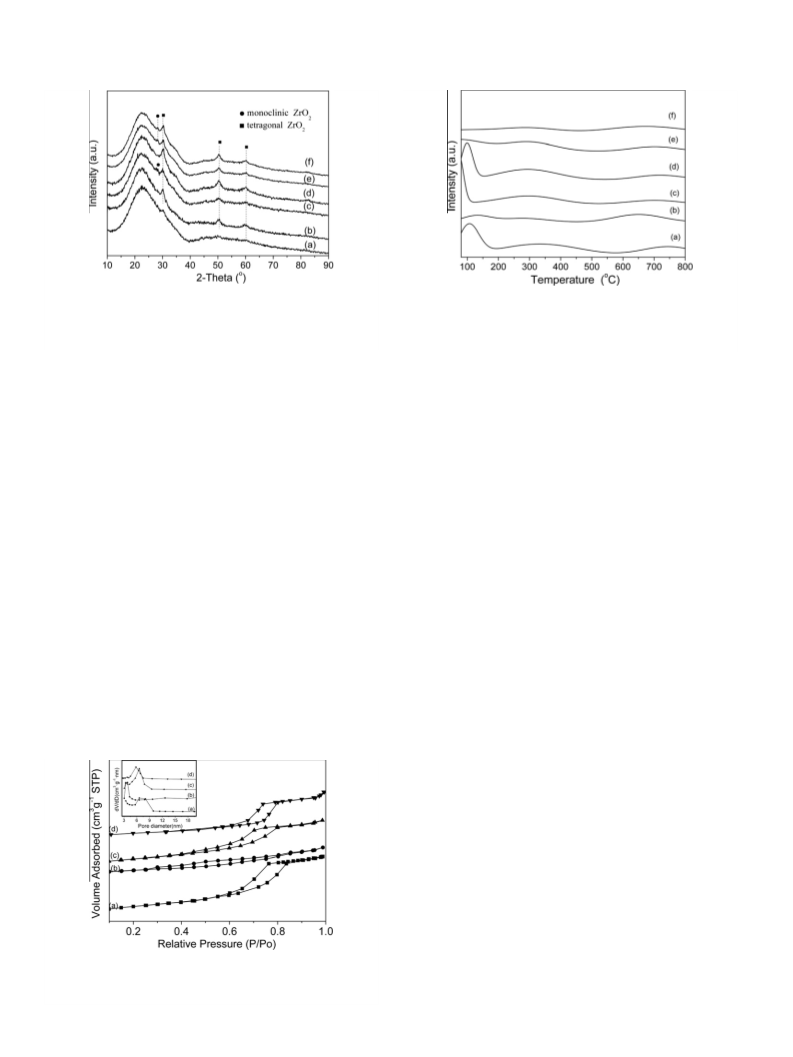
Al-promoted SO42--/ZrO2/SBA-15 catalysts were prepared and characterized by XRD, BET, ICP and NH3-TPD techniques. The influence of introducing aluminum on the structure and surface properties of the catalyst and the catalytic activity for dehydration of xylose to furfural has been investigated. The introduction of the Al stabilizes the tetragonal phase of the ZrO2 and thus increases the number and intensity of acid sites. Based on the characterization of the deactivated catalyst, the accumulation of byproducts is the main reason for the deactivation of the catalyst. Regeneration with H2O2 can completely recover the catalytic activity of the deactivated catalyst.
View More



Contact:+ 86 512 52491118
Address:1 Fuyu Road, Haiyu TownChangshu, Jiangsu, China
NINGBO PANGS CHEM INT’L CO.,LTD.
Contact:+86-574-27666845
Address:FLOOR 21,BUILDING NO.11,XIN TIAN DI,NO.689 SHI JI ROAD,NINGBO CHINA
Shanghai KFSL Pharmaceutical Technology Co.,Ltd.
Contact:+86-21-39971718
Address:859 jiadingchengliu shanghai
Goldwills Pharmaceuticals Co., Ltd.
Contact:0916-2237889 13991621155
Address:North Suburb of Hanzhong city, Shaanxi Province
Jingzhou TianHe Sci&Tech Chemical Co., Ltd.
Contact:86-716-8331612
Address:Jiangjin Road, #18, High-grade technology industries development district, Jingzhou city, Hubei province
Doi:10.1016/j.bioorg.2020.104356
(2020)Doi:10.1021/jp063362n
(2006)Doi:10.1016/j.tetlet.2006.05.166
(2006)Doi:10.1039/b414996c
(2004)Doi:10.1016/j.ica.2017.06.052
(2017)Doi:10.1080/00958972.2014.928289
(2014)