G.-W. Rao, W.-X. Hu / Bioorg. Med. Chem. Lett. 16 (2006) 3702–3705
3705
(f) Soloducho, J.; Doskocz, J.; Cabaj, J.; Roszak, S.
Tetrahedron 2003, 59, 4761.
central six-membered ring of 1d, the tetrazine ring, has
an obvious chair conformation and therefore is not
homoaromatic.
4. Synthesis of 1f or 1 g: azine (10 mmol) was dissolved in
ethanol (20 ml) with stirring. Hydrated hydrazine
(12 mmol, 80%) was added to the mixture in reflux
conditions. The mixture was refluented for 45 min, cooled,
filtered, and washed with ethanol (3· 10 ml). Solvent was
removed in vacuo and the residue was recrystallized from
ethanol or chloroform to give the product as a yellow
solid. Synthesis of 1d: (4-Chloro-phenyl)-acetonitrile
(50 mmol) and sulfur (1.0 g) were dissolved in anhydrous
ethanol (15 ml) with stirring in nitrogen. Hydrated
hydrazine (10 ml, 80%) was added to the mixture in an
ice bath. The mixture was stirred at room temperature for
2 h and then reflux conditions for 1–2 h, cooled, filtered,
and washed with anhydrous ethanol (3 · 10 ml). Solvent
was removed in vacuo and the residue was recrystallized
from ethanol or chloroform to give the product (1d) as a
colorless solid. IR (KBr, cmÀ1): 3446, 3267, 3182, 1678,
1491, 1442, 1417, 1235, 1209, 1083, 1014, 978, 932, 834,
741, 659, 586, 499, 429. 1H NMR (400 MHz, CDCl3)d:
3.48 (s, 4H, CH2), 6.13 (br, 2H, NH), 7.17–7.32 (m, 8H,
Ph). MS m/z (%): 333 (M+, 11), 334 (27), 332 (40), 127
(31), 125 (100), 116 (16), 90 (13), 89 (40), 63 (15). Anal.
Calcd for C16H14N4Cl2: C, 57.67; H, 4.23; N, 16.81.
Found: C, 57.82; H, 4.17; N, 16.79.Compounds of 1a–e
were prepared as described above for 1d. Synthesis of 3d:
compound 1d (10 mmol) was suspended in a 10% solution
of NaNO2 (25 ml) with stirring. Ether (15 ml) was added
to the mixture, a 10% solution of CH3COOH (15 ml) was
dropped to the mixture. The mixture was stirred at room
temperature for 5 h, filtered and the residue was recrys-
tallized from ethanol to give the product (3d) as a red
solid. IR (KBr, cmÀ1): 3050, 1607, 1553, 1491, 1378, 1088,
1014, 852, 785. 1H NMR (400 MHz, CDCl3)d: 4.57 (s,
4 H, CH2), 7.30–7.35 (m, 8H, Ph). MS m/z (%): 331 (M+,
0.73), 151 (34), 116 (100), 89 (27), 50 (8). Anal. Calcd for
C16H12N4Cl2: C, 58.02; H, 3.65; N, 16.92. Found: C,
58.34; H, 3.61; N, 16.73. Compounds of 3a–e were
prepared as described above for 3d.
The antitumor activities in vitro for these compounds
were evaluated by SRB method for A-549 and BEL-
7402 cells, and MTT method for P-388 and HL-60 cells.
The results are summarized in Tables 2–5.
Usually, when the concentration of the compound solu-
tion is 10À6 mol/L, the inhibition ratio of the solution to
cancer cell growth is more than 50%, or when the con-
centration of the compound solution is 10À5 mol/L,
the inhibition ratio of the solution to cancer cell growth
is more than 85%, the compound is considered as
strongly effective. According to this standard, it can be
found from Tables 2–5 that there is one compound
(3g) that has a very strong effect against to P-388 cells.
The inhibition ratio of the solution to cancer cell growth
is more than 80% in 10À8 mol/L. And there is one com-
pound (3e) that has a strong affect against HL-60 cells.
The different group of 3,6-positions has prodigious
diversity of antitumor activity. Changing the group of
3,6-positions is possible to improve the antitumor activ-
ity. So 1,4-dihydro-1,2,4,5-tetrazine is a kind of com-
pound which may have potential antitumor activities.
It is a good lead compound that warrants further
investigation.
Acknowledgments
The authors thank the National Natural Science Foun-
dation of China (20272053), for financial support and
National Center for Drug Screening, Shanghai, China,
for evaluation of antitumor activities.
5. Cohen, V. I. J. Heterocycl. Chem. 1978, 1, 1113.
6. (a) Brooker, P. J.; Parsons, J. H.; Reid, J.; West, P. J.
Pestic. Sci. 1987, 18, 179; (b) Chae, Y. B.; Chang, K. S.;
Kim, S. S. Taehan Hwahakhoe Chi 1967, 11, 85.
7. Wiley, P. F.. In Neunhoeffer, H., Wiley, P. F., Eds.;
Heterocyclic Compounds; John Wiley and Sons: New
York, 1983; vol. 33, p 1075.
8. Sauer, J.; Bauerlein, P.; Ebenbeck, W.; Gousetis, C.;
Sichert, H.; Troll, T.; Utz, F.; Wallfahrer, U. Eur. J. Org.
Chem. 2001, 14, 2629.
Supplementary data
Supplementary data associated with this article can be
9. Lang, S. A., Jr.; Johnson, B. D.; Cohen, E. J. Heterocycl.
Chem. 1975, 1, 1143.
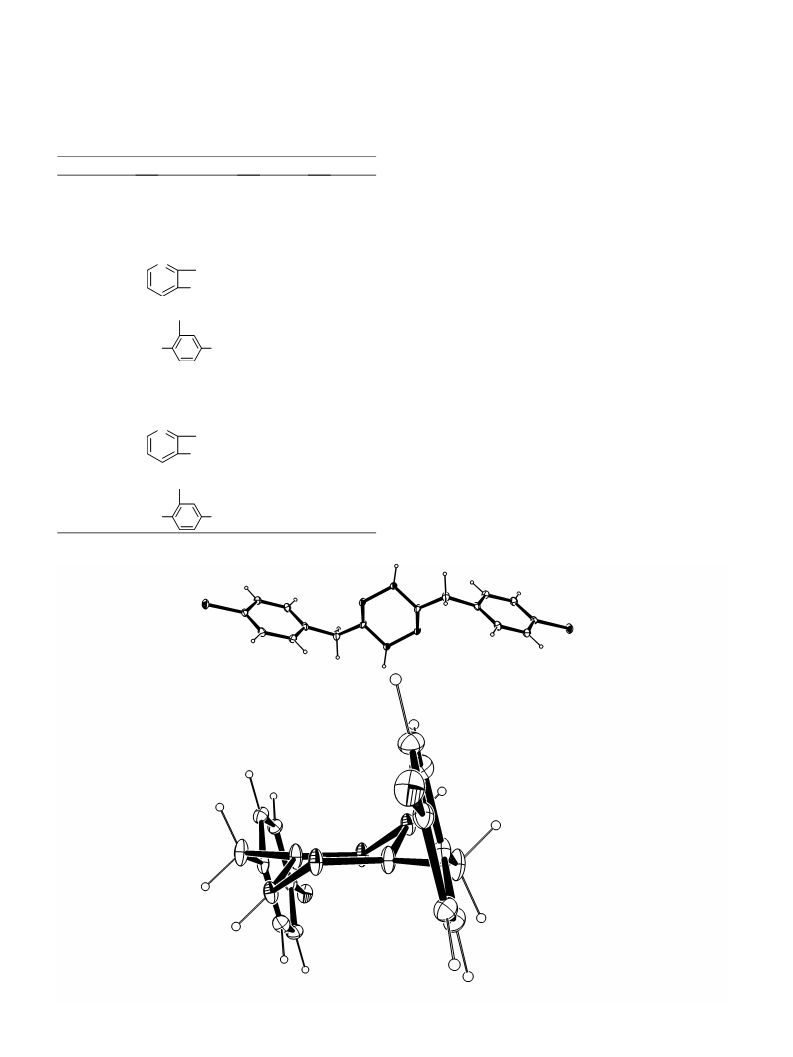
10. Crystal data of 1d. C8H7ClN2, M = 166.61, Monoclinic,
References and notes
˚
a = 5.833 (1), b = 5.131 (3), c = 26.250 (6) A, b = 93.05
1. (a) Neunhoeffer, H. In Katritzky, A. R., Ed.; Compre-
hensive Heterocyclic Chemistry, I; Pergamon: Frankfurt,
1984; vol. 3, p 531; (b) Sauer, J. In Boulton, A. J., Ed.;
Comprehensive Heterocyclic Chemistry, II; Elsevier:
Oxford, England, 1996; vol. 6, p 901.
2. (a) Eremeev, A. V.; Tikhomirov, D. A.; Tyusheva, V. A.;
Liepins, F. Khim. Geterotsikl. Soedin. 1978, 6, 753; (b)
Eremeev, A.V.; Tikhomirov, D.A.; Zidermane, A.
U.S.S.R. 686,336, 1980.
3
˚
(2)ꢁ, U = 784.5 (5) A , T = 293 (2) K, space group P 21/n,
Z = 4, Dc = 1.411 g/cm3, l(Mo-Ka) = 0.415 mmÀ1, 3798
reflections measured, 3599 unique (Rint = 0.0413) which
were used in all calculations. Fine R1 = 0.0437, wR
(F2) = 0.1421 (all data). Full crystallographic details of
1d have been deposited at the Cambridge Crystallographic
Data Centre and allocated the deposition number CCDC
255695.
11. Frisch, M. J.; Trucks, G. W.; Schlegel, H. B. et al. Gaussian
98, Revision A. 7, Pittsburgh PA: Gaussian Inc. 1998.
12. Jennison, C. P. R.; Mackay, D.; Watson, K. N.; Taylor,
N. J. J. Org. Chem. 1986, 51, 3034.
13. (a) Neugebauer, F. A.; Krieger, C.; Fischer, H.; Siegel, R.
Chem. Ber. 1983, 116, 2261; (b) Bemis, G. W.; Whittle, R.
R.; Mayo, S. L. Acta Crystallogr. 1984, C40, 2076.
3. (a) Dallacker, F. Monatsh. Chem. 1960, 91, 294; (b)
Abdel-Rahman, M. O.; Kira, M. A.; Tolba, M. N.
Tetrahedron Lett. 1968, 35, 3871; (c) Konishi, Osako,
Koto, Jap. Pat. 74, 04, 532, 1974; (d) Chai, L. L.; Sang,
H. P.; Tae, Y. K.; Eui, S. Y.; Byung, H. H. Bull.
Korean Chem. Soc. 1995, 16, 374; (e) Bentiss, F.;
´
Lagrenee, M.; Barbry, D. Tetrahedron 2000, 41, 1539;

 Rao, Guo-Wu
Rao, Guo-Wu
 Hu, Wei-Xiao
Hu, Wei-Xiao