Page 1 of 6
RSC Advances
Journal Name
DOI: 10.1039/C3RA43799J
Cite this: DOI: 10.1039/c0xx00000x
www.rsc.org/xxxxxx
ARTICLE TYPE
Iodide-catalyzed Amide Synthesis from Alcohols and Amines
a
a
a
a
a
a
Gao Wang, Qing-Ying Yu, Jian Wang, Shan Wang, Shan-Yong Chen* and Xiao-Qi Yu*
Received (in XXX, XXX) Xth XXXXXXXXX 20XX, Accepted Xth XXXXXXXXX 20XX
DOI: 10.1039/b000000x
5
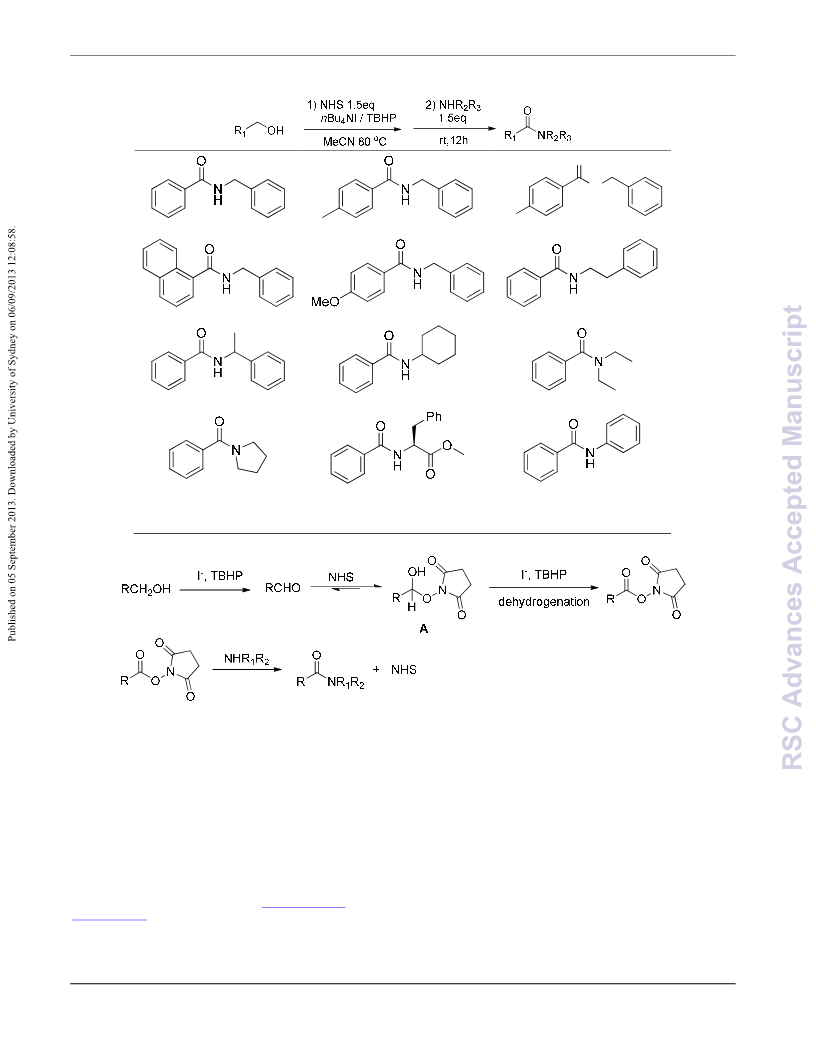
An efficient method to prepare amides by a cascade strategy was developed. Using nBu NI or NaI as the
4
catalyst and tertꢀbutyl hydroperoxide as the oxidant, various alcohols reacted with Nꢀhydroxysuccinimide
or Nꢀhydroxyphthalimide affording corresponding active esters in moderate to good yield. The resulted
active esters were converted into amides smoothly in one pot.
first metalꢀcatalyzed amidation reaction of aldehyde using Nꢀ
hydroxyimide as a dual promoter of aldehyde oxidation and
amines displacement. Barbas III groups reported the first
Introduction
8
1
1
2
2
0
5
0
5
Amide bond is one of the most abundant units in a wide range of
natural products, polymers, agrochemicals, and pharmaceuticals
with biologically relevant properties. The synthesis of amides
has therefore attracted considerable interest and a number of
methods have been devised. They are routinely prepared from the
acylation of amines with activated carboxylic acids, especially
with Nꢀhydroxyimide esters. Oxidative amidation of alcohols or
aldehydes are economically attractive alternatives to traditional
synthesis. Research during the past decade resulted in significant
organocatalytic amidation reaction of aldehyde using the same
9
4
0
strategy. We envisioned that an iodide reagent also could
1
catalyze the oxidation of alcohols into Nꢀhydroxyimide esters,
which facilitate the displacement of an amine. This tandem
strategy will alleviate structural dependence on amines.
2
Results and discussion
45
Our initial studies focused on the model reaction of
phenylmethanol 1a with Nꢀhydroxysuccinimide (NHS) 2a in
acetonitrile. Without a catalyst, no desired product was observed.
3
,4
progress in the field of amidation of aldehydes. Considering the
stability and the availability of alcohols, chemists have been
focusing on the direct conversion of alcohols and amines into
amides, which is more atomꢀeconomical and environmentally
benign. In spite of great progresses, the use of transitionꢀmetal
a
Table 1. Optimization of the reaction conditions
50
5
catalysts or the need for more than stoichiometric amounts of
6
hypervalent iodine (III) reagents limits the practical application
of this strategy.
Entry
Catalyst
Oxidant
Yield
1
2
3
4
5
6
7
8
ꢀ
aqueous TBHP
aqueous TBHP
aqueous TBHP
aqueous TBHP
aqueous TBHP
anhydrous TBHP
DTBP
N.D.
71%
82%
N.D.
N.D.
85%
N.D.
N.D.
NaI
nBu NI
4
I2
PhI(OAc)2
nBu NI
4
nBu NI
4
nBu NI
H O
2
4
2
b
9
nBu NI
anhydrous TBHP
anhydrous TBHP
80%
76%
4
c
1
0
nBu NI
4
Scheme 1. Methods to amidation of alcohols
a
1
a (0.5 mmol), NHS (0.75 mmol), catalyst (10 mol %), oxidant (4.0
3
0
b
equiv) in acetonitrile (2 mL) at 80 °C for 18 hours. Ethyl acetate as the
Wang et al developed an amide formation reaction of alcohol
with N,Nꢀdisubstituted formamides leading to N,Nꢀdisubstituted
amides without the use of a hypervalent iodine (III) reagent or a
c
solvent. Using 0.5 mmol of NHS. H O : 50% hydrogen peroxide in
water; DTBP: diꢀtertꢀbutylꢀperoxide.
2
2
7
metal catalyst. However, nitrogen source is limited to preꢀ
With NaI as the catalyst and aqueous tertꢀbutyl hydroperoxide
(
3
5
formed formamides. Quite recently, Yamamoto et al reported the
TBHP 70 wt.% in water) as the oxidant, the desired active ester
This journal is © The Royal Society of Chemistry [year]
[journal], [year], [vol], 00–00 | 1

 Wang, Gao
Wang, Gao
 Yu, Qing-Ying
Yu, Qing-Ying
 Wang, Jian
Wang, Jian
 Wang, Shan
Wang, Shan
 Chen, Shan-Yong
Chen, Shan-Yong
 Yu, Xiao-Qi
Yu, Xiao-Qi