VOL. 39, 1995
L-743,726 (DMP-266): A NOVEL HIV-1 RT INHIBITOR
2605
This compound was found to be a potent inhibitor of puri-
fied wild-type HIV-1 RT in vitro and of HIV-1 replication in
cell culture. In the latter respect, its activity was at least 10-fold
greater than that of L-697,661. In addition, L-743,726 exhibited
noted antiviral activity against an extensive panel of HIV-1
mutants constructed so that they expressed single and multiple
RT substitutions which mediate resistance to many of the de-
scribed NNRTIs. Analysis of similar data obtained with puri-
fied mutant RT enzymes (Table 1) suggested that the primary
interaction between the inhibitor and the RT was probably
mediated by the RT amino acid residues at positions 100 and
4. Carroll, S. S., M. Stahlhut, J. Geib, and D. B. Olsen. 1994. Inhibition of
HIV-1 reverse transcriptase by a quinazolinone and comparison with inhi-
bition by pyridinones. J. Biol. Chem. 269:32351–32357.
5
6
7
. Dueweke, T. J., F. J. Kezdy, G. A. Waszak, M. R. Deibel, and W. G. Tarpley.
1992. The binding of a novel bisheteroarypiperazine mediates inhibition of
human immunodeficiency virus type 1 reverse transcriptase. J. Biol. Chem.
267:27–30.
. Frank, K. B., G. J. Noll, E. V. Connell, and I. S. Sim. 1991. Kinetic interac-
tion of the human immunodeficiency virus type 1 reverse transcriptase with
the antiviral tetrahydroimidazo [4,5,1,jk]-[1,4]-benzodiazepine-2-(1H)-
thione compound, R82150. J. Biol. Chem. 266:14232–14236.
. Goldman, M. E., J. H. Nunberg, J. A. O’Brien, J. C. Quintero, W. A. Schleif,
K. F. Freund, S. L. Gaul, W. S. Saari, J. S. Wai, J. M. Hoffman, P. S.
Anderson, D. J. Hupe, E. A. Emini, and A. M. Stern. 1991. Pyridinone
derivatives: specific human immunodeficiency virus type 1 reverse tran-
scriptase inhibitors with antiviral activity. Proc. Natl. Acad. Sci. USA 88:
1
03. Both residues contribute to the defined NNRTI RT bind-
ing site (15, 18). Alterations at each of these sites had striking
adverse effects on L-743,726’s activity. Cell culture selection of
mutant virus with greatly reduced susceptibility to the inhibitor
resulted in a variant that expressed a combination of substitu-
tions at both residues (Table 2). In contrast to the relative ease
with which L-697,661-resistant virus was previously derived in
culture (10), derivation of L-743,726-resistant virus required
an extended number of passages and an extended selection
period, suggesting that the resistant mutant was not signifi-
cantly represented in the initial wild-type virus population.
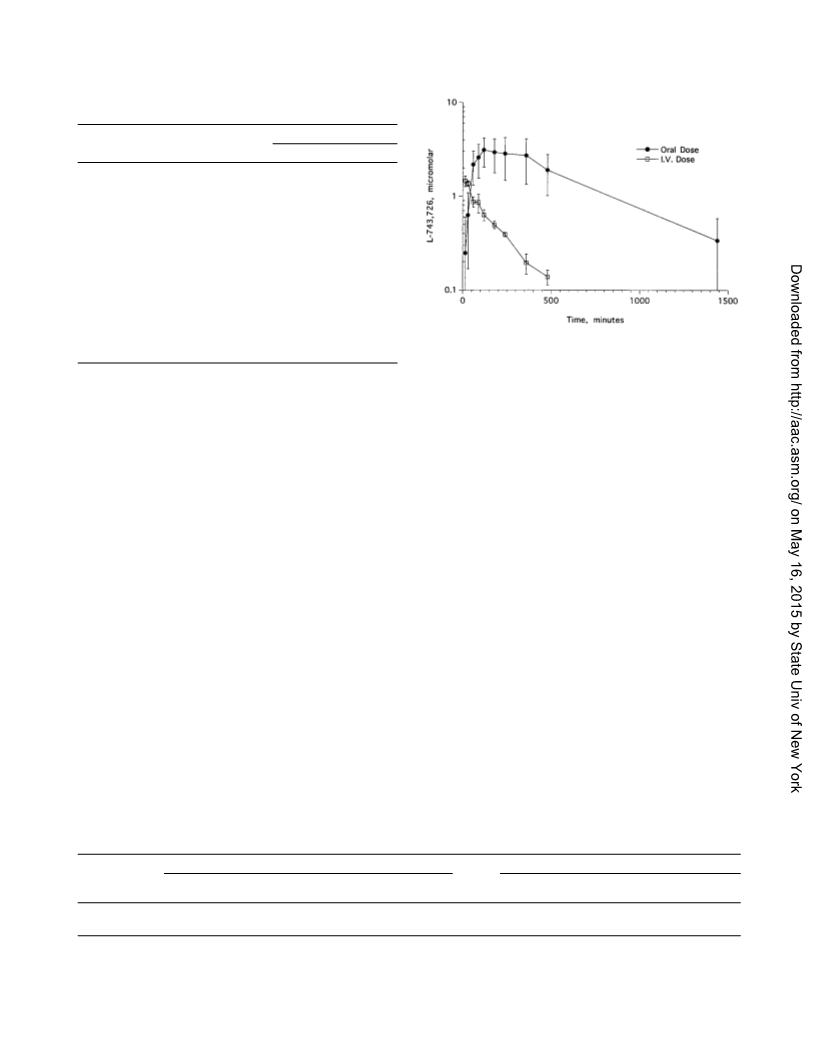
The compound’s pharmacokinetic characteristics in several
test animal species were also encouraging. Oral bioavailability
was good, as was the maintenance of reasonably high systemic
levels following the oral administration of an aqueous suspen-
sion. In a chimpanzee, a 10-mg/kg oral dose resulted in con-
centrations greater than 2.5 M at 24 h. This concentration of
compound would, in theory, be sufficient to suppress replica-
tion of all of the NNRTI-resistant mutants that express a single
RT amino acid substitution and some that express multiple
substitutions.
Of course, it is likely that monotherapy with L-743,726
would rapidly select for a highly resistant variant with amino
acid alterations at both residues 100 and 103. However, the
usefulness of the compound in combination therapy will as
likely be a reflection of its high degree of potency against
wild-type HIV-1 and its excellent bioavailability. In this regard,
L-743,726 represents a reasonable candidate for inclusion in
such a therapeutic regimen to determine whether the NNRTIs
will contribute significantly to the treatment of HIV-1-infected
persons.
6863–6867.
8
. Kohlsteadt, L. A., J. Wang, J. M. Friedman, P. A. Rice, and T. A. Steitz. 1992.
Crystal structure at 3.5Å resolution of HIV-1 reverse transcriptase com-
plexed with an inhibitor. Science 256:1783–1790.
9. Kopp, E. B., J. J. Miglietta, A. G. Shrutkowski, C. K. Shih, P. M. Grob, and
M. T. Skoog. 1991. Steady state kinetics and inhibition of HIV-1 reverse
transcriptase by a non-nucleoside dipyridodiazepinone, BI-RG-587, using a
heteropolymeric template. Nucleic Acids Res. 19:3035–3039.
10. Nunberg, J. H., W. A. Schleif, E. J. Boots, J. A. O’Brien, J. C. Quintero, J. M.
Hoffman, E. A. Emini, and M. E. Goldman. 1991. Viral resistance to human
immunodeficiency virus type 1-specific pyridinone reverse transcriptase in-
hibitors. J. Virol. 65:4887–4892.
1
1. Saag, M. S., E. A. Emini, O. L. Laskin, J. Douglas, W. I. Lapidus, W. A.
Schleif, R. J. Whitley, V. W. Byrnes, C. Hildebrand, J. C. Kappes, K. W.
Anderson, F. E. Massari, G. M. Shaw, and the L-697,661 Working Group.
1
993. A short clinical trial of L-697,661, a nonnucleoside inhibitor of HIV-1
reverse transcriptase. N. Engl. J. Med. 329:1065–1072.
1
2. Saari, W. S., J. S. Wai, T. E. Fisher, C. M. Thomas, J. M. Hoffman, C. S.
Rooney, A. M. Smith, J. H. Jones, D. L. Bamberger, M. E. Goldman, J. A.
O’Brien, J. H. Nunberg, J. C. Quintero, W. A. Schleif, E. A. Emini, and P. A.
Anderson. 1992. Synthesis and evaluation of 2-pyridinone derivatives as
HIV-1 specific reverse transcriptase inhibitors. 2. Analogs of 3-aminopyri-
din-2(1H)-one. J. Med. Chem. 35:3792–3802.
13. Sardana, V. V., E. A. Emini, L. Gotlib, D. J. Graham, D. W. Lineberger, W. J.
Long, A. J. Schlabach, J. A. Wolfgang, and J. H. Condra. 1992. Functional
analysis of HIV-1 reverse transcriptase amino acids involved in resistance to
multiple nonnucleoside inhibitors. J. Biol. Chem. 267:17526–17530.
1
4. Sheldrick, G. M. 1990. Phase annealing in SHELX-90: direct methods for
larger structures. Acta Crystallogr. A46:467–473.
15. Smerdon, S. J., J. Jager, J. Wang, L. A. Kohlstaedt, A. S. Chirino, A. J.
Friedman, P. A. Rice, and T. A. Steitz. 1994. Structure of the binding site for
nonnucleoside inhibitors of the reverse transcriptase of human immunode-
ficiency virus type 1. Proc. Natl. Acad. Sci. USA 91:3911–3915.
16. Spence, R. A., W. M. Kati, K. S. Anderson, and K. A. Johnson. 1995.
Mechanism of inhibition of HIV-1 reverse transcriptase by nonnucleoside
inhibitors. Science 267:988–993.
1
7. Stahlhut, M., Y. Li, J. H. Condra, J. Fu, L. Gotlib, D. J. Graham, and D. B.
Olsen. 1994. Purification and characterization of HIV-1 reverse transcriptase
having a 1:1 ration of p66 and p51 subunits. Protein Expr. Purif. 5:614–621.
8. Tantillo, C., J. Ding, A. Jacobo-Molina, R. G. Nanni, P. L. Boyer, S. H.
Hughes, R. Paulwels, K. Andries, P. A. J. Janssen, and E. Arnold. 1994.
Location of anti-AIDS drug binding sites and resistance mutations in the
three-dimensional structure of HIV-1 reverse transcriptase. J. Mol. Biol.
ACKNOWLEDGMENT
1
We are indebted to Jean Kaysen for assistance with manuscript
preparation.
243:369–387.
REFERENCES
1
9. Tucker, T. J., T. A. Lyle, C. M. Wiscount, S. F. Britcher, S. D. Young, W. M.
Sanders, W. C. Lumma, M. E. Goldman, J. A. O’Brien, R. G. Ball, C. F.
Homnick, W. A. Schleif, E. A. Emini, J. R. Huff, and P. S. Anderson. 1994.
Synthesis of a series of 4-(arylethynyl)-6-chloro-4-cyclopropyl-3,4-dihydro-
quinazolin-2(1H)-ones as novel non-nucleoside HIV-1 reverse transcriptase
inhibitors. J. Med. Chem. 37:2437–2444.
1
. Althaus, I. W., J. J. Chou, A. J. Gonzales, M. R. Deibel, K. C. Chou, F. J.
Kezdy, D. L. Romero, P. A. Aristoff, W. G. Tarpley, and F. Reusser. 1993.
Steady-state kinetic studies with the non-nucleoside HIV-1 reverse tran-
scriptase inhibitor U-87201E. J. Biol. Chem. 268:6119–6124.
2
. Byrnes, V. W., V. V. Sardana, W. A. Schleif, J. H. Condra, J. A. Waterbury,
J. A. Wolfgang, W. J. Long, C. L. Schneider, A. J. Schlabach, B. S. Wolanski,
D. J. Graham, L. Gotlib, A. Rhodes, D. L. Titus, E. Roth, O. M. Blahy, J. C.
Quintero, S. Staszewski, and E. A. Emini. 1993. Comprehensive mutant
enzyme and viral variant assessment of human immunodeficiency virus type
20. Wu, J. C., T. C. Warren, J. Adams, J. Proudfoot, J. Skiles, P. Raghavan, C.
Perry, I. Potocki, P. Farina, and P. M. Grob. 1991. A novel dipyridodiaz-
epinone inhibitor of HIV-1 reverse transcriptase acts through a nonsubstrate
binding site. Biochemistry 30:2022–2026.
21. Yonetani, T., and H. Theorell. 1964. Studies on liver alcohol dehydrogenase
complexes. III. Multiple inhibition kinetics in the presence of two competi-
tive inhibitors. Arch. Biochem. Biophys. 106:243–251.
1
reverse transcriptase resistance to the nonnucleoside inhibitors. Antimi-
crob. Agents Chemother. 37:1576–1579.
3
. Carroll, S. S., D. B. Olsen, C. D. Bennett, L. Gotlib, D. J. Graham, J. H.
Condra, A. M. Stern, J. A. Shafer, and L. C. Kuo. 1993. Inhibition of HIV-1
reverse transcriptase by pyridinone derivatives. J. Biol. Chem. 268:276–281.
22. Young, S. D. 1993. Non-nucleoside inhibitors of HIV-1 reverse transcriptase.
Perspect. Drug Discov. Des. 1:181–192.

 Young
Young
 Britcher
Britcher
 Tran
Tran
 Payne
Payne
 Lumma
Lumma
 Lyle
Lyle
 Huff
Huff
 Anderson
Anderson
 Olsen
Olsen
 Carroll
Carroll
 Pettibone
Pettibone
 O'Brien
O'Brien
 Ball
Ball
 Balani
Balani
 Lin
Lin
 Chen
Chen
 Schleif
Schleif
 Sardana
Sardana
 Long
Long
 et al.
et al.