Aginagalde et al.
JOCFeatured Article
shifts (NICS) were computed on B3LYP/6-31G* geometries
1
9
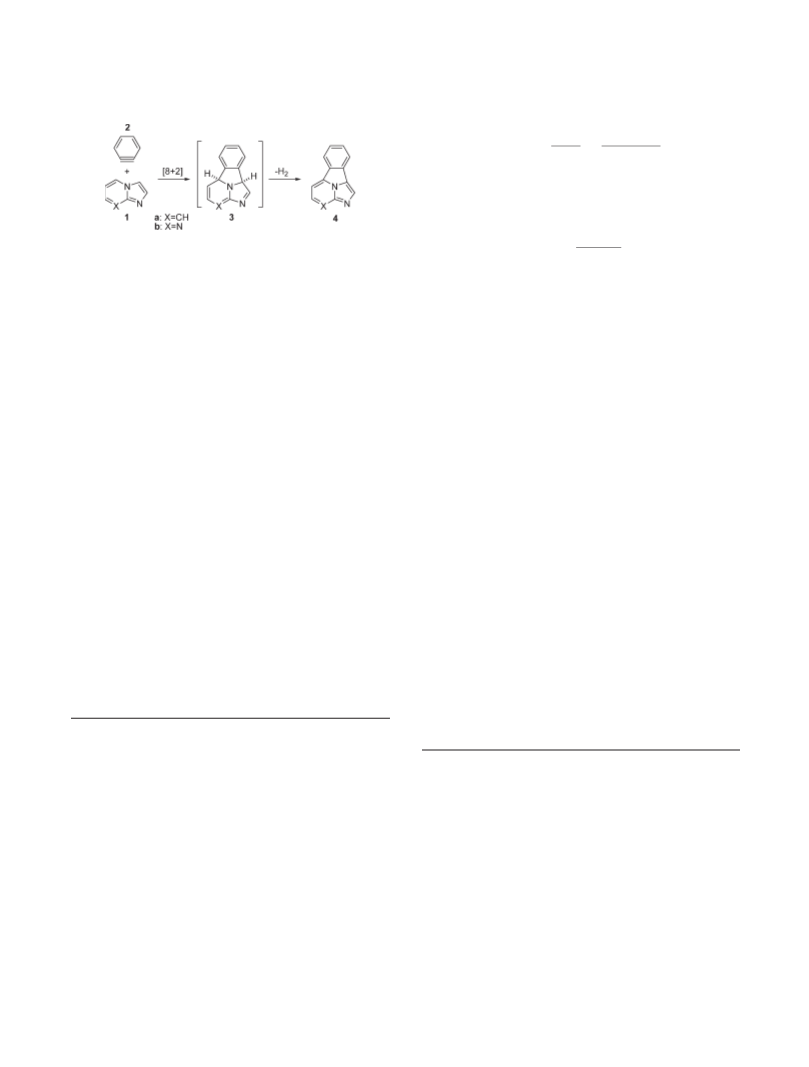
SCHEME 1. Reaction between Imidazo[1,2-a]pyridine 1a or
Imidazo[1.2-a]pyrimidine 1b and Benzine 2 to Yield [8 þ 2]
Cycloadducts 3a,b and Dehydrogenated Products 4a,b
2
0
using the GIAO method.
The synchronicities of several reactions were quantified
2
1
2
2
according to eq 1,
Sy ¼ 1 - 2
n
X
where n is the number of bonds being modified along the
1
jδBi -δBavj
δBav
ð1Þ
n -2
i ¼1
reaction coordinate and δB is the variation of the bond index
i
of a given atom pair i on going from the reactants (R) to the
products (P) via the corresponding transition structure (eq 2):
B Ti S -Bi
R
Given the generality of these reactions, we reasoned that the two
last families of bicyclic heterocycles 1 could be suitable sub-
strates for [8 þ 2] cycloadditions (Scheme 1). Since the starting
molecules 1 must lose aromaticity to yield [8 þ 2] cycloadducts
δBi ¼
ð2Þ
ð3Þ
P
R
B -B
i
i
The average value δBav is given by eq 3:
n
X
-
1
δBav ¼ n
δBi
3, we decided to use benzynes as tetraenophiles since these
i ¼1
highly reactive compounds should provide tetracyclic com-
pounds 3a,b efficiently. We expected that these intermediates
should aromatize to yield benzo[a]imidazo[5,1,2-cd]indolizines
4a and 2,3,9c-triazocyclopenta[j,k]fluorenes 4b. In addition, the
interesting photophysical properties reported for indolizino-
According to these definitions, for a perfectly synchronous
reaction a value of Sy = 1 is achieved.
All the stationary points were characterized by harmonic
2
3
analysis. Activation energies (ΔE
(ΔErxn) were computed including zero-point vibrational energy
ZPVE) corrections.
Car-Parrinello simulations of the intermediate structure 3a
a
) and reaction energies
6
[
4
3,4,5-ab]isoindoles constitute a stimulus to prepare compounds
a,b and to investigate the nature of the [π8s 2 ]
(
þ
24
π s
and [ 2 þ 6 þ 2 ] thermal processes required for their
25
σ s
π s
σ s
were performed using the Quantum Espresso package. The
norm-conserving pseudopotentials C.BLYP-mt.UPF, N.
BLYP-mt.UPF, and H.BLYP-vbc.UPF, obtained from the
Quantum Espresso distribution, were used to describe the
interaction between the core and valence electrons in carbon,
nitrogen, and hydrogen atoms, respectively. The valence wave
functions were expanded in a plane wave basis set with an energy
cut off in the range 40-100 Ry. The exchange-correlation
formation.
Within this context, in this paper we describe the mechan-
ism of the [ 8 þ 2 ] reaction between compounds 1 and
π
s
π s
benzynes as well as the conversion of intermediates 3 in
compounds 4. Next, we describe the preparation of 1-sub-
stituted derivatives of 4a,b via microwave-assisted reaction
between benzyne precursors and imidazo[1,2-a]pyridines
and imidazo[1,2-a]pyrimidines. Finally, the photophysics
of the final compounds thus prepared will be discussed.
2
6
functional was approximated by the BLYP method. The
electrons were assigned a fictitious mass of 400 au. The simula-
tions were performed in the NVT ensemble, where the tempera-
2
7
ture was controlled by the Nos ꢀe -Hoover chain thermostat,
Computational Methods
3
3
3
˚
˚
and V = 11.642 A = 1577.87 A . After reaching the electronic
ground state, the “ionic” positions were energy minimized using
the steepest descent algorithm. Then the “ionic” and electronic
degrees of freedom were propagated using the velocity-Verlet
All the time-independent computational studies reported in
9
10
this work were performed using the Gaussian 03 and Jaguar
programs, within the density functional theory (DFT) frame-
work. Both B3LYP and M06-2X hybrid functionals were
2
8
11
12
13
algorithm and the former gradually (using simulations at T =
0, 100, 200, and 300 K) heated to T = 400 K.
1
4
5
used along with the 6-31G* split-valence basis set. The accuracy
1
5,16
of both methods has been extensively tested for stable mole-
cules and pericyclic reactions. Wiberg bond indices were com-
1
7
Results and Discussion
1
puted using the NBO method. Nucleus-independent chemical
8
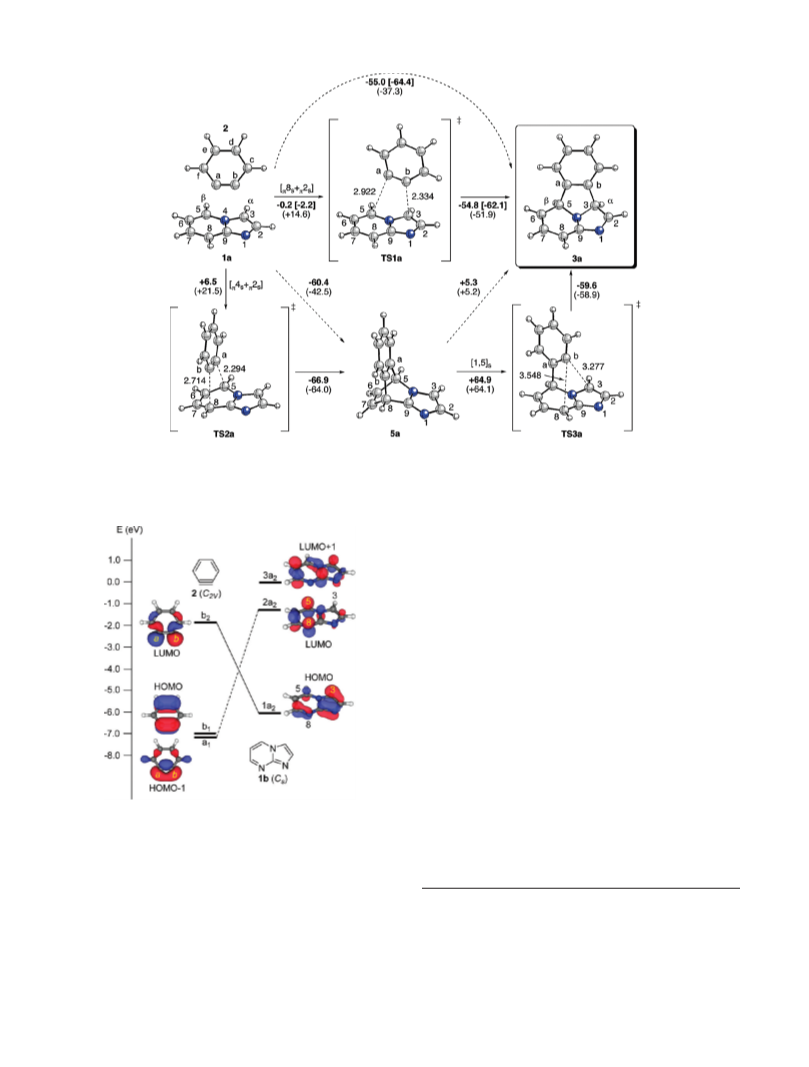
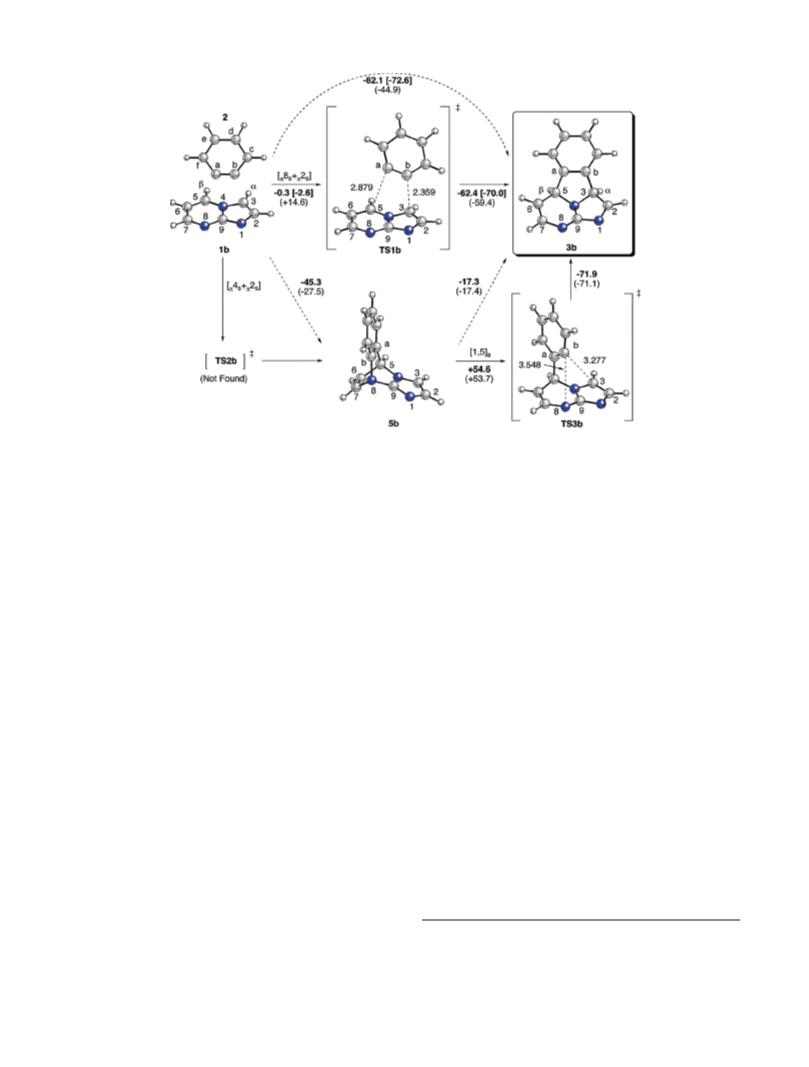
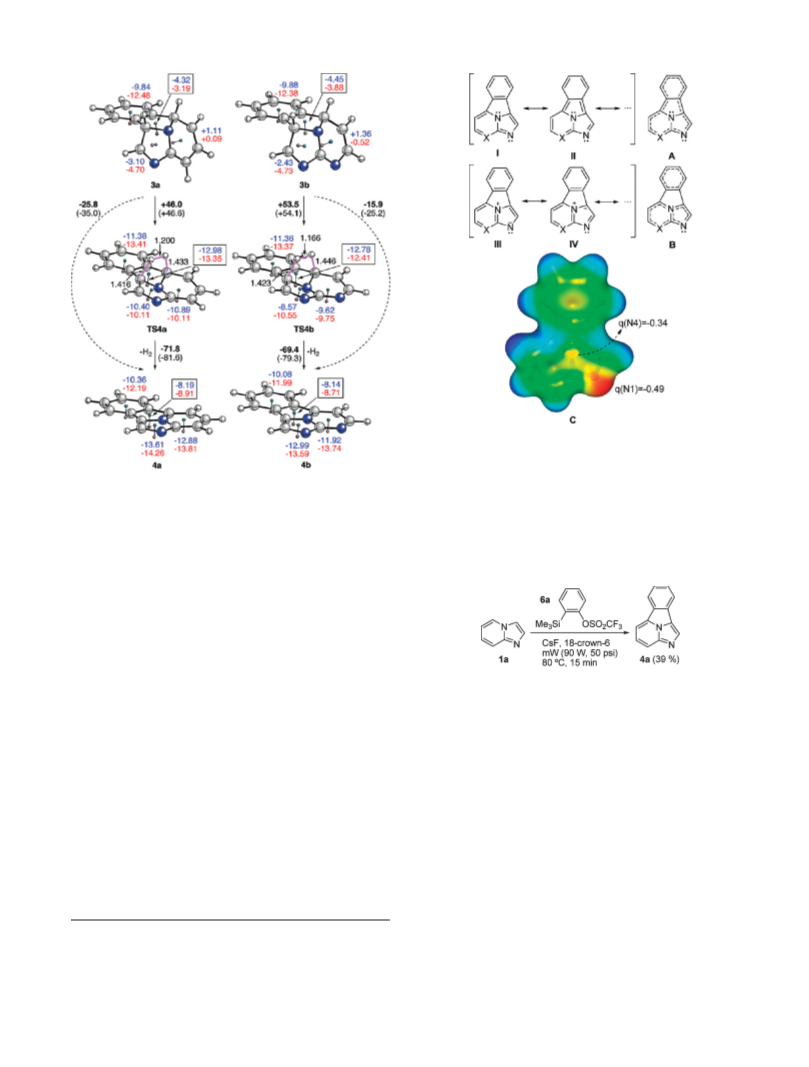
First, we analyzed the possible reactions paths leading to
8 þ 2] cycloadducts 3a,b starting from compounds 1a,b and
[
(9) Gaussian 03, Rev E.01; Frisch, M. J., Ed.; et al. Gaussian, Inc.:
Wallingford, CT, 2004.
(
(
10) Jaguar, version 7.5; Schr o€ dinger, LLC: New York, 2008.
11) (a) Parr, R. G.; Yang, W. Density-Functional Theory of Atoms and
(19) Schleyer, P. v. R.; Maerker, C.; Dransfeld, A.; Jiao, H.; Hommes, N.
Molecules; Oxford University Press: Oxford, 1989. (b) Ziegler, T. Chem. Rev.
S. R. v. E. J. Am. Chem. Soc. 1996, 118, 6317–6318.
(20) Wolinski, K.; Hilton, J. F.; Pulay, P. J. Am. Chem. Soc. 1990, 112,
8251–8260.
(21) Borden, W. T.; Loncharich, R. J.; Houk, K. N. Annu. Rev. Phys.
Chem. 1988, 39, 213–236.
1
991, 91, 651–667.
12) (a) Kohn, W.; Becke, A. D.; Parr, R. G. J. Phys. Chem. 1996, 100,
2974–12980. (b) Becke, A. D. J. Chem. Phys. 1993, 98, 5648–5652. (c) Becke,
(
1
A. D. Phys. Rev. A 1988, 38, 3098–3100.
(
(
13) Zhao, Y.; Truhlar, D. G. Theor. Chem. Acc. 2008, 120, 215–241.
14) Hehre, W. J.; Radom, L.; Schleyer, P. v. R.; Pople, J. A. Ab Initio
(22) (a) Moyano, A.; Peric ꢁa s, M. A.; Valentı
573–582. (b) Lecea, B.; Arrieta, A.; Lopez, X.; Ugalde, J. M.; Cossı
J. Am. Chem. Soc. 1995, 117, 12321–12314.
(23) McIver, J. W. Jr.; Komornicki, A. J. J. Am. Chem. Soc. 1972, 94,
2625–2633.
(24) Car, R.; Parrinello, M. Phys. Rev. Lett. 1985, 55, 2471–2474.
(25) Baroni, S.; Dal Corso, A.; De Gironcoli, S.; Giannozzi, P.; Cavazzoni,
´
C.; Ballabio, G.; Scandolo, S.; Chiarottı, G.; Focher, P.; Pasquarello, A.;
Lasonen, K.; Trave, A.; Car, R.; Marzari, N.; Kokalj, A. Quantum Espresso
Package 2005, http://www.quantum-espresso.org, accessed March 26, 2009.
(26) (a) Becke, A. D. J. Chem. Phys. 1992, 96, 2155–2160. (b) Lee, C.;
Yang, W.; Parr, R. G. Phys. Rev. B 1988, 37, 785–789.
(27) (a) Nos ꢀe , S. Mol. Phys. 1984, 52, 255–268. (b) Hoover, W. G. Phys.
Rev. A 1985, 31, 1695–1697.
(28) Verlet, L. Phys. Rev. 1967, 159, 98–103.
´
, E. J. Org. Chem. 1989, 54,
´
o, F. P.
Molecular Orbital Theory; Wiley: New York, 1986; pp 76-87 and references
cited therein.
(
15) Guner, V.; Khuong, K. S.; Leach, A. G.; Lee, P. S.; Bartberger, M.
D.; Houk, K. N. J. Phys. Chem. A 2003, 107, 11445–11459.
16) (a) Zhao, Y.; Truhlar, D. G. Acc. Chem. Res. 2008, 41, 157–167.
b) Chen, J.-L.; Hong, J.-T.; Wu, K.-J.; Hu, W.-P. Chem. Phys. Lett. 2009,
68, 307–312.
(
(
4
(
(
17) Wiberg, K. B. Tetrahedron 1968, 24, 1083–1096.
18) (a) NBO 5.0: Glendening, E. D.; Badenhoop, J. K.; Reed, A. E.;
Carpenter, J. E.; Bohmann, J. A.; Morales, C. M.; Weinhold, F., Theoretical
Chemistry Institute, University of Wisconsin: Madison, 2001. (b) Reed, A. E.;
Curtiss, L. A.; Weinhold, F. Chem. Rev. 1988, 88, 899–926. (c) Reed, A. E.;
Weinstock, R. B.; Weinhold, F. J. Chem. Phys. 1985, 83, 735–746.
J. Org. Chem. Vol. 75, No. 9, 2010 2777

 Aginagalde, Maialen
Aginagalde, Maialen
 Vara, Yosu
Vara, Yosu
 Arrieta, Ana
Arrieta, Ana
 Zangi, Ronen
Zangi, Ronen
 Cebolla, Vicente L.
Cebolla, Vicente L.
 Delgado-Camon, Arantzazu
Delgado-Camon, Arantzazu
 Cossio, Fernando P.
Cossio, Fernando P.