Inorganic Chemistry
Article
under various conditions was not successful. For 7: 29Si NMR
(INEPT, bb-decoupled, toluene/D2O, TMS) δ −91.0 (s, SiH3),
−151.9 (s, SiSi3Ge); 1H NMR (C6D6, TMS, relative intensity) δ 7.5−
7.1 (m, C6H5), 5.31 (s, 1H, GeHPh2), 3.50 (s, 9H, SiH3); MS [m/e
(relative intensity)] 350.0 (3%, M+), 229.0 (100%, Ph2GeH).
ACKNOWLEDGMENTS
■
We thank the FFG (Austrian Research Promotion Agency,
Wien, Austria) for financial support (Project 838476 “SILA-
VOLT”). We gratefully acknowledge support from NAWI Graz.
Reaction of 7 with CF3SO3H/iBu2AlH. The same procedure that
is described above was followed with 0.24 g (0.7 mmol, 1 equiv) of 7,
0.062 mL (0.7 mmol, 1 equiv) of triflic acid, and 0.13 mL (0.7 mmol, 1
REFERENCES
■
(1) (a) Amato, M.; Palummo, M.; Rurali, R.; Ossicini, S. Chem. Rev.
2014, 114, 1371−1412. (b) Schaffler, F. In Silicon-Germanium
Nanostructures; Shiraki, Y., Usami, N., Eds.; Woodhead Publishers
Ltd.: Cambridge, U.K., 2011; pp 399−432. (c) Kasper, E.; Oehme, M.
Phys. Status Solidi C 2009, 6, 700−703.
(2) Hartmann, J. M.; Andrieu, F.; Lafond, D.; Ernst, T.;
Bogumilowicz, Y.; Delaye, V.; Weber, O.; Rouchon, D.; Papon, A.
M.; Cherkashin, N. Mater. Sci. Eng., B 2008, 154−155, 76−84.
(3) Ritter, C. J.; Hu, C.; Chizmeshya, A. V. G.; Tolle, J.; Klewer, D.;
Tsong, I. S. T.; Kouveta kis, J. J. Am. Chem. Soc. 2005, 127, 9855−
9864.
(4) Horn, H.-G. Chemikerzeitung 1986, 110, 142−150.
(5) (a) Mackay, K. M.; Hosfield, S. T.; Stobart, S. R. J. Chem. Soc. A
1969, 0, 2937−2943. (b) Andrews, T. D.; Phillips, C. S. G. J. Chem.
Soc. A 1966, 0, 46−48.
1
equiv) of iBu2AlH. H and 29Si NMR analysis of the resulting liquid
̈
residue showed the formation of 8, Ph2GeH2, and further products of
unknown composition. Distillation of the crude material at 36−38 °C
and 0.05 mbar afforded a 1:2 mixture of 8 and Ph2GeH2 that could not
be separated. For 8: 29Si NMR (INEPT, bb-decoupled, toluene/D2O,
TMS) δ −90.9 (s, SiH3), −153.4 (s, SiSi3Ge); 1H NMR (C6D6, TMS,
relative intensity) 7.5−7.0 (m, C6H5), 4.43 (s, 2H, GeH2Ph), 3.45 (s,
9H, SiH3); MS [m/e (relative intensity)] 274.0 (2%, M+), 210.0
(100%, PhGeSiSiH3).
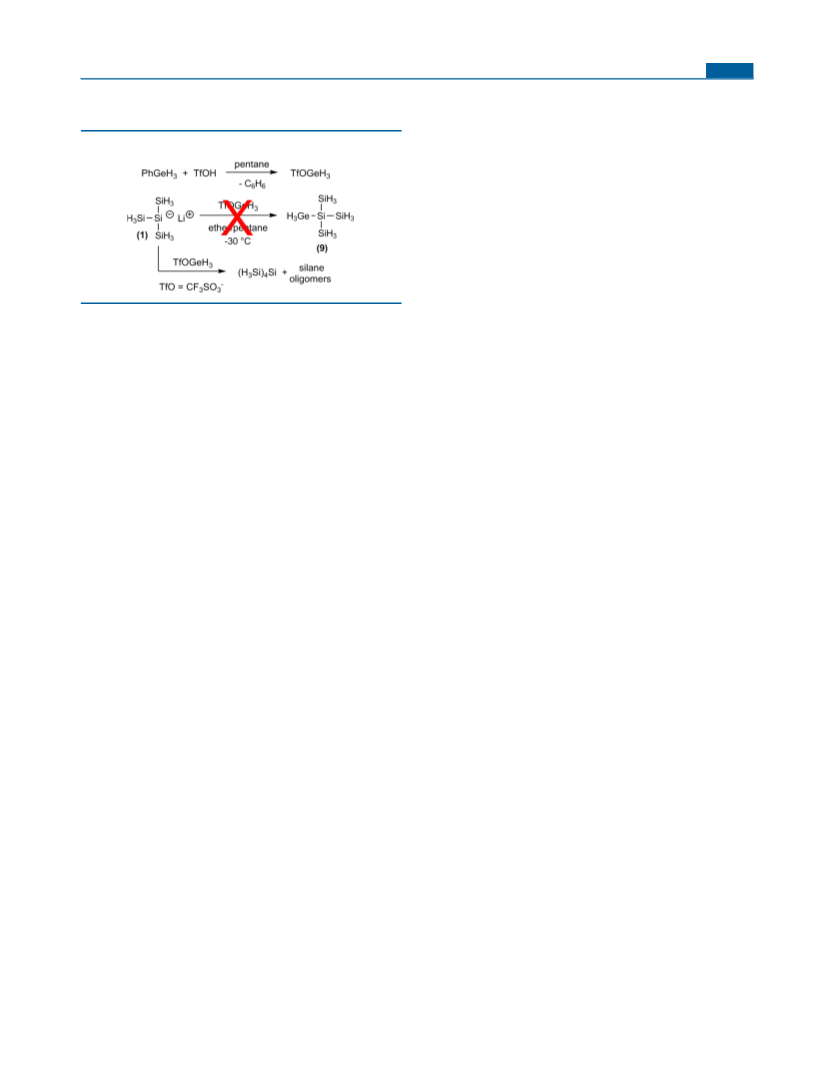
Reaction of 1 with CF3SO3GeH3. A volume of 0.29 mL (3.3
mmol, 1 equiv) of triflic acid was added to a solution of 0.50 g (3.3
mmol, 1 equiv) of PhGeH3 in 5 mL of toluene at −30 °C. After the
mixture had been stirred for 1 h at room temperature, the resulting
mixture was cooled to 0 °C and a solution of 1 in 10 mL of diethyl
ether [freshly prepared from 0.50 g (3.3 mmol, 1 equiv) of Si(SiH3)4
and 3.1 mmol (0.95 equiv) of MeLi] was added via a syringe. After the
mixture had been stirred for an additional 1 h at room temperature,
considerable amounts of insoluble polymeric material of unidentified
structure were obtained. 29Si NMR and GC/MS analysis of the soluble
fraction showed the formation of Si(SiH3)4 along with minor amounts
of other silicon hydrides.
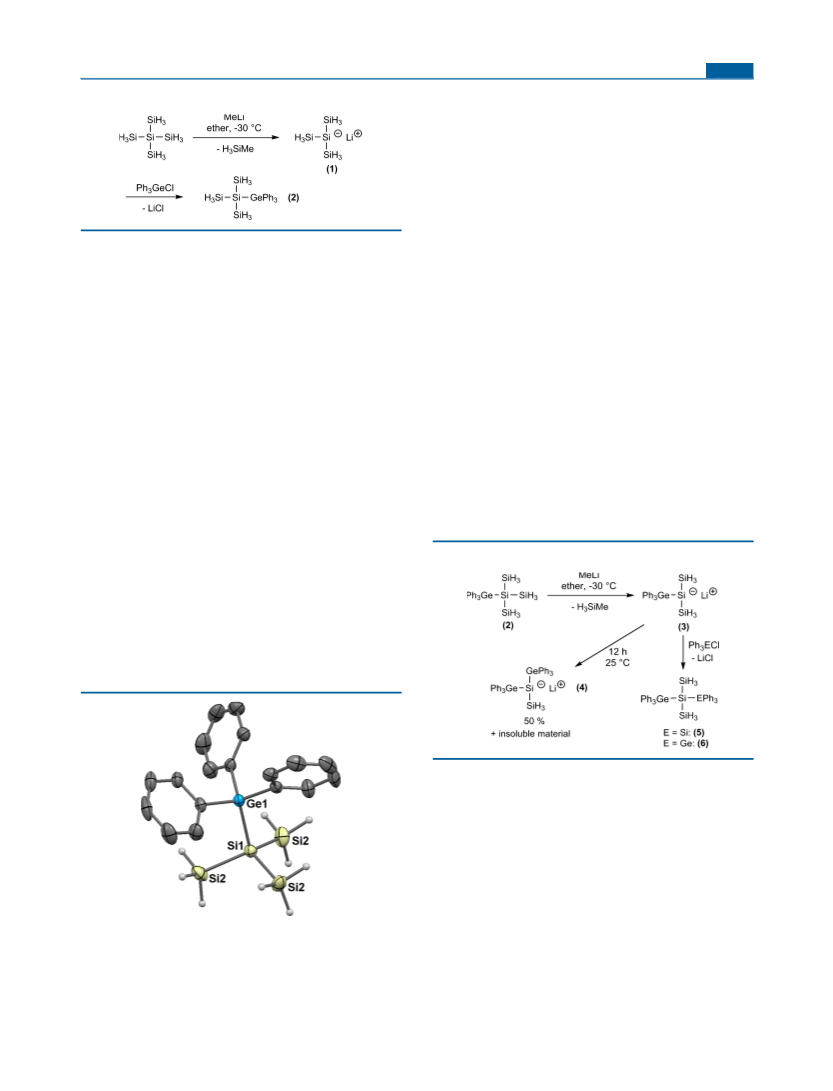
X-ray Crystallography. For X-ray structure analysis, suitable
crystals were mounted onto the tip of glass fibers using mineral oil.
Data collection was performed on a Bruker Kappa Apex II CCD
diffractometer at 100 K using graphite-monochromated Mo Kα (λ =
0.71073 Å) radiation. Details of the crystal data and structure
version 6.1 was used for the structure solution and refinement.20
Absorption corrections were applied using SADABS.21 All non-
hydrogen atoms were refined with anisotropic displacement
parameters. Hydrogen atoms were included in the refinement at
calculated positions using a riding model as implemented in
SHELXTL. Crystallographic data (excluding structure factors) have
been deposited with the Cambridge Crystallographic Data Centre as
supplementary publications CCDC-1450030 (2) and CCDC-1450031
(6). Copies of the data can be obtained free of charge on application
to The Director, CCDC, 12 Union Road, Cambridge CB2 1EZ, UK
(fax (internat.) + 44-1223/336-033; e-mail deposit@ccdc.cam.ac.uk).
(6) (a) Timms, P. L.; Simpson, C. C.; Phillips, C. S. G. J. Chem. Soc.
1964, 0, 1467−1475. (b) Timms, P. L.; Phillips, C. S. G. Inorg. Chem.
1964, 3, 606−607.
(7) Lobreyer, T.; Oberhammer, H.; Sundermeyer, W. Angew. Chem.
1993, 105, 587−588.
(8) Lobreyer, T.; Oeler, J.; Sundermeyer, W. Chem. Ber. 1991, 124,
2405−2410.
(9) Hofler, F.; Brandstatter, E. Monatsh. Chem. 1975, 106, 893−904.
̈
̈
(10) Stueger, H.; Mitterfellner, T.; Fischer, R.; Walkner, C.; Patz, M.;
Wieber, S. Chem. - Eur. J. 2012, 18, 7662−7664.
(11) Amberger, E.; Muhlhofer, E. J. Organomet. Chem. 1968, 12, 55−
̈
62.
(12) Parkanyi, L.; Hernandez, C.; Pannell, K. H. J. Organomet. Chem.
1986, 301, 145−151.
(13) Mallela, S. P.; Ghuman, M. A.; Geanangel, R. A. Inorg. Chim.
Acta 1992, 202, 211−217.
(14) (a) Zaitsev, K. V.; Oprunenko, Y. F.; Churakov, A. V.; Zaitseva,
G. S.; Karlov, S. S. Main Group Met. Chem. 2014, 37, 67−74.
(b) Zaitsev, K. V.; Kapranov, A. A.; Oprunenko, Y. F.; Churakov, A.
V.; Howard, J. A. K.; Tarasevich, B. N.; Karlov, S. S.; Zaitseva, G. S. J.
Organomet. Chem. 2012, 700, 207−213. (c) Uhlig, W. Chem. Ber.
1996, 129, 733−739. (d) Uhlig, W. J. Organomet. Chem. 1993, 463,
73−76.
(15) Literature reports δ1H = 5.61 ppm for Ph3GeH measured in a
CDCl3 solution: Clark, K. B.; Griller, D. Organometallics 1991, 10,
746−750.
ASSOCIATED CONTENT
(16) Literature reports δ1H = 5.00 ppm for Ph2GeH2 measured in a
■
S
* Supporting Information
THF-d8/pentane solution: Castel, A.; Riviere, P.; Satge,
Organometallics 1990, 9, 205−210.
(17) Pangborn, A. B.; Giardello, M. A.; Grubbs, R. H.; Rosen, R. K.;
Timmers, F. J. Organometallics 1996, 15, 1518−1520.
(18) Cannady, J. P.; Zhou, X. (Dow Corning Corp.) WO2008/
051328, 2008.
(19) Zaitsev, K. V.; Kapranov, A. A.; Oprunenko, Y. F.; Churakov, A.
V.; Howard, J. A. K.; Tarasevich, B. N.; Karlov, S. S.; Zaitseva, G. S. J.
Organomet. Chem. 2012, 700, 207−213.
(20) SHELX and SHELXL PC, version 5.03; Bruker AXS, Inc.:
Madison, WI, 1994.
(21) SADABS: Area-detection Absorption Correction; Bruker AXS, Inc.:
Madison, WI, 1995.
́
J.; Ko, H. Y.
The Supporting Information is available free of charge on the
Figures of 1H NMR spectra of 2, 5, and 6, figures of 29Si
NMR spectra of 3, 4, and 7, and tables giving crystal,
collection, and refinement data for the structures of
CIF files giving crystal, collection, and refinement data
for the structures of compounds 2 and 6 (CIF)
AUTHOR INFORMATION
■
Corresponding Author
Notes
The authors declare no competing financial interest.
E
Inorg. Chem. XXXX, XXX, XXX−XXX

 Stueger, Harald
Stueger, Harald
 Christopoulos, Viktor
Christopoulos, Viktor
 Temmel, Andrea
Temmel, Andrea
 Haas, Michael
Haas, Michael
 Fischer, Roland
Fischer, Roland
 Torvisco, Ana
Torvisco, Ana
 Wunnicke, Odo
Wunnicke, Odo
 Traut, Stephan
Traut, Stephan
 Martens, Susanne
Martens, Susanne