Angewandte
Communications
Chemie
Table 1: H2-driven enzymatic reduction of 1 under various conditions.[a]
Entry [1]
(mM)
[FMN]
(mM)
Conv.
Hyd1 TOF
[minÀ1 [c]
Hyd1
FMN
to [%][b]
]
TTN[d] TN[d]
1
2
3
4
2
2
5
10
20
20
10
0.5
0.1
0.1
0.1
0.1
0.1
0.1
100
100
95 {100}
62 {97}
24 {37}
{44}
20.4
7.8
4.8
5.4
5.4
8.4
9.6
3.0
2100
2100
5200
4
20
50
Figure 3. Hyd1-catalysed flavin reduction at different temperatures
(left: FMN; right: FAD). Conversion relative to stand-
Conversion at temp
ard=Conversion at 25
Â100%. The FMN 25–508C bars represent the
ꢀ C
10200 97
average of relative conversions calculated from duplicate experiments,
with the range represented as error bars. Reaction conditions: General
Procedure A (Supporting Information) in phosphate buffer (50 mM,
pH 8.0). Conversion was calculated after 30 min using UV-visible
spectroscopy.
5
7800
9300
9900
74
88
94
6[e]
7[f]
8[g]
{94}
20 then 0.1
{29} then
20200[i] 240[i]
24.2[h]
>99[i]
30 minutes relative to a standard reaction performed at 258C.
This standard temperature and stop time were selected to
leave room for improvement in conversions of FMN and FAD
at the higher temperatures. Reactions at 25–508C using FMN
were performed twice, and the corresponding bars indicate
the average relative conversion with the range of results
represented with error bars (Æ 3–12%). This level of repro-
ducibility is likely to extend to FAD owing to an identical
reaction set up. Results for FMN and FAD may not be
directly comparable due to different purity levels of the
cofactors which were obtained from different suppliers.
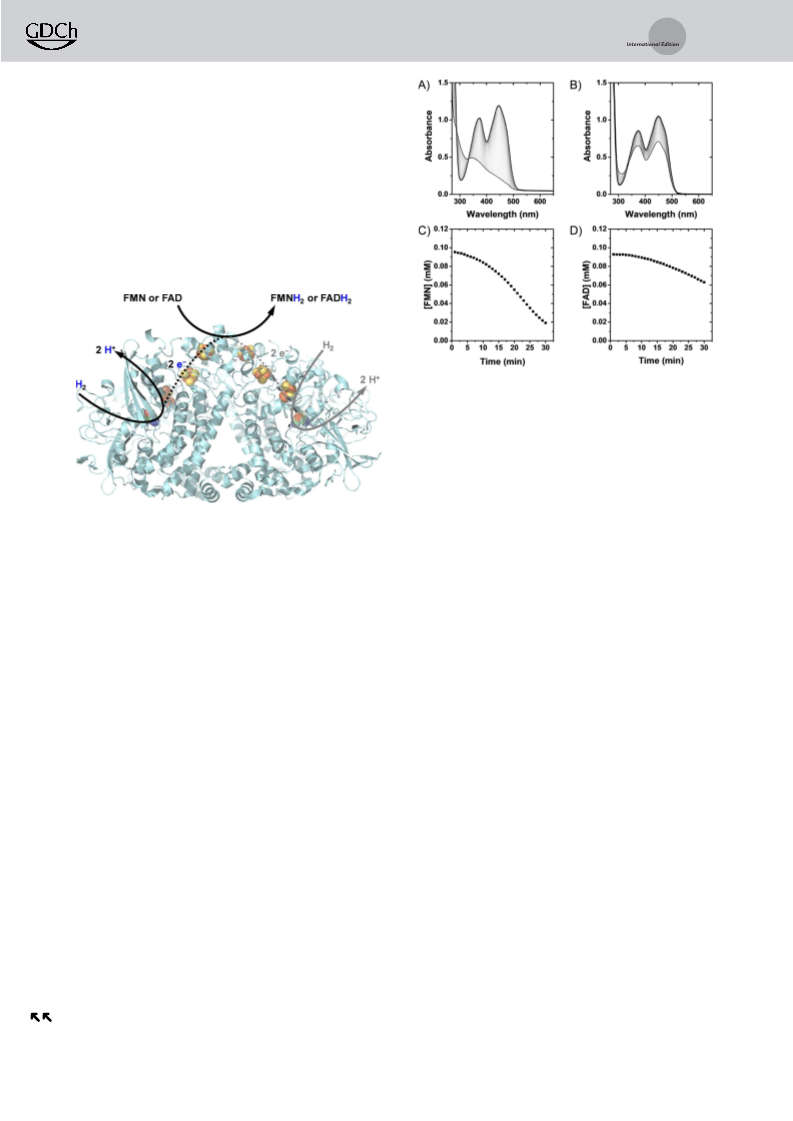
Conversion of FMN and FAD to the reduced forms after
30 min reaction time increased with temperature (Figure 3),
suggesting that Hyd1 is likely to open new doors to cofactor
recycling for flavoenzymes with optimal activity at higher
temperatures.
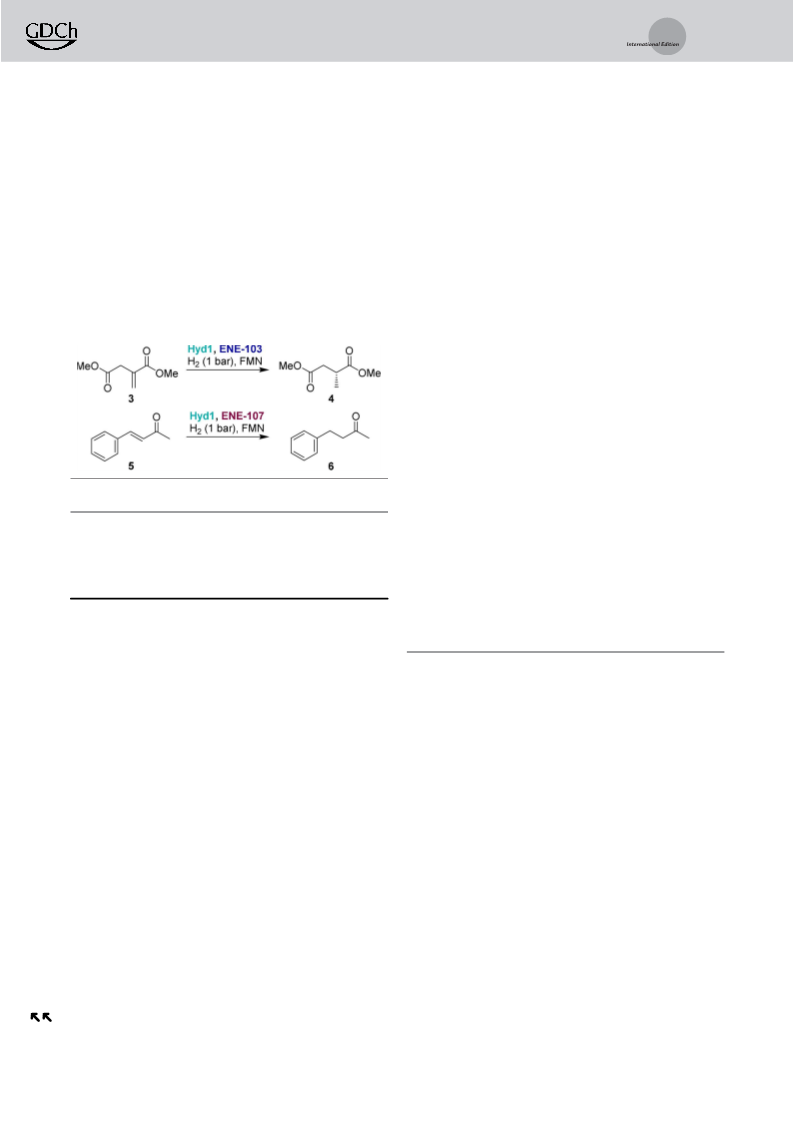
To demonstrate the utility of Hyd1 in biotechnologically-
relevant flavin recycling, we first coupled Hyd1-catalysed
flavin reduction with the OYE-type ene-reductase from
Thermus scotoductus, TsOYE,[32,33] to catalyze enantioselec-
tive reduction of ketoisophorone (1) to (R)-levodione (2,
Table 1). Reactions were conducted according to General
Procedure B (Supporting Information) and monitored using
chiral-phase GC-FID after extraction of the reaction mixture
into ethyl acetate (Supporting Information, Figure S13).
Enantiomeric excess (ee) was always > 99% at the first time
point but decreased to 86–92% from slow racemization under
alkaline conditions as previously reported.[34] Control experi-
ments confirmed good reproducibility (4.4% standard devia-
tion) and that each component is required for conversion
(Supporting Information, Tables S1,S2).
[a] Reaction conditions: In accord with General procedure B using 57 mg
Hyd1, 72 mg TsOYE in Tris-HCl (50 mM, pH 8), 1 vol% DMSO at room
temperature (208C–228C). [b] GC conversion to 2 at 15 h {and 24 h}.
[c] Hyd 1 turnover frequency (mol 2 per mol Hyd1 per min) was
calculated after 60 minutes. [d] Hyd1 total turnover number (mol 2 per
mol Hyd1) and FMN turnover number (mol 2 per molFMN) were
determined at the end of the reaction. [e] 4 bar H2. [f] 358C; some
evaporation of 1 and 2 was observed from GC-FID. [g] 71 mg Hyd1 was
used. [h] Reaction was fed with additional 72 mg TsOYE and 4.2 mM 1 at
66 h and 71 h, respectively. [i] Conversion, Hyd 1 TTN and FMN TN were
determined at 134 hours, additional time point data in the Supporting
Information, Figure S12.
tioselective reduction of 1 by [Cp*Rh(bpy)H]+ meant a care-
ful balance of catalysts was required in that case.[32] This was
not an appreciable issue with our biocatalytic system (Sup-
porting Information, Table S2). Increasing H2 pressure to
4 bar boosted conversion and Hyd1 TOF from 5.4 minÀ1 to
8.4 minÀ1, likely due to improved H2 availability (entries 5,6).
Like Hyd1, TsOYE has enhanced activity at elevated
temperatures,[33] therefore entry 4 was replicated at 358C (see
entry 7). Hyd1 TOF nearly doubled to 9.6 minÀ1 and 94%
conversion was achieved after 24 h, however GC-FID showed
that some of 1 and 2 likely evaporated.
To test stability over time, entry 5 was replicated using
71 mg Hyd1, and as the reaction neared full conversion an
additional 72 mg TsOYE then 4.2 mM 1 was added (66 h and
71 h, respectively, see entry 8). Though the reaction likely still
had active enzymes (Supporting Information, Figure S12), the
reaction was stopped for analysis at 134 h (5.5 days) after
which Hyd1 TTN reached 20200 and FMN TN 240. This
represents an improvement in stability over R. eutropha SH
(TTN 8400) for flavin recycling with TsOYE.[16] The 20200
TTN is of an appropriate order of magnitude for use as
a catalyst in the pharmaceutical and fine chemicals indus-
tries,[35] approaches values measured from commercial grade
enzymatic processes,[36] and there remains room for further
optimization to that end. The demonstrated continuous Hyd1
stability over time (Supporting Information, Figure S12) is an
important performance benchmark for potential commercial
applications, particularly in flow.[37] Furthermore, this appli-
The highest Hyd1 turnover frequency (TOF, 20.4 minÀ1)
and quantitative conversion after 15 h were achieved with
0.5 mM FMN and 2 mM 1 at room temperature (entry 1,
Table 1).
When 0.1 mM FMN was used with varying [1] (entries 2–
5), a Hyd1 total turnover number (TTN) of up to 10200 and
97 FMN turnovers (TN) were achieved after 24 h. This is
comparable to the FMN TN reported for formate-driven Rh-
catalyzed FMNH2 recycling, however background, non-enan-
Angew. Chem. Int. Ed. 2021, 60, 1 – 6
ꢀ 2021 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH
3
These are not the final page numbers!

 Cleary, Sarah E.
Cleary, Sarah E.
 Joseph Srinivasan, Shiny
Joseph Srinivasan, Shiny
 Paul, Caroline E.
Paul, Caroline E.
 Ramirez, Miguel A.
Ramirez, Miguel A.
 Reeve, Holly A.
Reeve, Holly A.
 Vincent, Kylie A.
Vincent, Kylie A.