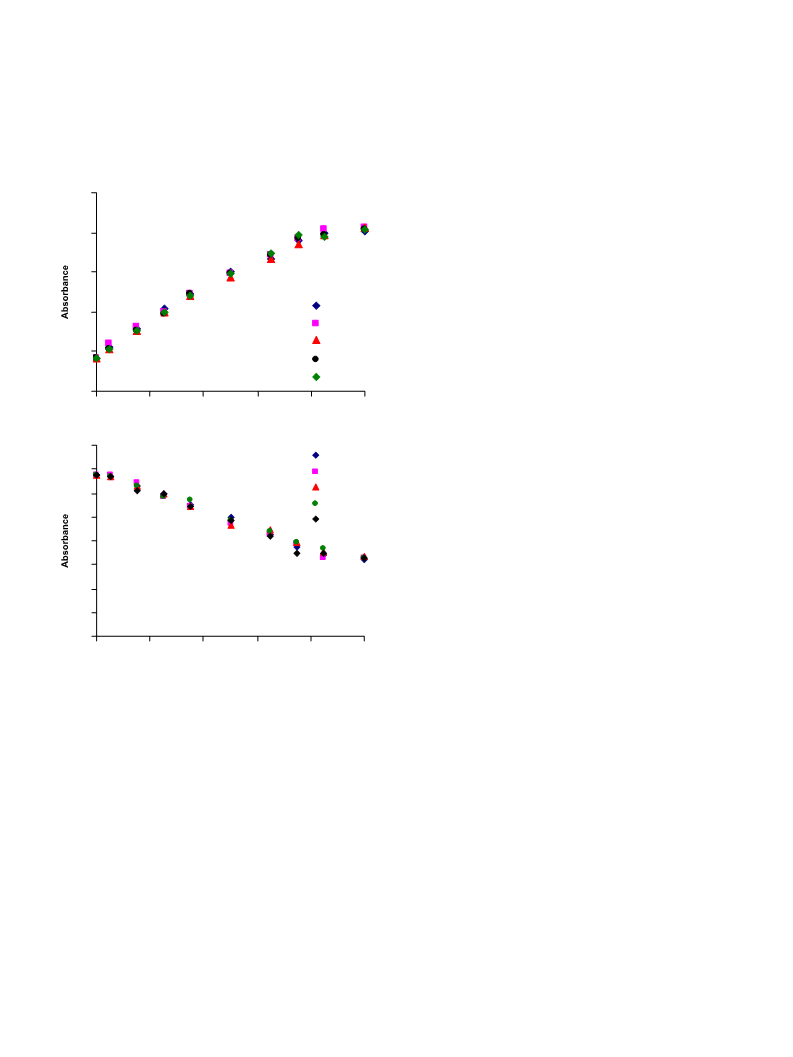N. Kaur, S. Kumar / Tetrahedron 64 (2008) 3168e3175
3175
Bioconjugate Chem. 2005, 16, 949e958; (c) Liu, C.-S.; Hernandez, R.;
Schuster, G. B. J. Am. Chem. Soc. 2004, 126, 2877e2884; (d) Williams,
T. T.; Dohno, C.; Stemp, E. D. A.; Barton, J. K. J. Am. Chem. Soc. 2004,
128, 8148e8158; (e) Pors, K.; Plumb, J. A.; Brown, R.; Spittle, P. T.;
Searcey, M.; Smith, P. J.; Patterson, L. H. J. Med. Chem. 2005, 48,
6690e6695; (f) Chaudhuri, P.; Majunder, H. K.; Bhattacharya, S.
J. Med. Chem. 2007, 50, 2536e2540; (g) Gatto, B.; Zagotto, G.; Sissi,
C.; Cera, C.; Uriarte, E.; Palu, G.; Capranico, G.; Palumbo, M. J. Med.
Chem. 1996, 39, 3114e3122; (h) Murdock, K. C.; Child, R. G.; Fabio,
P. F.; Angier, R. B. J. Med. Chem. 1979, 22, 1024e1030.
slit width of 1.0 nm and matched quartz cells. All metal ion
titrations were performed in CH3OH/H2O (1:1) at pH
7.0ꢂ0.1 (10 mM HEPES buffer). The pH and UVevis combi-
nation titrations were performed with CH3OH/H2O (1:1) un-
buffered solution. All absorption scans were saved as ASC
II files and further processed in ExcelÔ to produce all graphs
shown. Solutions of 3 and 4 were typically 100 mM.
Cu2þ binding characteristics, affinity and stoichiometries of
different complexes were assessed via titrations with Cu(NO3)2.
Titration data are fit with programme Specfit/32, which ana-
lyzes multi-wavelength data using an iterative method to obtain
5. (a) Segura, J. L.; Gomez, R.; Blanco, R.; Reinold, E.; Bauerlem, P. Chem.
Mater. 2006, 18, 2834e2847; (b) Dijk, E. H. V.; Myles, D. J. T.; Veen,
H. V. D.; Hummelen, J. C. Org. Lett. 2006, 8, 2333e2336; (c) Ma, H.;
Kang, M.-S.; Xu, Q.-M.; Kim, K.-S.; Jen, A. K.-Y. Chem. Mater. 2005,
17, 2896e2903; (d) Tutem, E.; Apak, R.; Sozgen, K. J. Inorg. Biochem.
1996, 61, 79e96; (e) Wang, H.; Hua, E.; Yang, P. Talanta 1995, 42,
1519e1524.
the association constant in terms of free or unbound Cu2þ
.
Acknowledgements
6. (a) Devaraj, S.; Saravanakumar, D.; Kandaswamy, M. Tetrahedron Lett.
2007, 48, 3077e3081; (b) Yen, Y.-P.; Ho, K.-W. Tetrahedron Lett.
2006, 47, 1193e1196; (c) Peng, X.; Wu, Y.; Fan, J.; Tian, M.; Han, K.
J. Org. Chem. 2005, 70, 10524e10531; (d) Miyaji, H.; Sessler, J. L.
Angew. Chem., Int. Ed. 2001, 40, 154e157; (e) Jimenez, D.; Martinez-
Manez, R.; Sancenon, F.; Soto, J. Tetrahedron Lett. 2002, 43, 2823e
2825; (f) Jose, D. A.; Kumar, D. K.; Ganguly, B.; Das, A. Org. Lett.
2004, 6, 3445e3448.
We thank CSIR and DST, New Delhi for the financial
assistance.
Supplementary data
Supplementary data associated with this article can be
7. (a) Kumar, S.; Kaur, N. Supramol. Chem. 2006, 18, 137e140; (b) Kaur,
N.; Kumar, S. Dalton Trans. 2006, 3766e3771; (c) Kaur, N.; Kumar, S.
Tetrahedron Lett. 2006, 47, 4109e4112.
References and notes
8. Known compounds with new molecular interaction results: (a) Zhang, L.;
Dong, S.; Zhu, l. Chem. Commun. 2007, 1891e1893; (b) Deraeve, C.;
Pitie, M.; Mazarguil, H.; Meunier, B. New J. Chem. 2007, 31, 193e
195; (c) Margulies, D.; Melman, G.; Shanzer, A. J. Am. Chem. Soc.
2006, 128, 4865e4871.
9. Some known Cu2þ chromogenic sensors: (a) Gunnlaugsson, T.; Leonard,
J. P.; Murray, N. S. Org. Lett. 2004, 6, 1557e1560; (b) Lee, J. S.; Lee,
S. S.; Jeong, I. Y.; Lee, J. Y.; Jung, J. H. Tetrahedron Lett. 2007, 48,
393e396; (c) Xiang, Y.; Tong, A.; Jin, P.; Ju, Y. Org. Lett. 2006, 8,
2863e2866; (d) Martinez, R.; Zapata, F.; Caballero, A.; Espinosa, A.; Tar-
raga, A.; Molina, P. Org. Lett. 2006, 8, 3235e3238; (e) Mu, H.; Gong, R.;
Ma, Q.; Sun, Y.; Fu, E. Tetrahedron Lett. 2007, 48, 5525e5529.
10. Simon, M. S. J. Am. Chem. Soc. 1963, 85, 1974e1977.
1. (a) Boumendjel, A.; Macalou, S.; Ahmed-Belkacem, A.; Blanc, M.;
Pietro, A. D. Bioorg. Med. Chem. 2007, 15, 2892e2897; (b) Katzhendler,
J.; Gean, K.-F.; Bar-Ad, G.; Tashma, Z.; Ben-Shoshan, R.; Ringel, I.;
Bachrach, U.; Ramu, A. Eur. J. Med. Chem. 1989, 24, 23e30; (c) Paull,
K. D.; Zee-Cheng, R. K. Y.; Cheng, C. C. J. Med. Chem. 1976, 19, 337e
339; (d) Krapcho, A. P.; Landi, J. J.; Shaw, K. J., Jr.; Phinney, D. G.;
Hacker, M. P.; McCormack, J. J. J. Med. Chem. 1986, 29, 1370e1373.
2. (a) Prodi, L.; Bolletta, F.; Montalti, M.; Zaccheroni, N. Coord. Chem. Rev.
2000, 205, 59e83; (b) Greig, L. M.; Philip, D. Chem. Soc. Rev. 2001, 30,
287e302; (c) Cram, D. J. Angew. Chem., Int. Ed. Engl. 1988, 27, 1009e
1020.
3. Colour Index, 3rd ed.; The society of dyers and colourists: Bradford, West
Yorkshire, 1971; Vol. 4, pp 4511e4591.
11. Xu, Z.; Qian, X.; Cui, J.; Zhang, R. Tetrahedron 2006, 62, 10117e10122.
12. Xu, Z.; Qian, X.; Cui, J. Org. Lett. 2005, 7, 3029e3032.
13. (a) For 3: Greenhalgh, C. W.; Hughes, N. J. Chem. Soc. C 1968, 1284e
1288; (b) For 5: Koeberle, K.; Steigerwald, C.; Schweizer, R. U.S. Patent
2,050,661, 1936; Chem. Abstr. 1936, 30, 6953.
4. (a) Bergeron, F.; Nair, V. K.; Wagner, J. R. J. Am. Chem. Soc. 2006, 128,
14798e14799; (b) Dias, N.; Gossens, J.-F.; Baldeyrou, B.; Lansiaux, A.;
Colson, P.; Salvo, A. D.; Bernal, J.; Turnbull, A.; Mincher, D. J.; Bailly, C.

 Kaur, Navneet
Kaur, Navneet
 Kumar, Subodh
Kumar, Subodh