Synthon-Assisted Short Oacid-H‚‚‚Owater H-Bonds
J. Phys. Chem. A, Vol. 108, No. 43, 2004 9415
(2) Jeffrey, G. A. An Introduction to Hydrogen Bonding; Oxford
University Press: New York, 1997.
(3) Scheiner, S. Hydrogen Bonding. Theoretical PerspectiVe; Oxford
University Press: Oxford, U.K., 1997.
Red Shift in the O-H Infrared Stretching Vibration. In
accordance with the covalent O-H bond lengthening in short-
strong H-bonds, the stretching frequency ,νs, of the COOH group
in 1 shows a red shift in the IR spectrum (KBr). The O-H
stretching vibrations of Oacid-H‚‚‚Ow in solid 1 appear at 1454,
1386, 1338, 1277, 1213, 1174, and 1134 cm-1. The broad peaks
at 3462 and 3219 cm-1 arise from the O-H stretching vibrations
of the COOH and water groups (see the Supporting Informa-
tion). These IR frequencies match the empirical νs(O-H) versus
(4) Desiraju, G. R.; Steiner, T. The Weak Hydrogen Bond in Structural
Chemistry and Biology; Oxford University Press: Oxford, U.K., 1999.
(5) Reviews: (a) Speakman, J. C. Struct. Bonding 1972, 12, 141. (b)
Huggins, M. L. Angew. Chem., Int. Ed. Engl. 1971, 10, 147. (c) Emsley, J.
Chem. Soc. ReV. 1980, 9, 91. (c) Jeffrey, G. A. Cryst. ReV. 1995, 4, 213.
(d) Gilli, G.; Gilli, P. J. Mol. Struct. 2000, 552, 1. (e) Steiner, T. Angew.
Chem., Int. Ed. 2002, 41, 48.
(6) Some selected recent papers: (a) Kuppers, H.; Takusagawa, F.;
Koetzle, T. F. J. Chem. Phys. 1985, 82, 56360. (b) Madsen, G. K. H.;
Wilson, C.; Nymand, C. T. M.; McIntyre, G. J.; Larsen, F. K. J. Phys.
Chem. A 1999, 103, 8684. (c) Steiner, T. Chem. Commun. 1999, 2299. (d)
Davidson, M. G.; Goeta, A. E.; Howard, J. A. K.; Lamb, V.; Mason, S. A.
New J. Chem. 2000, 24, 477. (e) Macchi, P.; Iversen, B. B.; Sironi, A.;
Chakoumakos, B. C.; Larsen, F. K. Angew. Chem., Int. Ed. 2000, 39, 2719.
(f) Remer, L. C.; Jenses, J. H. J. Phys. Chem. A 2000, 104, 9266. (g) Steiner,
T.; Schreurs, A. M. M.; Lutz, M.; Kroon, J. New J. Chem. 2001, 25, 174.
(h) Olovsson, I.; Ptasiewiczbak, H.; Gustafsson, T.; Majerz, I. Acta
Crystallogr. 2001, B57, 311. (i) Rodrigues, B. L.; Tellgren, R.; Fernandes,
N. G. Acta Crystallogr. 2001, B57, 353. (j) Parra, R. D.; Zeng, H.; Zhu, J.;
Zheng, C.; Zeng, X. C.; Gong, B. Chem.sEur. J. 2001, 7, 4353. (k)
Kumarswamy, K. C.; Kumarswamy, S.; Kommana, P. J. Am. Chem. Soc.
2001, 123, 12642. (l) Wilson, C. C. New J. Chem. 2002, 26, 1733. (m)
Janiak, C.; Scharmann, T. G.; Mason, S. A. J. Am. Chem. Soc. 2002, 124,
14010.
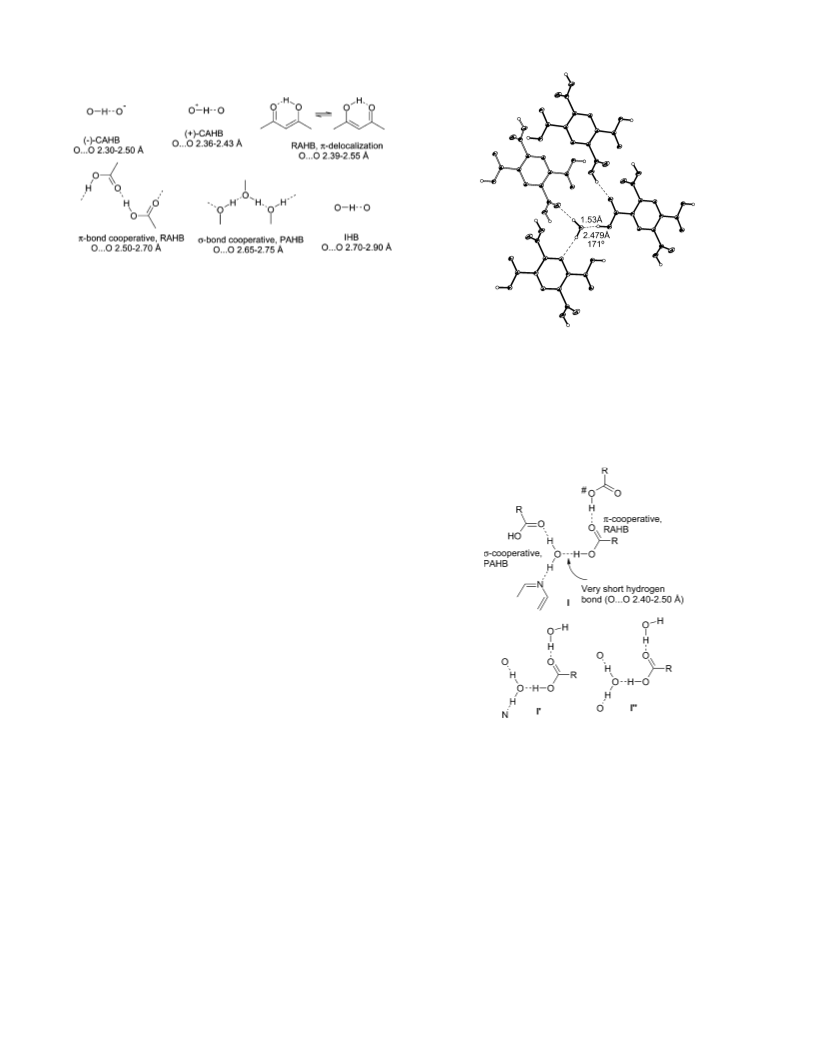
O‚‚‚O distance relation.7b,47 The hydrogen-bond energy of Oacid
-
H‚‚‚Ow (O‚‚‚O ) 2.49 Å, νs(O-H) ) 1350 cm-1) is estimated
as EHB ≈ 16 kcal mol-1 through comparison with a closely
related crystal structure.48 Differential scanning calorimetry and
thermal gravimetric analysis confirm the phase purity and
stoichiometry of tetraacid dihydrate 1 (Tonset H2O release at 144
°C; Tonset melting at 207 °C, decomposes).
Conclusions
We report the occurrence of short hydrogen bonds in pyrazine
di-, tri-, and tetracarboxylic acid dihydrates 1-5. A very short
O-H‚‚‚O hydrogen bond is characterized in tetraacid 1 and
triacid 5, and short O-H‚‚‚O hydrogen bonds are analyzed in
diacids 2, 3, and 4. The variable temperature neutron diffraction
analysis of dihydrate 1 and low-temperature X-ray diffraction
of dihydrates 2-5 provide an in-depth understanding of the short
hydrogen bond in multicenter synthons I and I′. The synergy
from resonance and polarization assistance in the finite, neutral
array is sufficient to result in short (strong) O-H‚‚‚O hydrogen
bonds when the carboxylic acid donor is activated. This novel
synthon-assisted hydrogen-bond shortening phenomenon is
postulated from the neutron diffraction crystal structure of 1,
verified through the X-ray structures of 2-5, and examples
retrieved from the Cambridge Structural Database (CSD) that
obey SAHB. The present study shows that not only charge and
resonance assistance but also polarization assistance can lead
to very short intermolecular O-H‚‚‚O hydrogen bonds (O‚‚‚O
) 2.4-2.5 Å). This is the first study of a very short Oacid-H‚
‚‚Ow hydrogen bond by variable temperature neutron diffraction
and, in general, a systematic analysis of short O-H‚‚‚O
hydrogen bonds in neutral arrays. The similarity of hydrogen-
bonded synthons discussed in this paper to multicenter short
H-bonds in enzyme-inhibitor crystal structures extends these
results to structure-based drug design. Tetraacid 1 and its amide
have potential in the crystal engineering of square network
architectures.
(7) (a) Reid, C. J. J. Chem. Phys. 1959, 30, 182. (b) Novak, A. Struct.
Bonding 1974, 18, 177.
(8) Humbel, S. J. Phys. Chem. A 2002, 106, 5517.
(9) Steiner, T. J. Phys. Chem. A 1998, 102, 7041.
(10) (a) Pietrzak, M.; Wehling, J.; Limbach, H.-H.; Golubev, N. S.;
Lo´pez, C.; Claramunt, R. M.; Elguero, J. J. Am. Chem. Soc. 2001, 123,
4338. (b) Bene, J. A. D.; Perera, S. A.; Bartlett, R. J.; Elguero, J.; Alkorta,
I.; Lo´pez-Leonardo, C.; Alajarin, M. J. Am. Chem. Soc. 2002, 124, 6393.
(c) Bene, J. A. D.; Perera, S. A.; Bartlett, R. J.; Ya´n˜ez, M.; Mo´, O.; Elguero,
J.; Alkorta, I. J. Phys. Chem. A 2003, 107, 3126. (d) Hibbert, F.; Emsley,
J. AdV. Phys. Org. Chem. 1990, 26, 255.
(11) (a) Flensburg, C.; Larsen, S.; Stewart, R. F. J. Phys. Chem. 1995,
99, 10130. (b) Mallinson, P. R.; Woz´niak, K.; Smith, G. T.; McCormack,
K. L. J. Am. Chem. Soc. 1997, 119, 11502. (c) Madsen, G. K. H.; Iversen,
B. B.; Larsen, F. K.; Kapon, M.; Reisner, G. M.; Herbstein, F. H. J. Am.
Chem. Soc. 1998, 120, 10040. (d) Madsen, D.; Flensburg, C.; Larsen, S. J.
Phys. Chem. 1998, 102, 2177.
(12) (a) Cleland, W. W. Biochemistry 1992, 31, 317. (b) Cleland, W.
W.; Kreevoy, M. M. Science 1994, 264, 1887. (c) Frey, P. A.; Whitt, S.
A.; Tobin, J. B. Science 1994, 264, 1927. (d) Dunitz, J. D. Science 1994,
264, 670. (e) Usher, K. C.; Remington, S. J.; Martin, D. P.; Drueckhammer,
D. G. Biochemistry 1994, 33, 7753. (f) Tobin, J. B.; Whitt, S. A.; Cassidy,
C. S.; Frey, P. A. Biochemistry 1995, 34, 6919. (g) Ash, E. L.; Sudmeier,
J. L.; De Fabo, E. C.; Bachovchin. Science 1997, 278, 1128. (h) Chen, J.;
McAllister, M. A.; Lee, J. K.; Houk, K. N. J. Org. Chem. 1998, 63, 4611.
(i) Massiah, M. A.; Viragh, C.; Reddy, P. M.; Kovach, I. M.; Johnson, J.;
Rosenberry, T. L.; Mildvan, A. S. Biochemistry 2001, 40, 5682. (j) Kim,
K. S.; Kim, D.; Lee, J. Y.; Tarkeshwar, P.; Oh, K. S. Biochemistry 2002,
41, 5300. (k) Schiøtt, B. Chem. Commun. 2004, 498.
(13) Ha¨ggman, L.; Lindblad, C.; Oskarsson, H.; Ullstro¨m, A.; Person,
I. J. Am. Chem. Soc. 2003, 125, 3631.
Acknowledgment. A.N. thanks the Department of Science
and Technology (SR/S5/OC-02/2002) for research funding. P.V.
and N.J.B. thank the CSIR and UGC for fellowships. University
of Hyderabad acknowledges financial support from the UPE
program. J.A.K.H. thanks the EPRSC for a Senior Research
Fellowship and support to H.P. R.M. thanks the ORS for
support. We thank Dr. M. T. Kirchner (University of Hyderabad)
for discussion.
(14) (a) Ohhara, T.; Uekusa, H.; Ohashi, Y.; Tanaka, I.; Kumazawa, S.;
Niimura, N. Acta Crystallogr. 2001, B57, 551. (b) Wilson, C. C.; Shankland,
K.; Shankland, N. Z. Kristallogr. 2001, 216, 303. (c) Wilson, C. C.; Goeta,
A. E. Angew Chem., Int. Ed. 2004, 43, 2095.
(15) Katz, B. A.; Spencer, J. R.; Elrod, K.; Luong, C.; Mackman, R.
L.; Rice, M.; Sprengeler, P. A.; Allen, D.; Janc, J. J. Am. Chem. Soc. 2002,
124, 11657.
(16) (a) Kim, K. S.; Suh, S. B.; Kim, J. C.; Hong, B. H.; Lee, E. C.;
Yun, S.; Tarakeshwar, P.; Lee, J. Y.; Kim, Y.; Ihm, H.; Kim, H. G.; Lee,
J. W.; Kim, J. K.; Lee, H. M.; Kim, D.; Cui, C.; Youn, S. J.; Chung, H. Y.;
Choi, H. S.; Lee, C.-W.; Cho, S. J.; Jeong, S.; Cho, J.-H. J. Am. Chem.
Soc. 2002, 124, 14268. (b) Suh, S. B.; Kim, J. C.; Choi, Y. C.; Yun, S.;
Kim, K. S. J. Am. Chem. Soc. 2004, 126, 2186.
(17) (a) Benoit, M.; Marx, D.; Parrinello, M. Nature 1998, 392, 258.
(b) Loubeyre, P.; LeToullec, R.; Wolanin, E.; Hanfland, M.; Hausermann,
D. Nature 1998, 397, 503.
(18) (a) Gilli, G.; Bellucci, F.; Ferretti, V.; Bertolasi, V. J. Am. Chem.
Soc. 1989, 111, 1023. (b) Bertolasi, V.; Gilli, P.; Ferretti, V.; Gilli, G. J.
Am. Chem. Soc. 1991, 113, 4917. (c) Gilli, P.; Bertolasi, V.; Ferretti, V.;
Gilli, G. J. Am. Chem. Soc. 1994, 116, 909. (d) Bertolasi, V.; Gilli, P.;
Ferretti, V.; Gilli, G. Chem.sEur. J. 1996, 2, 925. (e) Gilli, P.; Bertolasi,
V.; Ferretti, V.; Gilli, G. J. Am. Chem. Soc. 2000, 122, 10405. (f) Gilli, P.;
Bertolasi, V.; Pretto, L.; Ferretti, V.; Gilli, G. J. Am. Chem. Soc. 2004,
126, 3845.
Supporting Information Available: Synthesis of pyrazine
acids 1, 3, and 5, neutron diffraction and X-ray diffraction
experimental details, tables of crystal data, crystallographic files
of neutron and X-ray structures (cif format), electrostatic surface
potential maps of water, difference Fourier maps of 1, and IR
spectra of 1 (KBr and Nujol). This material is available free of
References and Notes
(1) Jeffrey, G. A.; Saenger, W. Hydrogen Bonding in Biological
Structures; Springer-Verlag: Berlin, 1991.

 Vishweshwar, Peddy
Vishweshwar, Peddy
 Babu, N. Jagadeesh
Babu, N. Jagadeesh
 Nangia, Ashwini
Nangia, Ashwini
 Mason, Sax A.
Mason, Sax A.
 Puschmann, Horst
Puschmann, Horst
 Mondal, Raju
Mondal, Raju
 Howard, Judith A. K.
Howard, Judith A. K.