
Angewandte Chemie - International Edition p. 8321 - 8328 (2021)
Update date:2022-08-22
Topics:
 Jiang, Wen-Shuang
Jiang, Wen-Shuang
 Ji, Ding-Wei
Ji, Ding-Wei
 Zhang, Wei-Song
Zhang, Wei-Song
 Zhang, Gong
Zhang, Gong
 Min, Xiang-Ting
Min, Xiang-Ting
 Hu, Yan-Cheng
Hu, Yan-Cheng
 Jiang, Xu-Liang
Jiang, Xu-Liang
 Chen, Qing-An
Chen, Qing-An
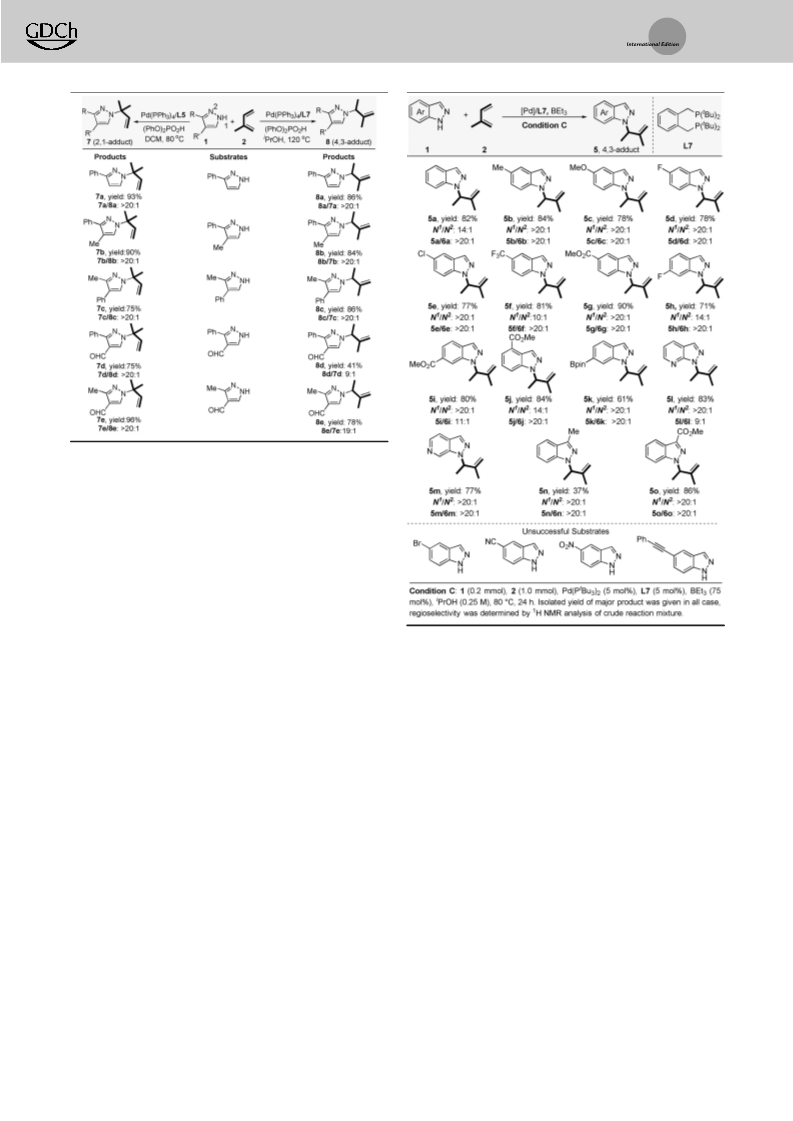
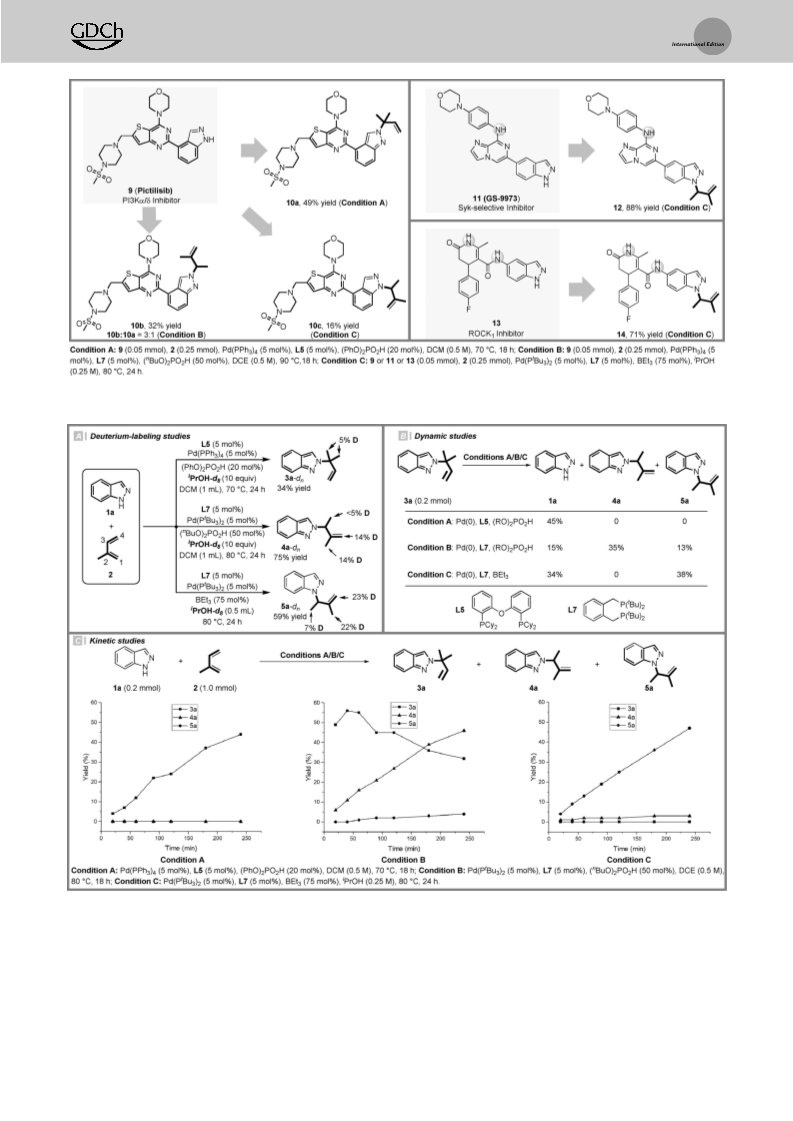
Depending on the reactant property and reaction mechanism, one major regioisomer can be favored in a reaction that involves multiple active sites. Herein, an orthogonal regulation of nucleophilic and electrophilic sites in the regiodivergent hydroamination of isoprene with indazoles is demonstrated. Under Pd-hydride catalysis, the 1,2- or 4,3-insertion pathway with respect to the electrophilic sites on isoprene could be controlled by the choice of ligands. In terms of the nucleophilic sites on indazoles, the reaction occurs at either the N1- or N2-position of indazoles is governed by the acid co-catalysts. Preliminary experimental studies have been performed to rationalize the mechanism and regioselectivity. This study not only contributes a practical tool for selective functionalization of isoprene, but also provides a guide to manipulate the regioselectivity for the N-functionalization of indazoles.
View More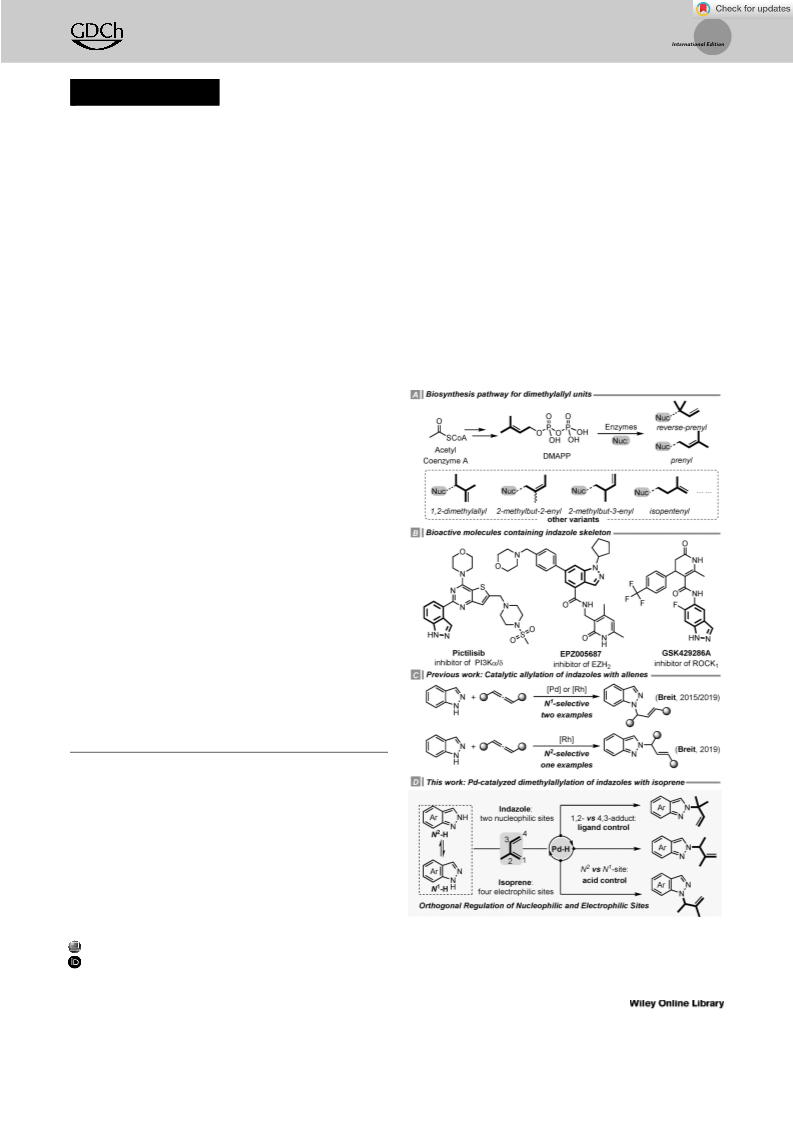





Shanghai Kangxin Chemical Co., Ltd
Contact:+86 21 60717227
Address:118,Ganbai Village,Waigang Town,Jiading District,Shanghai
Jining tiansheng chemical co.,ltd.
Contact:+86-537-5158722
Address:ROOM 1011, BLOCK B, CUIDU INTERNATIOAL BUSINESS CENTER, JINING CITY, CHINA
Xiamen Kaijia Imp & Exp Co., Ltd.
Contact:86-592-5101177
Address:Room406 Luhui Building No. 65 Haitian Road Huli Xiamen,China.
Contact:
Address:ROOM 1715, No#345 Jin Xiang Road, Pudong District
Jiangsu Zenji Pharmaceuticals LTD
Contact:+86-025-83172562; +1-224-888-1133(USA)
Address:No.5 Xinmofan Road
Doi:10.1016/j.saa.2016.10.004
(2017)Doi:10.1021/j100210a037
(1982)Doi:10.1007/BF01518019
()Doi:10.1007/BF00905301
()Doi:10.1016/0040-4039(93)89010-N
(1993)Doi:10.1007/s13738-015-0762-1
(2016)