Full Paper
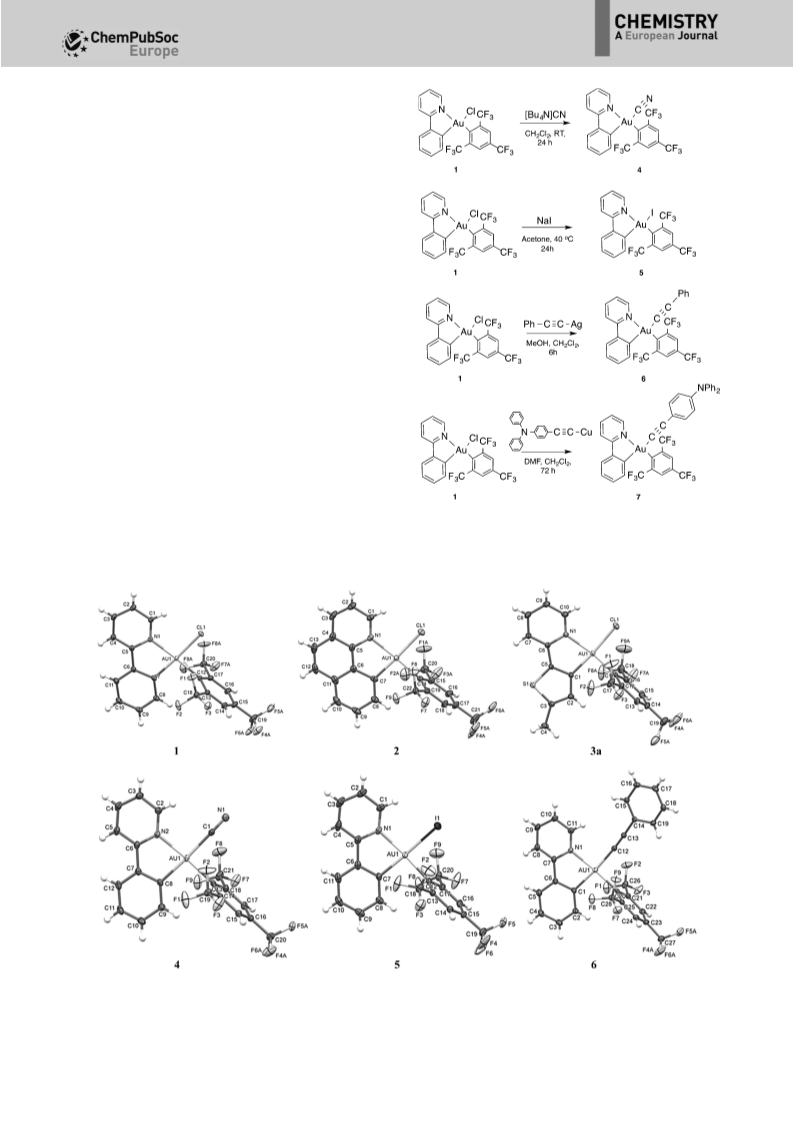
MeOH was stirred with complex 1 in CH2Cl2 at room tempera-
ture (Scheme 2).[10a] Further work-up and purification using
column chromatography gave complex 6 in 30% yield. Adopt-
ing similar reaction conditions for the preparation of 7 resulted
in a dismal yield. However, optimal reaction conditions were
successfully obtained by generating in situ the CuI acetylide
starting from (4-diphenylamino-phenyl)trimethylsilyl alkyne,
CsF and CuI in DMF,[10b] which after subsequent treatment with
complex 1 in DMF gave the desired complex 7 in 49% yield
(Scheme 2). Extensive characterization of complexes 6 and 7
equivalent ligands, especially with the constraints of our che-
lating cyclometalated ligands (N^C). The compound
(5m-thpy)AuCl(Fmes) presents two polymorphic crystal struc-
¯
tures 3a (monoclinic, P21/c) and 3b (triclinic, P1), both poly-
morphs are reported in our manuscript (see the Supporting In-
formation for 3b). The bond distances AuꢁN and AuꢁCN^C
were found to lie in the ranges 2.083(2)–2.117(2) ꢁ and
2.004(3)–2.052(3) ꢁ. They are significantly longer than those re-
ported in the X-ray structure of the starting dichloride com-
plex[11] which exhibits values of 2.034(1) and 1.950(2) ꢁ, respec-
tively. The former Au-N distances are also slightly longer than
those found for analogous N^C AuIII complexes, between
2.052(2) and 2.082(2).[7] The bond distance AuꢁCl in complexes
1–3 is in the range 2.3384(12)–2.3525 (6) ꢁ. These values are
very similar to the one of 2.361(8) ꢁ reported in the dichloride
complex, for which a structural trans influence was observed
for the AuꢁCl bond distances (a shorter value of 2.282(5) ꢁ is
reported when trans to the pyridyl nitrogen). In 1–3 the chlo-
ride ligand is always trans to the Au-CN^C bond and the AuꢁCl
bond length shows no real deviation with the change of the
cyclometalating core. The same observation can be done for
the AuꢁCFmes bond length which only varies by ꢂ0.014 ꢁ in
1–6. The AuꢁCalkynyl bond trans to the carbon atom of the pyr-
idyl ring in 6 is 2.039(3) ꢁ and falls in the range of 1.993(2)–
2.052(2) ꢁ found for analogous complexes.[7] The bond angle
XꢁAuꢁCFmes (X=Cl, CCN, I or CCCPh) is always nearly ideal around
908 in the range 88.12(10)–91.93(12)8, except for 3a where it is
significantly larger with 93.13(8)8. The polymorphic structures
3a and 3b, which present significantly different ClꢁAuꢁCFmes
bond angles of 89.25(13) and 93.13(8)8 are very good examples
to support the evidence that the geometric parameters, espe-
cially bond and dihedral angles, have to be considered with
care because they can be perturbed by packing forces and in-
termolecular interactions. The opposite NꢁAuꢁCN^C chelate
bite angles of the complexes are all smaller than 83.18(12)8
(for 2) while the neighboring CN^CꢁAuꢁCFmes and NꢁAuꢁX
bond angles are contrarily greater than 92.74(13)8 and
94.34(8)8, respectively (except for 3a for which the former
angle is 91.59(11)8; Table 1). The Fmes ligand is almost perpen-
dicular to the chelating metallacycle in each structure. The di-
hedral angle defined by the mean planes of the N^CAuXCFmes
core and the six-membered ring of the Fmes ligand is in the
narrow interval of 88.49(7)-89.43(8)8. For complex 6 the dihe-
dral angle shows a larger devia-
1
was carried out using H and 13C NMR, 2D NMR studies, ele-
mental analysis and the chemical identity was additionally cor-
roborated for 6 by a single crystal X-ray diffraction analysis.
The 13C NMR spectrum in CD2Cl2 showed two characteristic res-
onances at d=126.2 and 134.2 ppm for complex 6 that can be
attributed to the Ca and Cb of the alkyne bound to the AuIII
metal center, respectively. Similar resonances were shifted
downfield for 7 at d=115.9 and 103.5 ppm.
The influence of the different ancillary ligands on thermal
stability of the selected complexes 1, 4 and 7 were evaluated
in the solid state using thermogravimetric analysis (TGA) (see
Figures S1–S3 in the Supporting Information,). While 4 bearing
the cyanide ligand showed the highest stability with the onset
of total degradation (Td) at 3008C, 1 with the chloride ligand
was found to be the least stable with a Td at 2238C. Complex
7 has a Td at 2358C. An initial weight loss (T~5%) was mea-
sured to be 608C for complex 1 and 1148C for both complexes
4 and 7. These relative stabilities are in accordance with the
different kinds of bonding situations of the secondary ancillary
ligands to the AuIII center.
Structural characterization
X-ray diffraction studies were performed for complexes 1–6
with single crystals obtained by slow evaporation of the con-
centrated solution of the complexes in dichloromethane with
a layer of pentane at 0–58C. The perspective views of the mo-
lecular structures are shown in Figure 1. Table 1 displays select-
ed bond lengths and angles for all structures while the crystal-
lographic details are provided in the Supporting Information. A
distorted square planar geometry is observed resulting from
the coordination environment around the gold center. This be-
havior is expected for square-planar d8 systems with non-
tion from the idealized 908 angle
but only by about 58 with
Table 1. Selected bond lengths (ꢁ) and angles (8) for 1–6.
84.97(9)8. The phenyl ring of the
1
2
3a
3b
4
5
6
alkynyl ligand is almost coplanar
with the metallacycle and conse-
quently perpendicular to the
Fmes ligand with dihedral angles
of 13.1(2) and 74.10(9)8 between
the mean planes, respectively.
The intermolecular Au···Au dis-
tances of 6.1874(3) ꢁ for 3b and
6.2639(4) ꢁ for 4 were found to
be the shortest ones among all
AuꢁN
2.083(2)
2.019(2)
2.014(2)
2.3525(6)
6.5872(2)
81.49(9)
94.23(10)
88.98(7)
95.30(6)
2.089(2)
2.005(3)
2.017(3)
2.3452(8)
6.8605(2)
83.18(12)
93.59(13)
88.90(9)
94.34(8)
2.117(2)
2.004(3)
2.019(3)
2.3414(8)
7.4738(1)
80.89(10)
91.59(11)
93.13(8)
94.51(7)
2.103(4)
2.005(4)
2.015(4)
2.3384(12)
6.1874(3)
81.54(17)
94.55(18)
89.25(13)
94.63(11)
2.085(2)
2.038(3)
2.027(3)
2.045(3)
6.2639(4)
80.98(11)
93.08(11)
90.13(11)
95.77(10)
2.100(3)
2.037(3)
2.028(3)
2.6557(3)
7.7450(5)
80.95(14)
93.99(14)
88.12(10)
96.96(9)
2.086(3)
2.052(3)
2.023(3)
2.039(3)
7.1495(4)
80.80(12)
92.74(13)
91.93(12)
94.65(12)
AuꢁCN^C
AuꢁCFmes
AuꢁX[a]
Au···Au[b]
NꢁAuꢁCN^C
CN^CꢁAuꢁCFmes
CFmesꢁAuꢁX
XꢁAuꢁN
[a] X=Cl (1–3b), CCN (4), I (5) or CCCPh (6). [b] Shortest Au···Au distance in the crystal structure.
Chem. Eur. J. 2014, 20, 2585 – 2596
2588
ꢀ 2014 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Szentkuti, Alexander
Szentkuti, Alexander
 Bachmann, Michael
Bachmann, Michael
 Garg, Jai Anand
Garg, Jai Anand
 Blacque, Olivier
Blacque, Olivier
 VenKatosan, Koushik
VenKatosan, Koushik