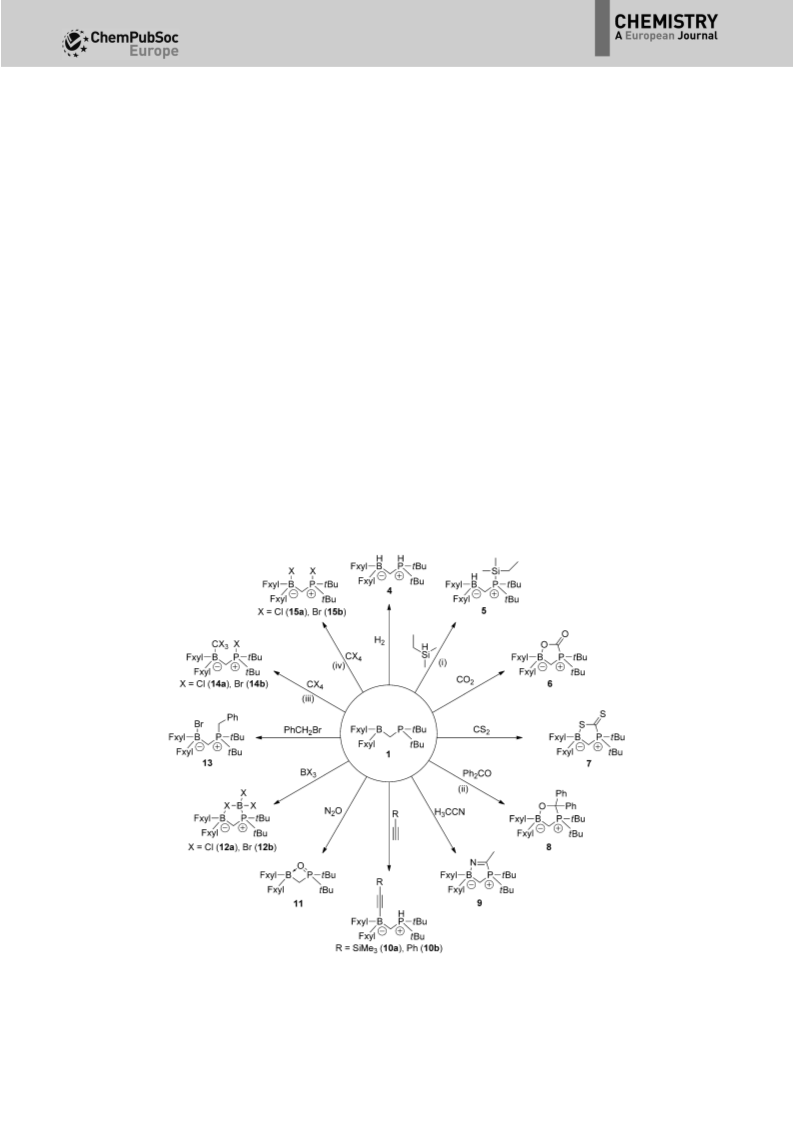
Full Paper
The reaction between 1 and CO , another standard FLP sub-
lecular structure of 9 in the solid state shows an endocyclic CÀ
2
strate, takes
a
similar course to the reaction between
N distance of 1.258(10) and an exocyclic CÀC distance of
[22]
[38]
tBu PCH BPh and CO2.
An almost-planar, five-membered,
air- and moisture-stable heterocycle with an exocyclic C=O
1.505(10) , which are typical values of C=N bonds
and
2
2
2
2
3
[39]
C(sp )ÀC(sp ) single bonds, respectively. We therefore con-
clude that 9 is the imine rather than the enamine tautomer. In
contrast to the adduct of Nçth et al., 9 is thermally stable up
to 1208C.
1
3
double bond is formed (6). The corresponding C NMR signal
appears at 168.3 ppm, in good agreement with the shift re-
ported for the literature-known system mentioned above
(
167.8 ppm). An analogous structure to 6 is obtained from
Reactions of P/B FLPs with terminal alkynes are governed by
the basicity of the phosphine: FLPs containing less basic phos-
phines tend to add to the CꢀC bond, whereas the use of
1
and CS (7). Compound 7 has a red-purple color, characteris-
2
[30–32]
tic of phosphine–CS adducts.
CS activation by P/B Lewis
2
2
pairs is far less common than CO2 activation, and the only
strongly basic phosphines (e.g., tBu P) results in deprotonation
3
[
40]
known examples are the addition of CS to tBu PNꢀBtmp
of the alkyne to give phosphonium alkynylborate salts. Ac-
2
2
[33]
(
Htmp=2,2,6,6-tetramethylpiperidine)
and
Et PC(Ph)= cordingly, 1 cleaves the terminal CÀH bonds of Me SiCCH and
2
3
[
34]
C(nBu)B(nBu2).
PhCCH with generation of 10a and 10b, respectively. Phos-
Whereas aldehydes have already been reported to react
phine protonation is evidenced by doublets of multiplets at
[
12,35,36]
31
1
with P/B FLPs,
the Ph CO adduct 8 is a rare example of
about 53 ppm in the P NMR spectra with J(P,H) coupling
2
1
an activated ketone. In a related case, Ph CO undergoes
constants of 450 Hz. The corresponding H resonances appear
2
1
a [2+2] cycloaddition with the phosphinoborane tBu PBFlu
at about 5 ppm as doublets of triplets ( J(H,P)=450 Hz,
2
3
11
(
HBFlu=9-borafluorene). The primary product then undergoes
heterolytic cleavage of the PÀB bond to furnish
tBu PCPh OBFlu. The room-temperature H NMR spectrum of
J(H,H)=4.5 Hz). B NMR signals are observed at À14.5 ppm.
As a further characteristic, the BCꢀC signals are broadened
[
16]
1
13
1
1
13
beyond detection in the C{ H} NMR spectrum. A H– C
HMBC experiment, however, revealed chemical shifts of
131.9 ppm (10a) and 109.8 ppm (10b). The proposed molecu-
lar structures of 10a and 10b were further corroborated by X-
ray crystallography (see the Supporting Information).
2
2
8
shows poorly resolved phenyl resonances. Steric repulsion
between the Ph and tBu substituents likely restricts intramolec-
ular motion and/or causes an association/dissociation equilibri-
um between FLP 1, the ketone, and 8. To clarify this point, we
also recorded NMR spectra of 8 at elevated temperatures. The
Stephan and co-workers trapped N O with a bimolecular P/B
2
3
1
[41]
P NMR signal (84.4 ppm) became severely broadened at 508C
FLP to obtain tBu PN=NOB(C F ) .
Although kinetically
3
6 5 3
11
and completely vanished at 808C; similarly, the B NMR reso-
nance of 8 (4.9 ppm) was no longer detectable in the high-
temperature spectrum. Both signals reappeared when the
sample was cooled back to room temperature. Moreover, the
colorless solution of 8 adopts the yellow color of free 1 on
heating, but becomes colorless again on cooling. Adduct for-
stable, the compound loses N with formation of the phos-
2
phine oxide adduct tBu P=OB(C F ) on photolysis or heating
3
6 5 3
to 1358C. In contrast, the intramolecular phosphine oxide
adduct 11 was already generated when an n-pentane solution
11
of 1 was stored under N O at 48C in the dark. The B NMR res-
2
onance of 11 appears at 7.5 ppm and thus in the typical shift
[
28]
mation of the FLP with Ph CO is thus a reversible dynamic pro-
range of tetracoordinate boron nuclei.
Compared to the
2
31
1
cess. Accordingly, compound 8 is hydrolyzed much more readi-
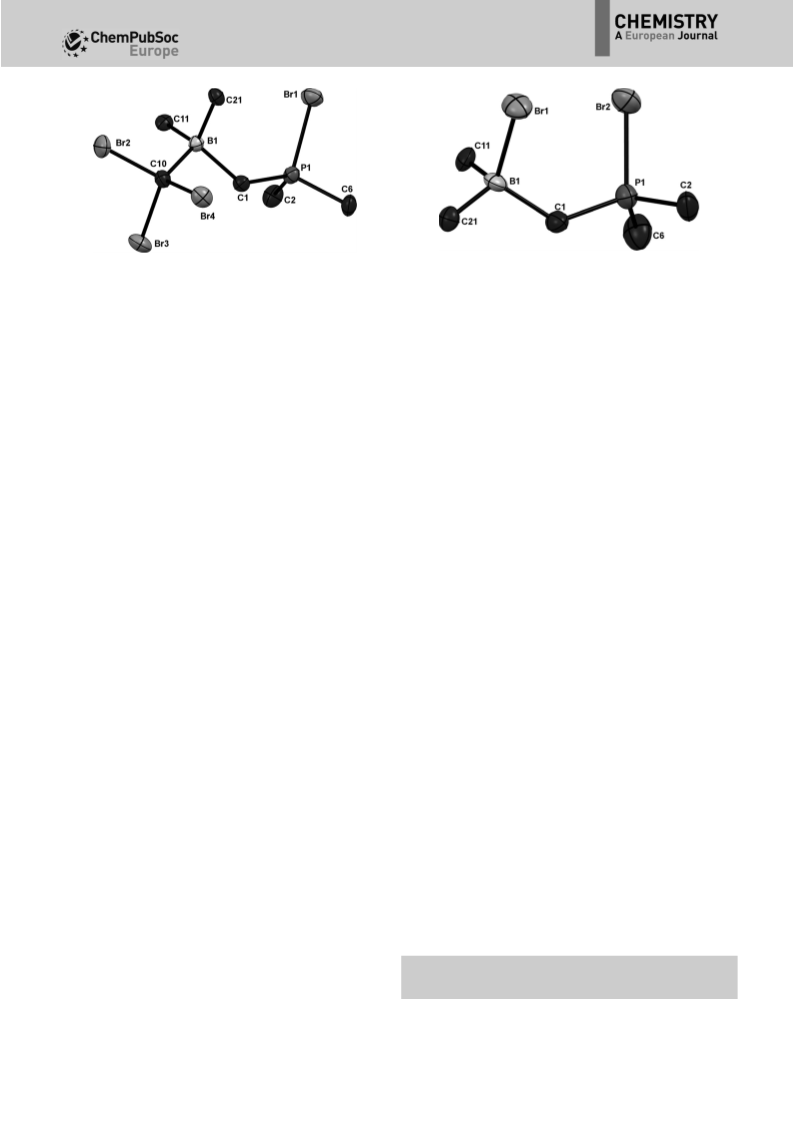
ly than compound 6. As a major hydrolysis product, we identi-
fied tBu P(H)CH B[OB(Fxyl) ](Fxyl) by X-ray crystallography and
P{ H} NMR resonance of 1 (25.9 ppm), the signal of 11 is shift-
ed to lower field (113.1 ppm). In the solid state, 11 has a P=O
bond length of 1.576(2) and a BÀO bond length of
1.612(3) . Both these bonds are significantly longer than
2
2
2
2
NMR spectroscopy (see the Supporting Information for more
details). This species is formally derived from (Fxyl) BOH by
those of the related intramolecular adduct tBu P(m-O)(m-
2
2
OÀH addition to 1.
C H )B(C F ) featuring a five-membered heterocycle (P=O
6
4
6 5 2
[
42]
Geminal FLP 1 efficiently catalyzes the hydrosilylation of
Ph CO with EtMe SiH (12 mol% catalyst loading, room temper-
1.546(2), BÀO 1.550(2) ).
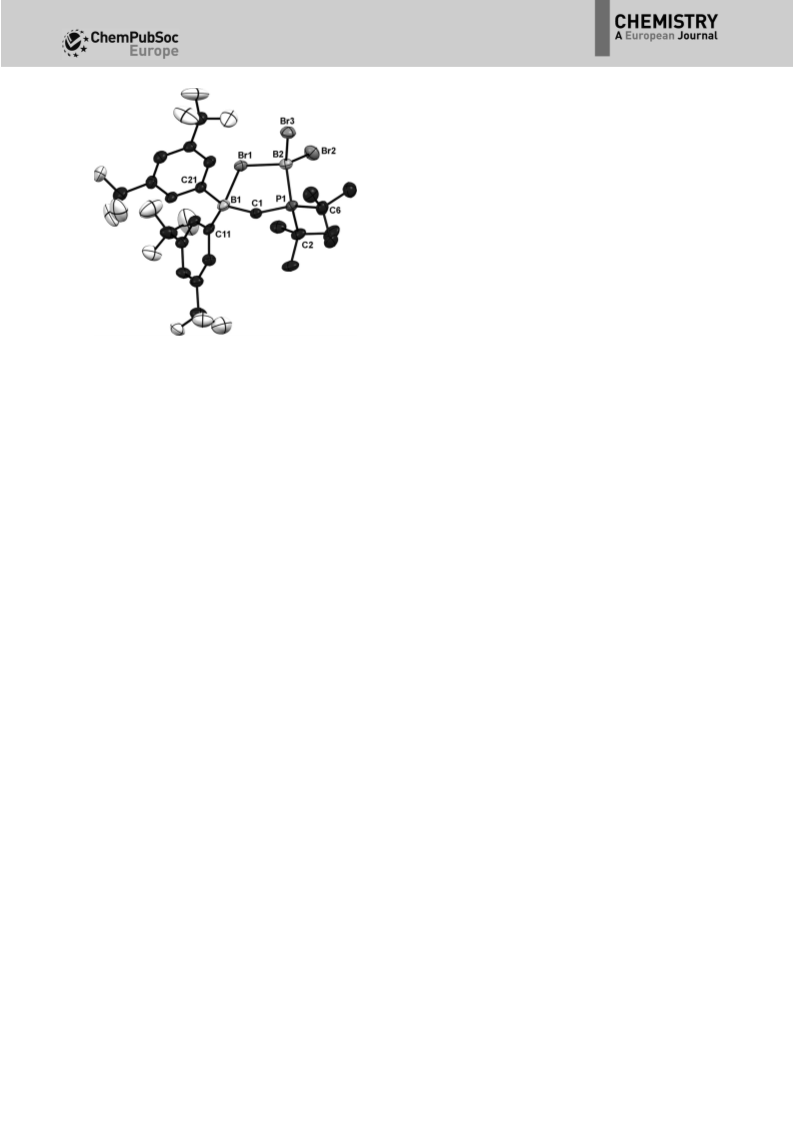
The serendipitous finding of the (Fxyl) BBr adduct 2 drew
2
2
2
[37]
ature, 30 min, C D ).
Note that 1 not only interacts with
our attention to the possibility of trapping BCl and BBr , too.
6
6
3
3
Ph CO, but also with EtMe SiH (cf. 5), the other reagent of the
Previously Uhl and co-workers prepared cyclic adducts be-
2
2
hydrosilylation sequence.
tween BX (X=F, Cl, Br, I) and the P/Al FLP Mes PC[=C(H)Ph]-
3
2
[
23a]
FLP 1 not only traps compounds containing a C=O bond,
AltBu2.
Interestingly, the products with X=F and Cl proved
but also adds to the CꢀN bond of H CCN to give the five-
to be thermally stable and could be stored at room tempera-
3
membered cyclic compound 9. The only comparable example
ture, whereas the adducts with X=Br and I decomposed
[
23a]
of a P/B-mediated H CCN activation was described by Nçth
above 08C.
In the case of FLP 1, both the BCl adduct 12a
3
3
and co-workers, who again used the species tBu PNꢀBtmp. At
and the BBr adduct 12b are isolable under ambient condi-
tions. We did not observe any signs of substituent scrambling
2
3
room temperature, they observed kinetically controlled forma-
tion of the imine fragment PC(CH )=NB. On thermal treatment,
between the two B atoms of 12a or 12b. BX binding results
in downfield shifts of the P NMR resonances from 25.9 ppm
3
3
31
the imine tautomerized to the thermodynamically preferred
[
33]
enamine PC(=CH )N(H)B. In the case of 9, we found a proton
in free 1 to 39.4 and 38.6 ppm in 12a and 12b, respectively
2
3
11
resonance at 1.88 ppm (d, J(H,P)=4.9 Hz) with an integral of
(broadened 1:1:1:1 quartets). In turn, the FLP B NMR signals
13
3
H, assignable to a CH group. The corresponding C NMR
experience an upfield shift from 63 ppm (1) to 35 ppm (12a)
or 34 ppm (12b), attributable to a certain degree of intramo-
3
2
signal was observed at 26.5 ppm (d, J(C,P)=47 Hz). The mo-
Chem. Eur. J. 2016, 22, 3478 – 3484
3481 ꢀ 2016 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Samigullin, Kamil
Samigullin, Kamil
 Georg, Isabelle
Georg, Isabelle
 Bolte, Michael
Bolte, Michael
 Lerner, Hans-Wolfram
Lerner, Hans-Wolfram
 Wagner, Matthias
Wagner, Matthias