
ChemCatChem p. 2382 - 2388 (2013)
Update date:2022-08-10
Topics:
 Zheng, Zhanfeng
Zheng, Zhanfeng
 Zhao, Jian
Zhao, Jian
 Liu, Hongwei
Liu, Hongwei
 Liu, Jiangwen
Liu, Jiangwen
 Bo, Arixin
Bo, Arixin
 Zhu, Huaiyong
Zhu, Huaiyong
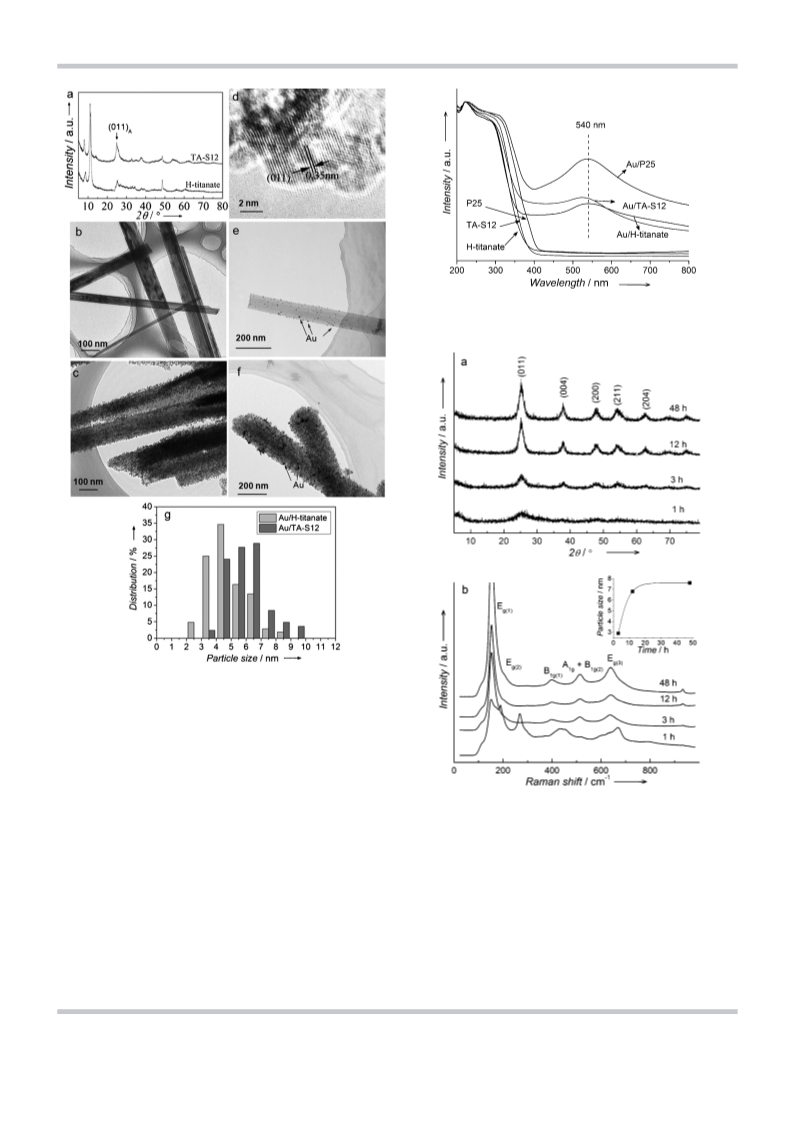
Anatase TiO2 nanocrystals were painted on H-titanate nanofibers by using an aqueous solution of titanyl sulfate. The anatase nanocrystals were bonded solidly onto the titanate fibers through formation of coherent interfaces at which the oxygen atoms were shared by the nanocrystals and the fiber. This approach allowed us to create large anatase surfaces on the nanofibers, which are active in photocatalytic reactions. This method was also applied successfully to coat anatase nanocrystals on surfaces of fly ash and layered clay. The painted nanofibers exhibited a much higher catalytic activity for the photocatalytic degradation of sulforhodamineB and the selective oxidation of benzylamine to the corresponding imine (with a product selectivity >99%) under UV irradiation than both the parent H-titanate nanofibers and a commercial TiO2 powder, P25. We found that gold nanoparticles supported on H-titanate nanofibers showed no catalytic activity for the reduction of nitrobenzene to azoxybenzene, whereas the gold nanoparticles supported on the painted nanofibers and P25 could efficiently reduce nitrobenzene to azoxybenzene as the sole product under visible light irradiation. These results were different from those from the reduction on the gold nanoparticles photocatalyst on ZrO2, in which the azoxybenzene was the intermediate and converted to azobenzene quickly. Evidently, the support materials significantly affect the product selectivity of the nitrobenzene reduction. Finally, the new photocatalysts could be easily dispersed into and separated from a liquid because of their fibril morphology, which is an important advantage for practical applications.
View More






LIAOYANG WANRONG CHEMICALS COMPANY LIMITED
Contact:86-419-2390789
Address:XINLI VILLAGE , DONG NINGWEI COUNTY,TAIZIHE DISTRICT, LIAOYANG , LIAONING
Guangxi Shanyun Biochemical Science and Technology Co., Ltd
Contact:+86-0772--6828887
Address:#2 Industrial Park of Luzhai County, Liuzhou, Guangxi, China
QINGDAO HONG JIN CHEMICAL CO.,LTD.
website:http://www.hongjinchem.com
Contact:+86-532-83657313
Address:2ND Building, 8 Shangqing Road, Qingdao, Shandong Province, China.
Jining tiansheng chemical co.,ltd.
Contact:+86-537-5158722
Address:ROOM 1011, BLOCK B, CUIDU INTERNATIOAL BUSINESS CENTER, JINING CITY, CHINA
Jinhua City Mingzhu Pharmaceutical Co.,Ltd.
Contact:15857995878 0579-82207761
Address:No.169 Shenze Road, New Area,Jinpan Development Zone, Jinhua
Doi:10.1002/jps.2600770217
(1988)Doi:10.1016/j.ejmech.2019.111976
(2020)Doi:10.1246/cl.2010.728
(2010)Doi:10.1023/A:1017536430239
(2000)Doi:10.1039/c8cp02352b
(2018)Doi:10.3762/bjoc.9.174
(2013)