Antimitotic agents bearing adamantane residue
Russ. Chem. Bull., Int. Ed., Vol. 70, No. 3, March, 2021
553
1
13
Compound 5a. Н NMR (CDCl ), : 1.66—1.77 (m, 12 H,
С NMR (CDCl ), : 28.3, 36.9, 39.2, 41.2 (C(1)Ad)); 66.2
3
3
HAd); 2.02 (m, 3 H, HAd); 2.33 (s, 2 H, AdCH ); 3.85 (s, 3 H,
(ArCH ); 101.5 (OCH O); 108.6 (C(2)); 109.9 (C(5)); 122.0
2
2 2
OCH ); 4.68 (s, 2 H, CH OH); 6.91 (dd, 1 H, C(5)H, J = 1.65 Hz,
(C(6)); 130.7 (C(1)); 147.7 (C(4)); 148.1 (C(3)); 177.8 (C=O).
3
2
+
+
J = 8.07 Hz); 6.99—7.01 (m, 1 H, C(6)H); 7.02 (d, 1 H, C(3)H,
MS (MALDI-TOF): found m/z 314 [M] , 337 [M + Na] , 353
J = 1.65 Hz). 1 С NMR (CDCl ), : 28.7, 33.1, 36.8, 42.3, 48.6
3
[M + K] ; C calculated for C19H22O 314.4.
+
3
4
(
(
AdCH ); 55.7 (OCH ); 65.1 (CH OH); 111.0 (C(3)); 118.9
(2E)-3-(3-Hydroxy-4-methoxyphenyl) adamantane-1-prop-
2-en-1-one (8). Sodium hydroxide (0.029 g, 0.728 mmol) was
added with stirring to a solution of 1-adamant-1-yl ethanone
(0.1 g, 0.56 mmol) in ethanol (5 mL). 3-Hydroxy-4-meth-
oxybenzaldehyde (0.085 g, 0.56 mmol) was added to the mixtu-
re, and the reaction mixture was stirred at ~20 C for 24 h. The
solvent was evaporated under reduced pressure. The residue was
chromatographed. Ethyl acetate—petroleum ether (40—70 С,
gradient 1 : 7—1 : 5) served as the eluent for chromatography.
2
3
2
C(5)); 122.9 (C(6)); 139.1 (C(4)); 139.7 (C(1)); 151.2 (C(2));
1
69.7 (C=O). Found (%): C, 72.65; H, 7.89. C20H26O . Calcul-
4
ated (%): C, 72.70; H, 7.93.
1
Compound 5b. Н NMR (CDCl ), : 1.62—1.63 (m, 9 H,
3
HAd); 1.67—1.73 (m, 8 H, HAd); 1.76—1.77 (m, 7 H, HAd); 1.97
(
(
6
m, 3 H, HAd); 2.02 (m, 3 H, HAd); 2.13 (s, 2 H, AdCH ); 2.33
2
s, 2 H, AdCH ); 3.83 (s, 3 H, OCH ); 5.08 (s, 2 H, CH O);
2 3 2
.94 (dd, 1 H, C(5)H, J = 1.7 Hz, J = 8.0 Hz); 6.98 (d, 1 H,
1
3
1
C(3)H, J = 1.7 Hz); 7.01 (d, 1 H, C(6)H, J = 8.0 Hz). С NMR
The yield was 0.088 g (50%), white crystals, m.p. 142 С. Н NMR
(
(
(
CDCl ), : 28.5, 28.6, 32.9, 33.1, 36.7, 36.8, 42.2, 42.4, 48.5
(CDCl ), : 1.72—1.81 (m, 6 H, HAd); 1.88 (d, 6 H, HAd,
3
3
AdCH ); 48.9 (AdCH ); 55.7 (OCH ); 65.4 (CH O); 112.4
J = 2.8 Hz); 2.09 (m, 3 H, HAd); 3.94 (s, 3 H, OCH ); 5.67
2
2
3
2
3
C(3)); 120.5 (C(6)); 122.9 (C(5)); 134.9 (C(4)); 139.5 (C(1));
51.1 (C(2)); 169.5 (C=O); 171.5 (C=O). MS (MALDI-TOF):
(m, 1 H, OH); 6.84 (d, 1 H, J = 8.2 Hz); 7.03 (d, 1 H, CH=CH—
C(O), J = 15.5 Hz); 7.06 (dd, 1 H, J = 2.0 Hz, J = 8.2 Hz); 7.23
(d, 1 H, J = 2.0 Hz); 7.59 (d, 1 H, CH=CH—C(O), J = 15.5
1
+
+
+
found m/z 506 [M] , 529 [M + Na] , 545 [M + K] ; calculated
1
3
for C32H42O 506.
Hz). С NMR (CDCl ), : 28.0, 36.6, 38.2, 45.4 (C(1)Ad); 56.0
5
3
4
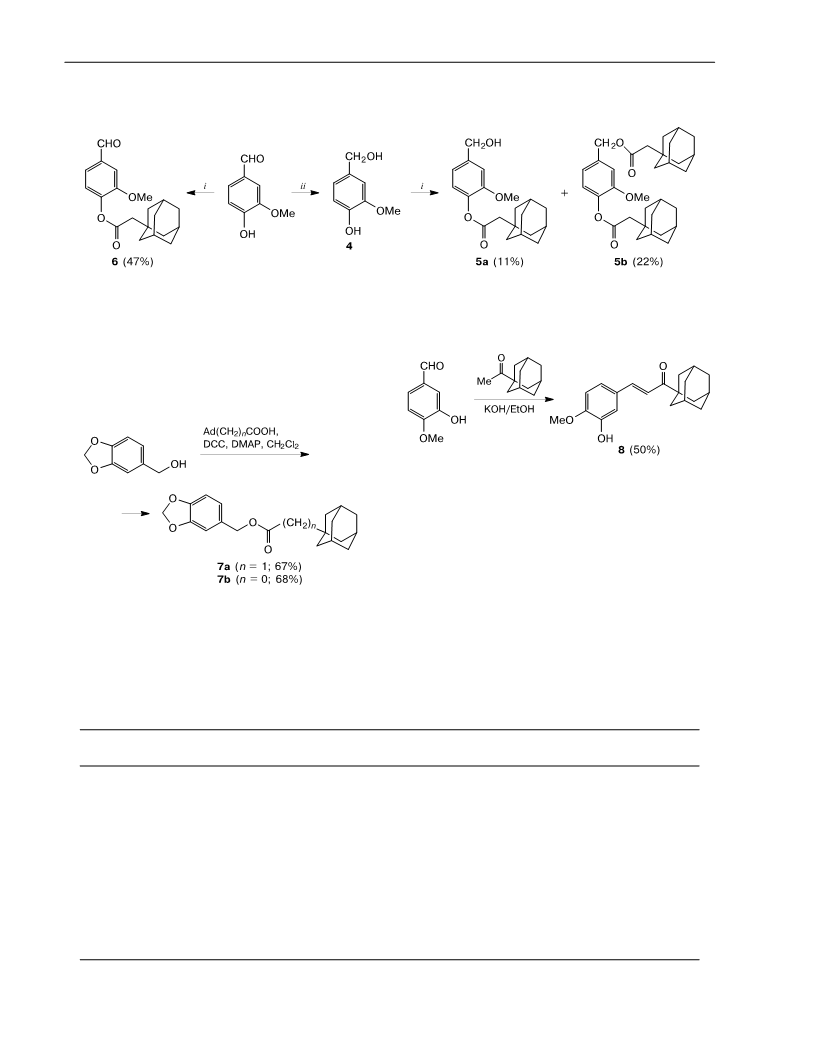
-Formyl-2-methoxyphenyl-adamantane-1-acetate (6) was
(OCH ); 110.4, 112.7 (OCH O); 118.6, 122.5 (CH=CHC(O));
3 2
synthesized according to the general procedure from 1-adaman-
taneacetic acid (0.128 g, 0.66 mmol) and 3-methoxy-4-hydroxy-
benzaldehyde (0.100 g, 0.66 mmol). Ethyl acetate—petroleum
ether (40—70 С, 1 : 7) served as the eluent for chromatography.
128.7, 142.6 (CH=CHC(O)); 145.7, 148.4, 203.9 (C=O).
Found (%): C, 76.82; H, 7.71. C20H24O . Calculated (%):
3
C, 76.89; H, 7.74.
Morphology of cells and microtubules was studied on lung
carcinoma cells A549 (CCL-185™) by immunofluorescence
microscopy. The cells were cultivated on small cover glasses,
incubated for 24 h together with the tested compounds in
1
The yield was 0.101 g (47%), white powder, m.p. 90 С. Н NMR
(
2
CDCl ), : 1.66—1.77 (m, 12 H, HAd); 2.02 (m, 3 H, HAd);
3
.35 (s, 2 H, AdCH ); 3.90 (s, 3 H, OCH ); 7.20 (d, 1 H, C(6)
2 3
–
1
H, J = 7.9 Hz); 7.47 (dd, 1 H, C(5)H, J = 1.8 Hz, J = 7.9 Hz);
a concentration of 10 or 100 mol L at 37 С and 5% CO2
(0.5% DMSO was used as the negative control).The fixed
cells were stained using primary mouse monoclonal antibodies
to -tubulin (Sigma, USA) followed by incubation with second-
ary goat antibodies against mouse immunoglobulins fluores-
cently labeled with AlexaFlour488 (Molecular Probes, USA).
The cells were analyzed with a Nikon Diaphot 300 microscope
(Nikon GmbH, Germany) equipped with a SenSys camera
(Photometrics, Germany). The procedure was described in
1
3
7
(
1
1
(
.50 (d, 1 H, C(3)H, J = 1.7 Hz); 9.94 (s, 1 H, C(O)H). С NMR
CDCl ), : 28.6, 33.1, 36.7, 42.2, 48.4 (AdCH ); 55.8 (OCH );
3
2
3
10.7 (C(3)); 123.5 (C(6)); 124.6 (C(5)); 135.0 (C(4));
45.0 (C(1)); 151.9 (C(2)); 168.9 (C=O), 191.0 (C(O)H). MS
+
+
MALDI-TOF): found m/z 351 [M + Na] , 367 [M + K] ;
calculated for C20H24O 328.4.
4
1
,3-Benzodioxol-5-ylmethyl adamantane-1-acetate (7a) was
synthesized using the general procedure from 1-adamantanea-
1
4—16
cetic acid (0.1 g, 0.51 mmol) and 1,3-benzodioxol-5-ylmethanol
detail.
(
0.078 g, 0.51 mmol). Ethyl acetate—petroleum ether (40—70 С,
This work was financially supported by the Russian
Science Foundation (Project No. 19-13-00084). The
synthesis and biotesting of compound 7а were performed
in terms of state assignment AAAA-A16-116032250004-2.
The authors are grateful to the German Academic Exchange
Service.
1
0
1
: 10) served as the eluent for chromatography. The yield was
1
.112 g (67%), pale yellow oily liquid. Н NMR (CDCl ), :
3
.58—1.68 (m, 12 H, HAd); 1.93 (s, 3 H, HAd); 2.07 (s, 2 H,
AdCH ); 4.97 (s, 2 H, ArCH ); 5.93 (s, 2 H, OCH O); 6.75
2
2
2
(
d, 1 H, C(5)H, J = 7.66 Hz); 6.82 (dd, 2 H, C(2)H, C(6)H,
1
3
J = 1.73 Hz, J = 8.81 Hz). С NMR (CDCl ), : 29.0, 33.2, 37.1,
3
4
1
(
2.8 (C(1)Ad)); 49.3 (AdCH ); 66.2 (ArCH ); 101.5 (OCH O);
2 2 2
This paper does not contain description of studies on
animals or humans.
08.6 (C(2)); 109.5 (C(5)); 122.6 (C(6)); 130.4 (C(1)); 147.9
C(4)); 148.1 (C(3)); 172.0 (C=O). MS (MALDI-TOF): found
+
+
+
The authors declare no competing interests.
m/z 328 [M] , 351 [M + Na] , 367 [M + K] ; calculated for
C20H24O 328.4.
4
1
,3-Benzodioxol-5-ylmethyl adamantane-1-carboxylate (7b)
References
was synthesized according to the general procedure from 1-ada-
mantaneacetic acid (0.1 g, 0.56 mmol) and 1,3-benzodioxol-5-
ylmethanol (0.085 g, 0.56 mmol). Ethyl acetate—petroleum ether
1. N. A. Zefirov, E. V. Nurieva, Yu. A. Pikulina, A. V. Ogon´kov,
B. Wobith, S. A. Kuznetsov, O. N. Zefirova, Russ. Chem.
Bull., 2017, 66, 1503.
2. N. A. Zefirov, Y. A. Evteeva, A. I. Krasnoperova, A. V.
Mamaeva, E. R. Milaeva, S. A. Kuznetsov, O. N. Zefirova,
Mendeleev Commun., 2020, 30, 421.
(
40—70 С, 1 : 10) served as the eluent for chromatography. The
1
yield was 0.120 g (68%), white solid, m.p. 74—75 С Н NMR
(
CDCl ), : 1.65—1.72 (m, 6 H, HAd); 1.88 (d, 6 H, HAd,
3
J = 2.89 Hz); 1.99 (s, 3 H, HAd); 4.97 (s, 2 H, ArCH ); 5.94
2
(
6
s, 2 H, OCH O); 6.76 (dd, 1 H, C(5)H, J = 0.58 Hz, J = 7.22 Hz);
3. L. Wanka, Kh. Iqbal, P. R. Schreiner, Chem. Rev., 2013,
113, 3516.
2
.79 (dd, 2 H, C(2)H, C(6)H, J = 1.59 Hz, J = 8.53 Hz).

 Zefirov
Zefirov
 Mamaeva
Mamaeva
 Krasnoperova
Krasnoperova
 Evteeva, Yu. A.
Evteeva, Yu. A.
 Milaeva
Milaeva
 Kuznetsov
Kuznetsov
 Zefirova
Zefirova