Green Chemistry
Page 8 of 9
chromatography using a Varian 450ꢀGC apparatus equipped with
a split/splitless injection mode and a flame ionization detector
CTQ2009ꢀ11376 (subꢀprogramme PPQ)). The contribution of
Leire LorenzoꢀIbarreta to the experimental work is also gratefully
acknowledged.
2
7
according to the procedure previously reported. Retention times
min): glycidol, 6,67; ethylene glycol (internal standard), 7,96;
(
5
glycerol, 12,59; GDC, 15,15; GC, 15,77.
Notes and references
Elemental analysis were carried out by the Servicios Técnicos
de Investigación of the University of Alicante (Alicante, Spain) in
a Thermo Finnigan 1112 Series Flash Elemental Analyzer
a
6
6
7
7
8
8
9
9
0
5
0
5
0
5
0
5
TECNALIA, TECNALIA-Energy, Department of Bioenergy, Parque
Tecnológico de Álava, Leonardo Da Vinci, 11, 01510 Miñano, Spain.
Fax:
+34
945198117;
Tel:
+34
629087981;
E-mail:
(ThermoFisher Scientific).
b
Universidad Alfonso X el Sabio, Department of Industrial Technology,
Avda de la Universidad 1. 28696 Villanueva de la Cañada, Madrid,
Spain. Fax: +34 91 8109781; Tel: +34 629087981; E-mail:
1
0
NMR spectra were carried out by Dr. Carmen Sanmartín of the
Pharmaceutical and Organic Chemistry Department of the
University of Navarra (Navarra, Spain) in a 400 MHz BRUKER
AC NMR spectrometer using tetramethylsilane as internal
reference and deuterated dimethyl sulfoxide as solvent.
1
F.M. Kerton, in Alternative Solvents for Green Chemistry; RSC Green
Chemistry Book Series, RSC Publishing, Cambridge, 2009, pp. 103ꢀ
1
5
Conclusions
1
04.
J. Kahre, T. Loehl, H. Tesmann, and H. Hensen, DE Pat., 19 756 454,
999.
JEFFSOL® GC, Glycerine Carbonate in Beauty & Personal Care,
Huntsman Technical Bulletin, 2011,
2
3
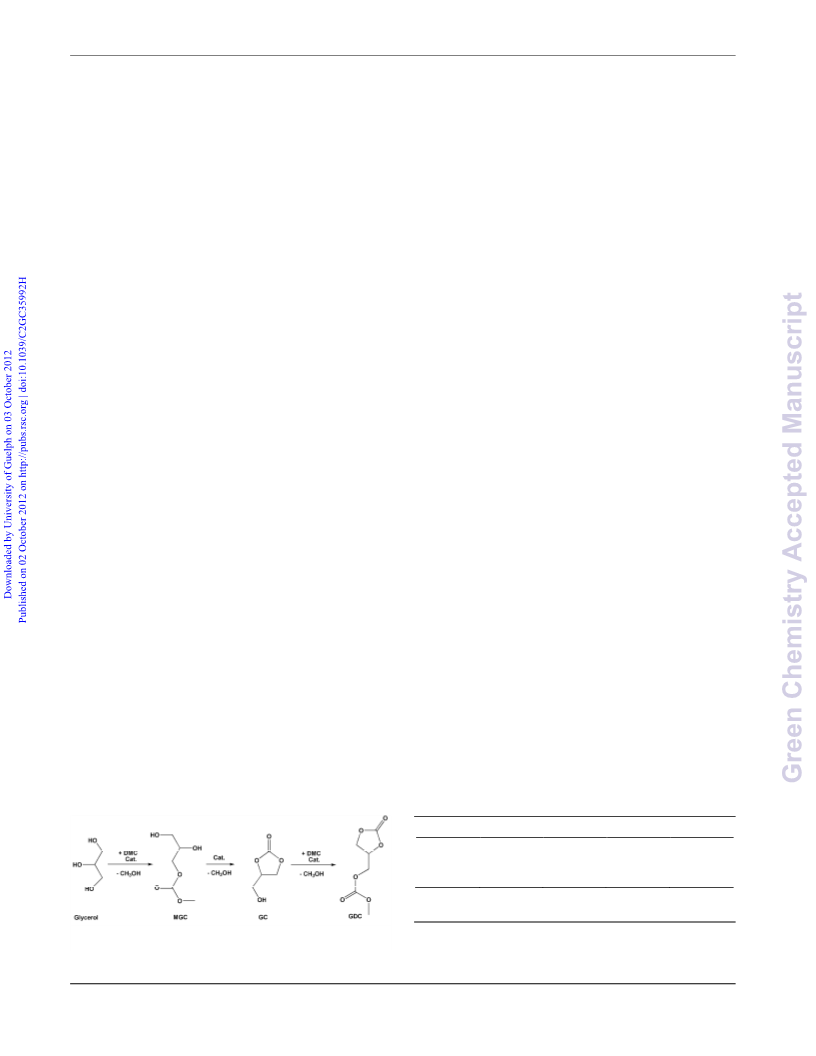
The synthesis of glycerol 1,2ꢀcarbonate (GC) by
transesterification of glycerol with dimethyl carbonate (DMC)
can be carried out by using triethylamine (TEA) as a facile
separable homogeneous catalyst. Reaction rate increases
dramatically with temperature. At a TEA/glycerol molar ratio of
1
2
2
3
3
4
4
5
0
5
0
5
0
5
0
4
5
D.P. Abraham, US Pat. Appl., 2011/0117445A1, 2011.
C. Magniont, G. Escadeillas, C. OmsꢀMulton and P. De Caro, Cem.
Concr. Res.. 2010, 40, 1072ꢀ1080.
0
.3 reaction is fast at refluxing temperature: a > 99% glycerol
conversion and a 91% GC yield are achieved in about 1 h with a
DMC/glycerol molar ratio of 4. However, at this high
TEA/glycerol molar ratio is difficult to stop the reaction in the
target molecule and glycerol dicarbonate (GDC) is also formed in
a 8% yield. GDC yield increases both with TEA/glycerol and
with DMC/glycerol molar ratios.
A good reaction control can be achieved by decreasing the
TEA/glycerol molar ratio but at the expense of a longer reaction
time. Thus, a 99% glycerol conversion and a 98% GC yield are
obtained in 2.5 h with a TEA/glycerol molar ratio of 0.1 and a
DMC/glycerol molar ratio of 4 at refluxing temperature. This low
TEA/glycerol molar ratio also prevents the evolution of GC to
GDC during GC isolation by evaporation under reduced pressure,
which in any case must be carried out at ≤ 40ºC.
6
7
A.T. Brooker, EP Pat., 2 380 958, 2011.
J.R. OchoaꢀGómez, O. GómezꢀJiménezꢀAberasturi, C. Ramírezꢀ
López and Mikel Belsué, Org. Process. Res. Dev., 2012, 16, 389ꢀ399.
8
9
1
1
1
M.J. Climent, A. Corma, P. De Frutos, S. Iborra, M. Noy, A. Velty
and P. Concepción, J. Catal., 2010, 269, 140ꢀ149.
Z. Mouloungui, J.W. Yoo, C. Gachen and A. Gaset, EP Pat., 0 739
888, 1996.
0 H.ꢀJ.Cho, H.ꢀM. Kwon, J. Tharun, and D.ꢀW. Park, J. Ind. Eng.
Chem., 2010, 16, 679ꢀ683.
1 G. Rokicki, P. Rakoczy, P. Parzuchowski, M. Sobiecki, Green Chem.,
2
005, 7, 529ꢀ539.
2 J.R. OchoaꢀGómez, O. GómezꢀJiménezꢀAberasturi, B. Maestroꢀ
Madurga, A. PesqueraꢀRodríguez, C. RamírezꢀLópez, L. Lorenzoꢀ
Ibarreta, J. TorrecillaꢀSoria and M.C. VillaránꢀVelasco, Appl. Catal.
A, 2009, 336, 315ꢀ324;
1
1
3 J. Li and T. Wang, J. Chem. Thermodyn, 2011, 43, 731ꢀ736.
4 F.S.H. Simanjuntak, T.K. Kim, S.D. Lee, B.S. Ahn, H.S. Kim and L.
Hyunjoo, Appl. Catal. A, 2011, 401, 220ꢀ225.
Above 90ºC glycidol is also obtained. Its synthesis can be
justified by means of a mechanism in which TEA abstracts a
proton from the hydroxyl moiety of GC resulting in the formation 100 15 J. Li and T. Wang, Reac. Kinet. Mech. Cat., 2011, 102, 113ꢀ126.
of the strong nucleophile 1,3ꢀdioxolanꢀ2ꢀoneꢀ4ꢀmethoxide, which
1
6 M. Du, Q. Li, W. Dong, T. Geng and Y. Jiang, Res. Chem. Intermed.,
012, 38, 1069ꢀ1077.
7 A. Takagaki, K. Iwatani, S. Nishimura and K. Ebitani, Green Chem.,
010, 12, 578ꢀ581.
2
evolves to glycidol with CO evolution through an intramolecular
2
1
nucleophilic attack of the negatively charged oxygen atom to the
methyne carbon in the ring.
GDC formation can be avoided working at conversions below
2
105 18 R. Bai, S. Wang, F. Mei, T. Li and G. J. Li, Ind. Eng. Chem. 2011, 17,
777ꢀ781.
1
2
9 M. Malyaadri, K. Jagadeeswaraiah, P.S. Sai Prasad and N. Lingaiah,
Appl. Catal. A, 2011, 401, 153ꢀ157.
0 (a) M. Tudorache, L. Protesescu, S. Coman and V. I. Parvulescu,
Green Chem., 2012, 14, 478ꢀ48219; (b) K.H. Lee, C.H. Park and E.Y.
Lee, Bioprocess Biosyst. Eng. 2010, 33, 1059ꢀ65; (c) S.C. Kim, Y.H.
Kim, H. Lee, D.Y. Yoon and B.K. Song, J. Mol. Catal. B: Enzym.
1
00% but in this case GC is highly impurified with glycerol after
solvent removal by evaporation. To separate both chemicals a
liquidꢀliquid extraction method has been developed using both
methyl isobutyl ketone (MIBK) and DMC as selective extraction
solvents for GC. GC can be extracted selectively at room
temperature in a 100% yield from a glycerol/GC mixture
consisting of 80 wt% GC by using a MIBK/mixture mass ratio of
1
1
1
10
15
2
007, 49, 75ꢀ78.
2
2
1 C. Chiappe and S. Rajamani, Pure Appl. Chem. ASAP article, 2012,
84, 755ꢀ762.
2 (a) M.G. Alvarez, A.M. Segarra, S. Contreras, J.E. Sueiras, F. Medina
and F. Figueras, Chem. Eng. J., 2010, 161, 340–345; (b) M.G.
Alvarez, M. Plíšková, A.M. Segarra, F. Medina and F. Figueras,
Appl. Catal. B, 2012, 113-114, 212ꢀ220.
1
.6 and three extraction steps. GC purity is higher than 98% as
shown by gas cromatography.
Acknowledgments
20 23 (a) L. Zhang, W. Guo, D. Liu, J. Yao, L. Ji, N. Xu and E. Min, Energy
Fuels, 2008, 22, 1353–1357; (b) L. Wang, Z. Tang, W, Xu and J.
Yang, Catal. Commun., 2007, 8, 1511ꢀ1515.
Thanks are due to the Ministerio de Economía y Competitividad
of the Spanish Government for financial support (project
5
5
8
| Journal Name, [year], [vol], 00–00
This journal is © The Royal Society of Chemistry [year]

 Ochoa-Gomez, Jose R.
Ochoa-Gomez, Jose R.
 Gomez-Jimenez-Aberasturi, Olga
Gomez-Jimenez-Aberasturi, Olga
 Ramirez-Lopez, Camilo
Ramirez-Lopez, Camilo
 Maestro-Madurga, Belen
Maestro-Madurga, Belen