558
A. Joseph et al. / Electrochimica Acta 137 (2014) 557–563
CPE has been used for lead sensing. These authors achieved selec-
concentration. In another study [19] a CPE was modified with a
range of 1.0 × 10−8 to 1.0 × 10−1 M. Studies have also been carried
out with other polymer modified CPEs for the detection of Pb2+ and
other metal ions [20,21]. Iminodiacetic acid (IDA), as a selective
metal coordinating ligand, has been used for remediation of lead
[22] as well as for other kinds of sensing applications [23].
2.3. Synthesis procedures
2.3.1. Synthesis of diethyliminodiacetate (1)
About 11 g of iminodiacetic acid (0.0826 mol) was dispersed
in 75 ml of absolute ethanol. To this 6 ml of concentrated sulfu-
ric acid was added dropwise over time and the reaction mixture
was refluxed for 24 h. Then the mixture was allowed to cool down
to room temperature and the excess acid was neutralized with 1 M
sodium hydroxide. The organic layer was collected by filtration and
the filtrate was diluted with 200 ml of dichloromethane. This was
then washed three times with 50 ml brine solution in each step.
The organic layer was evaporated under vacuum to get a yellowish
liquid. Yield: 9.04 g. NMR (DMSO-d6, 400 MHz): ␦ 4.19 (4H, q), 3.45
(4H, s), 2.13 (NH, s), 1.26 (6H, m). HRMS: 212 (M + Na). The NMR
spectrum is shown in Fig. S1 (Supporting Information).
In the present investigation, a CPE has been modified with imi-
nodiacetic acid functionalized polypyrrole (IDA-PPy) and evaluated
for selective recognition of Pb2+ ion from aqueous media, using
differential pulse anodic stripping voltammetry (DPASV) as the
measuring technique. The pyrrole was initially pre-functionalized
with iminodiacetic acid (IDA) and then chemically polymerized
into IDA-PPy. The advantage of using polypyrrole as the supporting
backbone is its inherent electron transport ability which improves
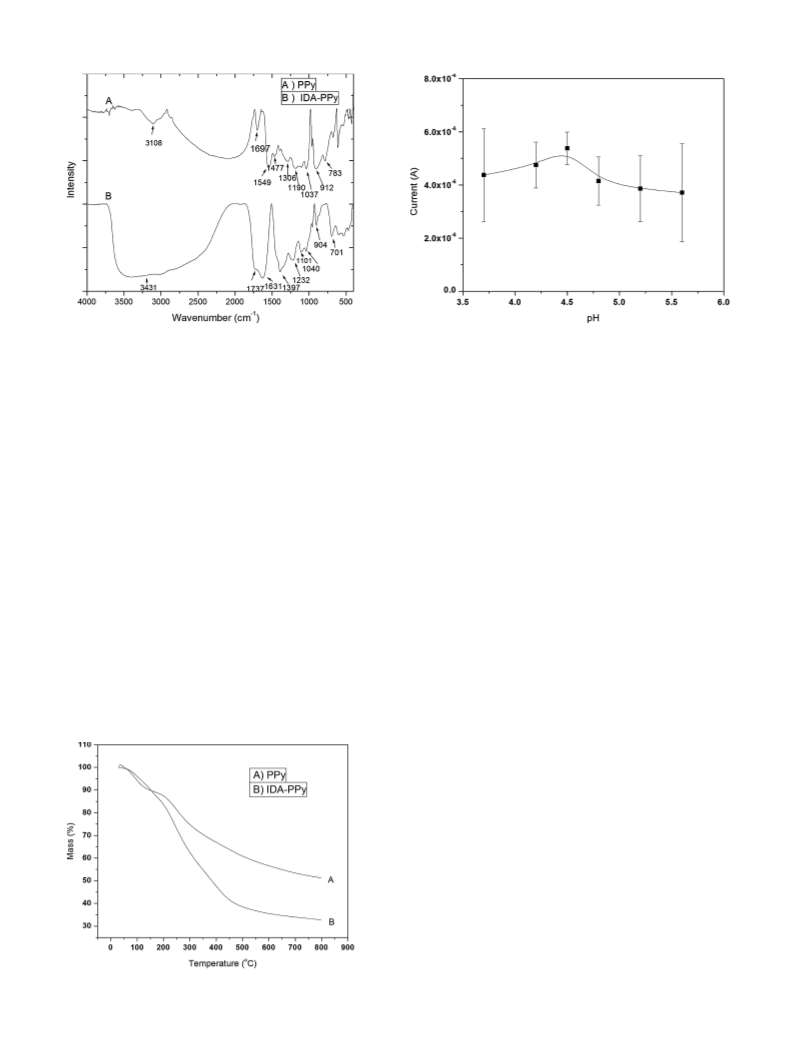
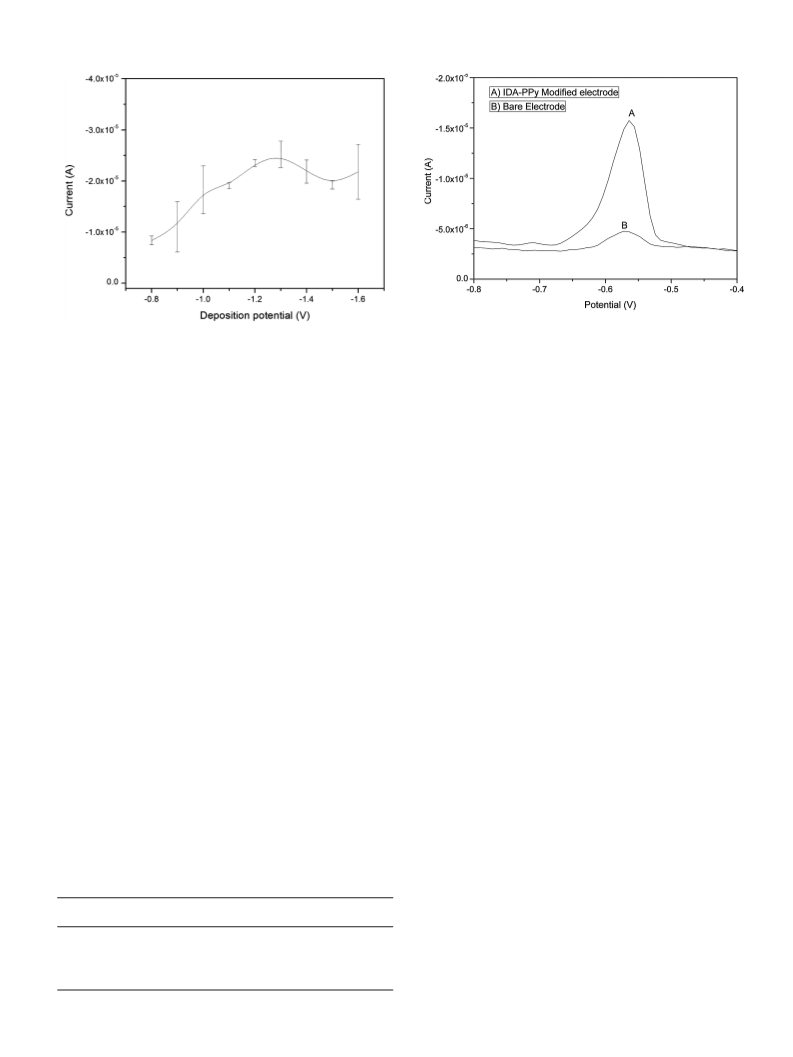
the detection range of metal ions. Various parameters affecting
the efficacy of IDA-PPy modified CPE, such as concentration of
depositing solution, pH of depositing solution, deposition poten-
tial, deposition time, and scan rate, have been optimized to achieve
maximum performance. The specificity of the IDA-PPy CPE for Pb2+
ions has been confirmed based on the comparison of the responses
2.3.2. Synthesis of N-glycidyl pyrrole (EPy) (2)
About 100 g of potassium hydroxide was dissolved in 150 ml
of D.I. water and cooled to 0 ◦C in an ice bath. To this 4.2 g of
tetrabutyl ammonium hydrogen sulfate was added followed by
dropwise addition of 45.1 ml of epichlorohydrin (0.58 mol). This
mixture was allowed to form a homogeneous solution by vigor-
ous stirring for 5 min. 20 ml of pyrrole (0.29 mol) was then added
dropwise over 20 min while keeping the temperature below 5 ◦C
whereafter stirring was continued for another 6 h at 0 ◦C. The reac-
tion mixture was poured into an excess of D.I. water and the organic
layer was extracted with diethylether, washed with three batches
of brine solution of 150 ml each, and dried over Na2SO4. The sol-
vent was evaporated under reduced pressure and the residue was
purified by vacuum distillation using a short-neck distillation appa-
ratus under high vacuum at 250 ◦C to get EPy. The distillate was
collected in portions and the last fraction yielded a colorless liq-
uid, which turned yellowish upon long exposure to atmosphere.
To prevent this, the last batch distillate was stored in a refrigera-
tor under dark conditions prior to use in subsequent experiments.
GCMS: 122.77 (retention time 10.15 s). NMR (CDCl3, 400 MHz): ␦
6.79 (1H, t), 6.08 (1H, t), 4.21 (1H, dd), 3.91 (1H, dd), 3.2 (1H, p), 2.78
(1H, q), 2.53 (1H, m). The NMR spectrum also showed the presence
of unreacted epichlorohydrin in small fractions. The NMR spectrum
and the GCMS results are shown in Fig. S2 (Supporting Information).
of individual solutions of different metal ions, namely Cu2+, Cd2+
,
Co2+, Hg2+, Ni2+ and Zn2+, evaluated at or below 1 × 10−7 M concen-
tration under identical conditions. Furthermore, the interference of
these metal ions for Pb2+ ion sensing has been investigated from
binary solutions of chosen concentrations. The practical efficacy of
the developed lead ion sensor has been assessed using an industrial
effluent sample.
2. Experimental
2.1. Chemicals, reagents and other materials
All the chemicals were analytical grade reagents and purchased
from local suppliers and used as such. These included imino-
diacetic acid (Merck Ltd., India), epichlorohydrin (Sisco Research
Laboratories Pvt. Ltd., India), tetrabutyl ammonium hydrogen sul-
fate, ethanol, and potassium hydroxide (s d Fine-chem Ltd., India).
Pyrrole (Spectrochem Pvt. Ltd., India) was dried over potassium
hydroxide and purified by distillation under vacuum prior to use.
High purity analytical grade acetic acid and sodium acetate (Sigma
Aldrich) were used for buffer solution preparation. Potassium
nitrate of analytical grade (s d Fine-chem Ltd., India) was used
as supporting electrolyte. Graphite powder of average grains size
100 m and light-weight paraffin oil (s d Fine-chem Ltd., India)
were used for making the electrodes. Deionized (D.I.) water (Milli-Q
system, > 15 Mꢀ cm) was used for all experiments, and all reactions
were carried out under inert argon atmosphere.
2.3.3. Synthesis of 1-N,N’-diacetylacetate-2-hydroxy-3N-pyrrolyl
propane (3)
About 8.2 g of EPy (colorless, 0.0667 mol) and 15.2 g of
diethyliminodiacetate (0.0805 mol) were mixed with 3 ml of abso-
lute ethanol and stirred for 24 h at 70 ◦C in an inert atmosphere. The
solution was purified by column chromatography (100–200 silica
gel, 25% ethyl acetate/petroleum ether) to get a yellowish viscous
liquid. Yield: 3.82 g. GCMS: 311.87 (retention time 22.76 s). NMR
(CDCl3, 400 MHz): ␦ 6.8 (2H, s), 5.9 (2H, s), 4.83 (1H, s), 4.1 (4H, q),
3.79 (2H, d), 3.55 (4H, s), 2.6 (2H, s), 1.2 (6H, t). The NMR and GCMS
results are shown in Fig. S3 (Supporting Information).
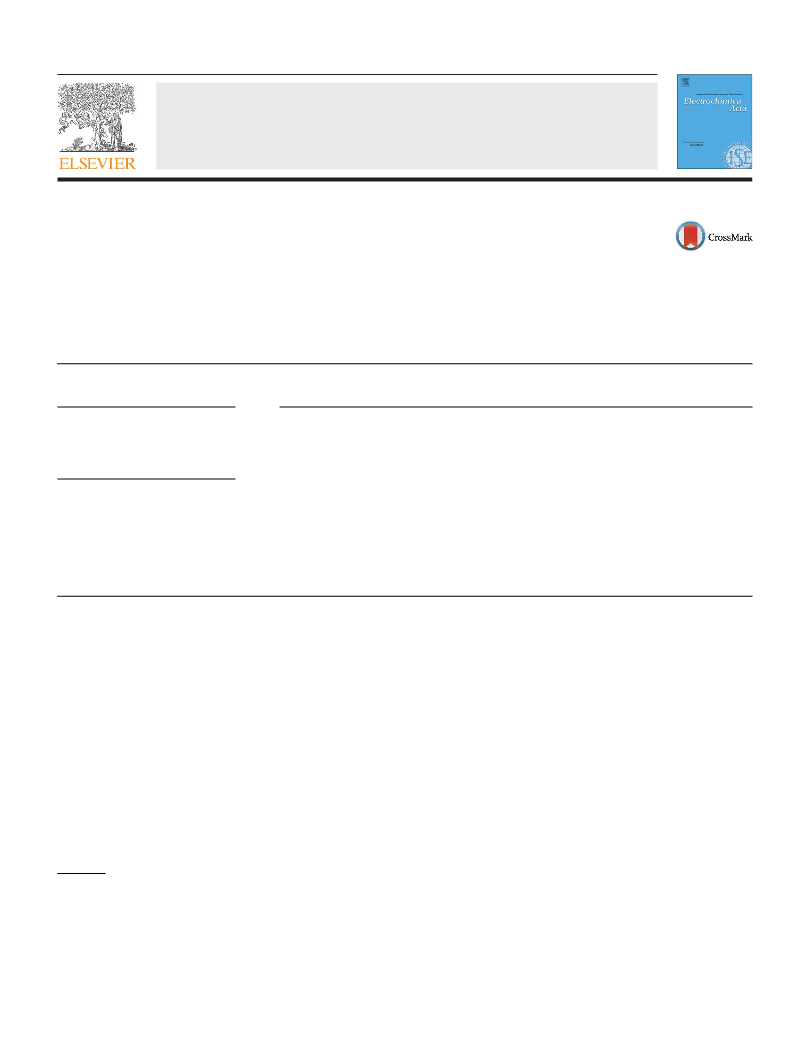
2.2. Characterization techniques
All the synthesized products were characterized after each step
using NMR and GCMS techniques. NMR spectra were recorded
using a Bruker 400 MHz instrument and GCMS data using a Thermo
Trace GC Ultra instrument. Fourier transform infrared (FTIR) spec-
tra were recorded in a Thermo-Nicolet 6700 instrument using the
KBr pellet technique. Thermogravimetric analysis was carried out
under ultrapure argon atmosphere using a Netzsch STA 409 instru-
ment at a heating rate of 10 ◦C min−1. The crucible was preheated
and the base line was corrected before the analysis of each sam-
ple. Atomic absorption spectrophotometric analysis of the chosen
metal ion samples was carried out using a Thermo Electron Corpo-
ration Series AA spectrometer adopting standard procedures.
2.3.4. Polymerization of
1-N,N’-diacetylacetate-2-hydroxy-3N-pyrrolyl propane
5.15 g of the synthesized compound (3) (0.0193 mol) was mixed
with a 1:1 mixture of conc. HCl and D.I. water and refluxed for 12 h.
5.74 g of ammonium persulfate (0.0251 mol) in 20 ml of D.I. water
was added dropwise and stirred for 24 h. The solution turned dark
brown in color. The pH was adjusted to 2.5 by the slow addition
of dilute NaOH solution to initiate the precipitation process. The
solution was then kept in a refrigerator overnight to precipitate out
the polymer, which was later washed with D.I. water and decanted
many times until the washings became colorless. The black residue

 Joseph, Alex
Joseph, Alex
 Subramanian, Sankaran
Subramanian, Sankaran
 Ramamurthy, Praveen C.
Ramamurthy, Praveen C.
 Sampath, Srinivasan
Sampath, Srinivasan
 Kumar, R. Vasant
Kumar, R. Vasant
 Schwandt, Carsten
Schwandt, Carsten