and, in general, H-ZSM-5 is inactive for CO þ H
2
reactions in
the absence of H S.
2
Therefore, for the maximum selectivity to methanethiol, a
relatively non-functionalised surface is required, which has
2 3
minimal acidity. Clearly, a-Al O provides such a surface,
but it is interesting to note that high methanethiol selectivities
were also observed with SiO , BN and SiC. At the present
3
time, the reactivity of a-Al O is perhaps too low and it will
2
2
be necessary to produce a higher area formulation; this may
be achieved through comminution, which has proved success-
3
2
ful in other catalysed systems.
It is interesting to consider the possible mechanisms by
which methanethiol is formed on the surface of a-Al from
the reaction of CO þ H þ H S. In a number of previous stu-
COS has been proposed as an intermediate in the
conversion of CO and H S mixtures. Although no H was
2 3
O
2
2
1
8,20,25
dies
2
2
introduced with the reactants, H2 could be formed via the
formation of COS or via water gas shift reactions under the
experimental conditions:
Scheme 1 Modified mechanism of the Fischer–Tropsch process
incorporating a reaction of a C1 intermediate with a surface SH
group (* denotes surface intermediates).
ꢁ
CO þ H2S Ð COS þ H2
COS þ 3H2 Ð CH3SH þ H2O
CO þ H2O Ð CO2 þ H2
ð1Þ
ð2Þ
ð3Þ
ð4Þ
ð5Þ
ꢁ
CO to form either an adsorbed CH3S
intermediate or
ads
adsorbed CH
surface –SH groups would disrupt the chain propagation step
and lead exclusively to the formation of a C product, namely
CH SH, which is wholly consistent with Schulz–Flory poly-
merisation kinetics. This mechanism would also readily
explain the observation that when H S is present the products
3
SH. The high nucleophility of the sulfur in the
COS þ H2O Ð CO2 þ H2S
1
2
COS Ð CO2 þ CS2
3
2
3
The synthesis of CH SH via hydrogenation of COS is con-
sidered to involve a surface methylthiolate intermediate
2
2
.
5
are almost exclusively thiols or sulfur-containing molecules.
In conclusion, in this study of methanethiol formation from
CH
to the formation of by-product methane. In our studies, we
have purposely introduced additional H and, hence, this path-
way could be possible. However, with the higher concentra-
tions of H present, the formation of COS via reaction (1)
would be suppressed and the CH ads intermediate would be
expected to be hydrogenated more readily, leading to higher
selectivities of CH . In this way, it is not considered feasible
that this mechanism would lead to the, almost, exclusive
formation of CH SH, which is observed in our studies.
It is, of course, possible that methanethiol is formed by the
reaction of H S with methanol:
S
3 ads
Further hydrogenation of this intermediate leads
the reaction of a CO/H
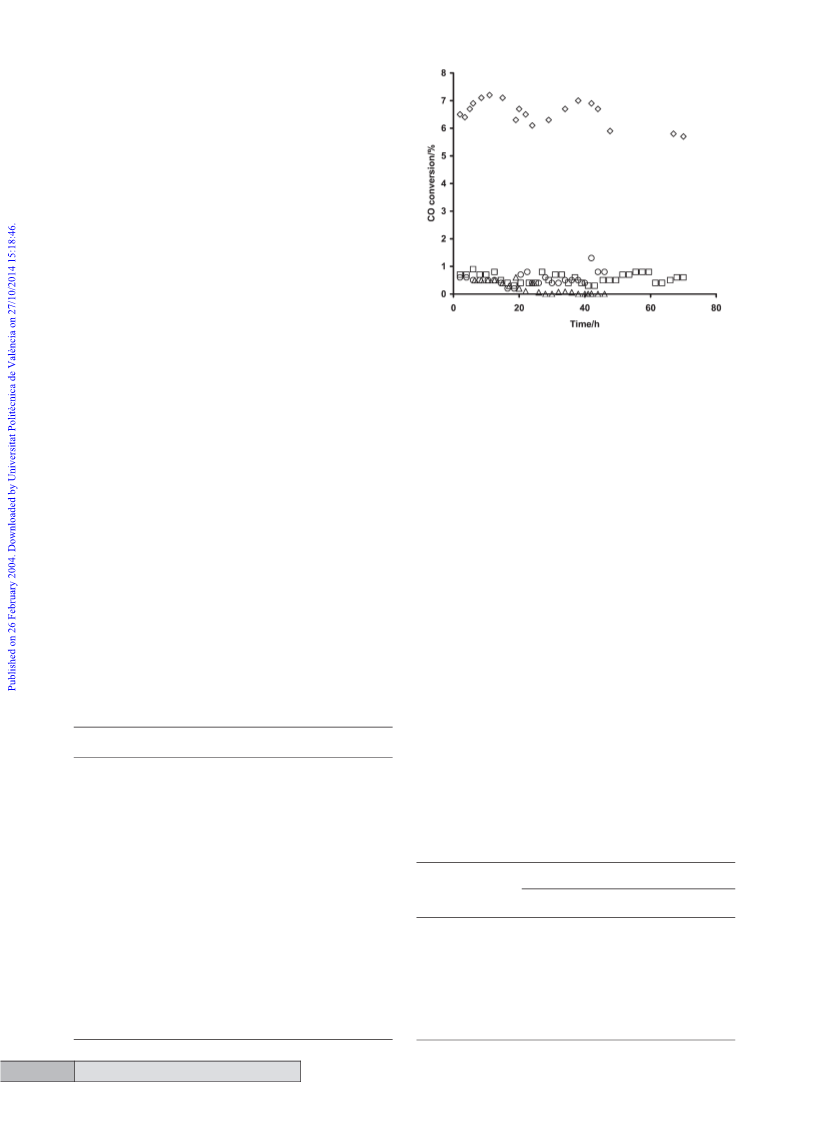
very high selectivities can be observed (ꢄ98%) when using
the relatively simple a-Al as catalyst. It is essential that sur-
2 2
/H S mixture, we have found that
2
2 3
O
2
face acidity is minimised, since when acid sites are present
hydrocarbon formation via the consecutive reaction of CH SH
is observed. A small amount of by-product thiophene is
formed along with the methanethiol, and if the activity of
the a-Al O catalyst can be significantly improved, then this
2 3
reaction may be of interest for the syntheses of these valuable
chemical intermediates.
3
S
3
4
3
2
CO þ 2H2 Ð CH3OH
CH3OH þ H2S Ð CH3SH þ H2O
ð6Þ
ð7Þ
Acknowledgements
The precedence for such a proposal comes from the present
industrial route for the synthesis of methanethiol via reaction
We thank the EPSRC for financial support.
1
1
7) and the observation that furan can exchange sulfur with
(
sulfided alumina catalysts as part of the mechanism by which
9
References
thiophene is formed. However, in our studies with g-Al
2 3
O
in the absence of H S, the rate of methanol formation is low
and the CO conversion is considerably enhanced when H S
2
2
1
2
3
4
5
M. V. Twigg, Catalyst Handbook, Wolfe Publishing Ltd., Frome,
UK, 1989.
J. P. Hindermann, G. J. Hutchings and A. Kiennemann, Catal.
Rev. Sci. Eng., 1993, 35, 1.
Y. Q. Yang, S. J. Dai, Y. Z. Yan, R. C. Lin, D. L. Tang and
H. B. Zhang, Appl. Catal., A, 2000, 192, 175.
L. S. Fuller, in Kirk-Othmer Encyclopaedia of Chemical Techno-
logy, 4th edn., Wiley, London, 1997, vol. 24, p. 34.
R. K. Russell and J. B. Press, Theophines and their Benzo Deriva-
tives, 1st edn., Elsevier, Amsterdam, 1996, vol. 2, p. 679.
R. T. Clark and J. A. Elkins, Jr., US Pat. 5,283,369, 1994.
J. Barrault, M. Guisnet, J. Lucien and R. Maurel, US Pat.
is introduced. Also, at low temperatures and low CO conver-
sion (Table 2) no methanol is observed, but if this was the pri-
mary product from the reaction of CO with H2 then some
residual methanol could have been expected to be observed
ꢀ
at 200 C. Both of these observations suggest that, for a-
2 3
Al O , reaction (7) is not the main pathway by which methane-
thiol is formed.
An alternative possibility is that a modification of the pro-
cess occurring during the Fischer–Tropsch reaction takes
place. The Fischer–Tropsch process is considered to follow a
6
7
4
,143,052, 6th March 1979.
8
9
0
1
2
N. R. Clark and W. E. Webster, UK Pat. 1,345,203, 10th May
1972.
surface polymerisation mechanism in which a surface C inter-
1
mediate reacts to form a surface C
2
intermediate as outlined in
S is introduced into the reactant mixture
the surface of the a-Al catalyst can be expected to comprise
SH. These would be much more nucleophilic with respect to
carbon compared with the surface –OH groups present in the
absence of H S. It is possible that the –SH groups could inter-
cept a C surface intermediate formed in the hydrogenation of
B. W. L. Southward, L. S. Fuller, G. J. Hutchings, R. W. Joyner
and R. A. Stewart, Chem. Commun., 1998, 2541.
B. W. L. Southward, L. S. Fuller, G. J. Hutchings, R. W. Joyner
and R. A. Stewart, Chem. Lett., 1998, 55, 207.
A. V. Mashkina, V. M. Kudenkov, E. A. Paukshtis and V. Y.
Mashkin, Kinet. Katal., 1992, 33, 904.
2
Scheme 1. When H
2
1
1
1
2 3
O
–
2
H. C. Woo, Y. G. Kim, I. S. Nam, J. S. Chung and J. S. Lee,
Catal. Lett., 1993, 20, 221.
1
T h i s j o u r n a l i s Q T h e R o y a l S o c i e t y o f C h e m i s t r y a n d t h e
C e n t r e N a t i o n a l d e l a R e c h e r c h e S c i e n t i f i q u e 2 0 0 4
N e w . J . C h e m . , 2 0 0 4 , 2 8 , 4 7 1 – 4 7 6
475

 Zhang, Baojian
Zhang, Baojian
 Taylor, Stuart H.
Taylor, Stuart H.
 Hutchings, Graham J.
Hutchings, Graham J.