
Journal of the Chinese Chemical Society p. 1270 - 1277 (2020)
Update date:2022-08-11
Topics:
 Perez, Ser John Lynon P.
Perez, Ser John Lynon P.
 Atayde, Eduardo C.
Atayde, Eduardo C.
 Arco, Susan D.
Arco, Susan D.
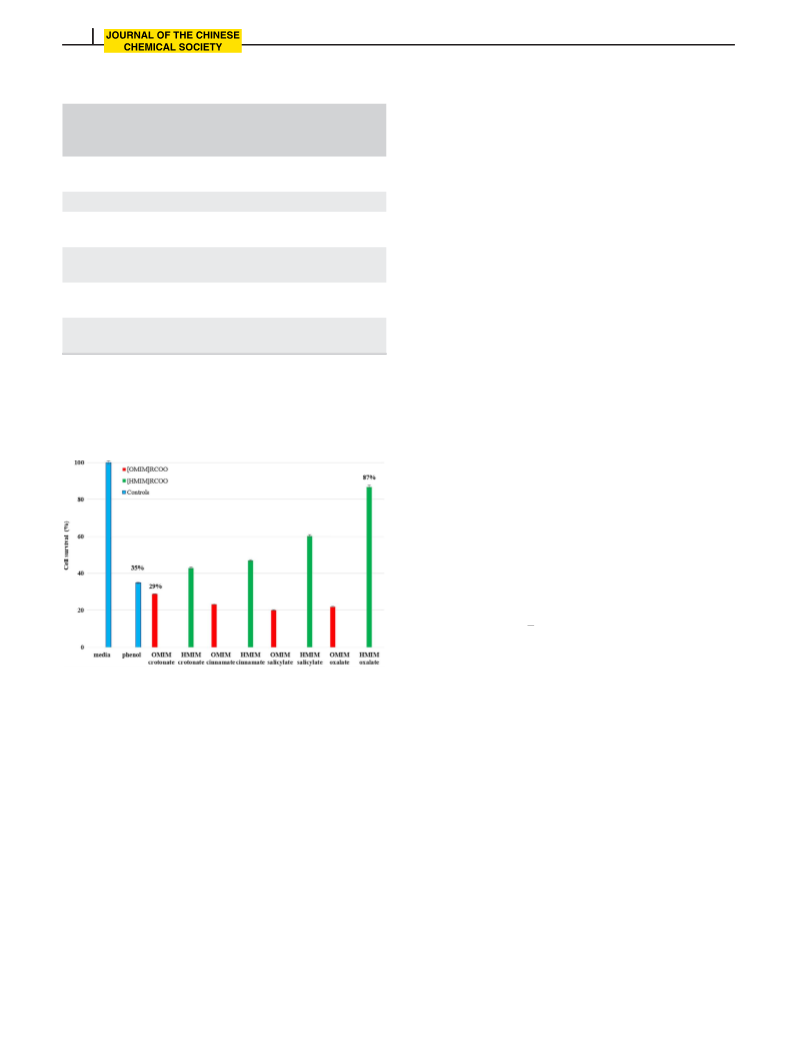
In an effort to discover lead compounds that may be of importance as potent antifungal agents, a series of novel 1-alkyl-3-methylimidazolium carboxylate ionic liquids were efficiently synthesized through a solvent-free ultrasound-assisted quaternization reaction of 1-methylimidazole and alkyl bromides RBr (R = hexyl, octyl) followed by an anion exchange process with selected carboxylate anions (cinnamate, salicylate, crotonate, and oxalate). Quantitative yields obtained were in the range of 86–94%. Structure characterization was done using FT-IR, 1H-NMR, and 13C-NMR spectroscopic techniques. All the synthesized compounds showed in vitro antifungal activity against the fungus Candida albicans with the minimum inhibitory concentrations found to be less than or equal to 1%. Preliminary cytotoxicity assays (trypan blue exclusion and MTT) were performed on all ionic liquids and findings revealed higher lymphocyte viability in 1-hexyl-3-methylimidazolium carboxylate ionic liquids than in 1-octyl-3-methylimidazolium counterparts. No extensive toxicity effect was observed with the carboxylate anion variation. Among the tested compounds, 1-hexyl-3-methylimidazolium crotonate and 1-hexyl-3-methylimidazolium oxalate exhibited the lowest cytotoxicity in the trypan blue exclusion and MTT assays, respectively. Together, our results highlight the potential of carboxylate-based ionic liquids in the development of next-generation antifungal drugs.
View More






BrightGene Bio-Medical Technology Co., Ltd.
website:https://en.bright-gene.com/
Contact:+86-512-62551801
Address:Building C25 - C31, No. 218 Xinghu Road, Suzhou Industrial Park, Suzhou, Jiangsu, China.
Contact:+86-25-83719363
Address:106-7 Chunnan Rd, Chunxi Town, Gaochun, Nanjing, China
ANHUI CHEM-BRIGHT BIOENGINEERING CO.,LTD
Contact:86-561-4080321
Address:No.8 Lieshan Industrial Zone of Huaibei
Contact:0833-5590788/5590338/5590055
Address:Victory in the town of Red Star Village,Mount Emei City,industrial concentration area storage processing logistics parkpark
Contact:027-87677569
Address:Room 2203, yujingmingmen Buidling One, xiongchu Road, wuhan city, hubei province, China
Doi:10.1080/00397910802604224
(2009)Doi:10.1016/j.ejmech.2014.01.015
(2014)Doi:10.1016/j.molcata.2013.03.004
(2013)Doi:10.1002/anie.201807814
(2018)Doi:10.1055/s-0032-1318148
(2013)Doi:10.1016/S0040-4020(01)86322-5
(1993)