Full Paper
tures in Scheme 2); the shape is typical of the emission of the
fused carbazole core of the SIA derivatives. The small Stokes
shifts are consistent with the high rigidity of the present D-
spiro-A derivatives. In addition, the emission QYs (in solution
relative to quinine sulfate) have been calculated to be between
33 and 39% (SIA-F: 33%, SIA-DAF: 38% and SIA-TXO2: 39%).
These relatively high and similar QYs clearly confirm that the
emissions are almost integrally due to transitions within the IA
unit (see the TD-DFT discussion above). Indeed, in such D-
spiro-A compounds, very low QYs are usually observed when
the HOMO and LUMO are spatially separated, leading to for-
bidden (or very disfavoured) through-space optical transi-
tions.[16b] Thus, the corresponding PA dyes, SPA-DAF and SPA-
TXO2, possess an unstructured emission spectrum with a corre-
sponding very low QY (0.1 and 4.1%, respectively), which is
characteristic of photoinduced intramolecular charge transfer
(ICT).[16b] Thus, the rigidification of the donor unit in D-spiro-A
compounds significantly increase the QY of the resulting dyes.
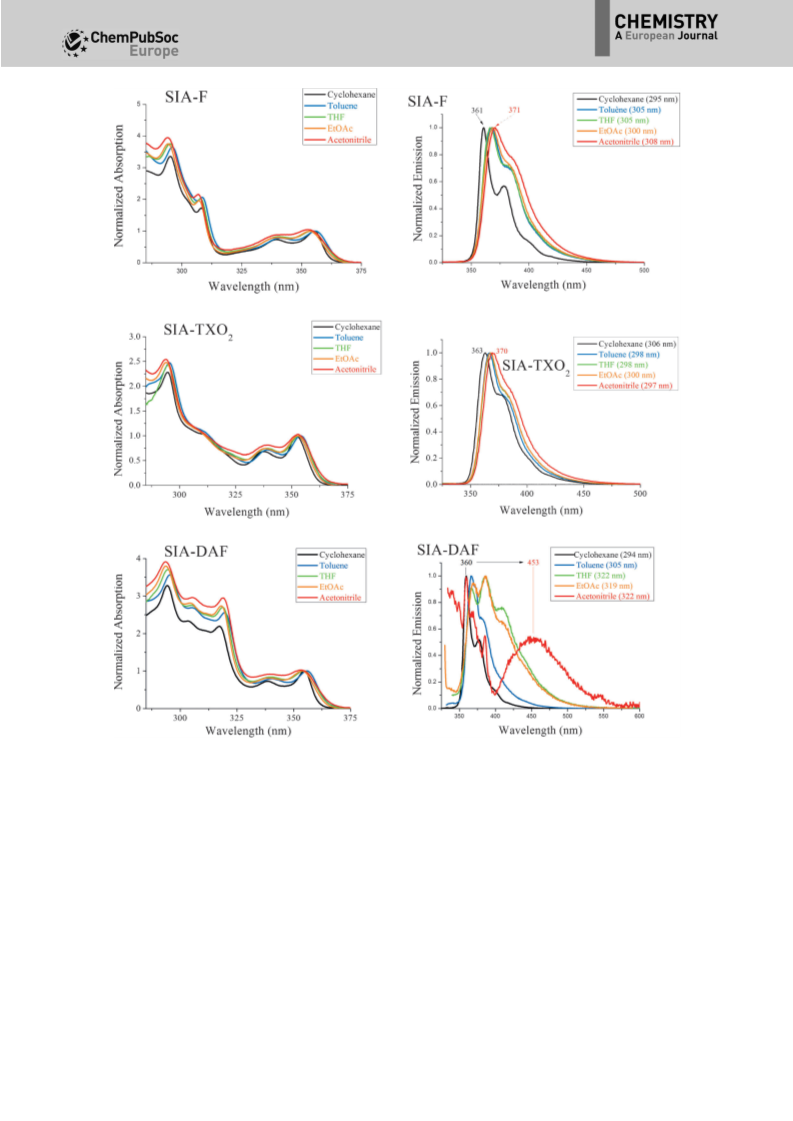
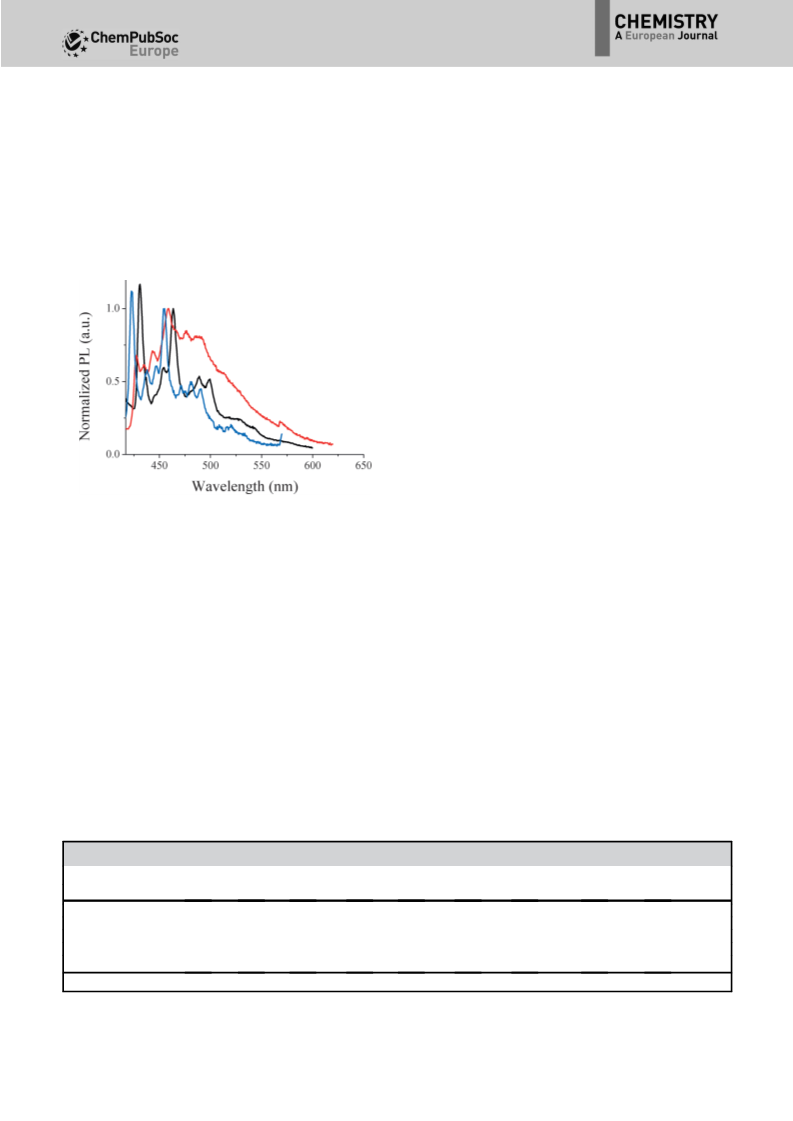
In the solid state, the absorption spectra (Figure 5, bottom
left) remain similar to those recorded in solution, with only
a redshift of 5 nm. Such similarity between absorption spectra
in solution and in the solid state clearly indicates that there are
very weak intermolecular interactions in the ground state in
thin films. Regarding the solid-state fluorescent properties
(Figure 5, bottom right) for SIA-F and SIA-TXO2, only a small
redshift is observed between solution and thin-film emissions
(10 nm for SIA-F and 6 nm for SIA-TXO2); this again indicates
weak interactions in the excited state. The solid-state fluores-
cent spectrum of SIA-DAF nevertheless appears poorly re-
solved, with a very weak emission at l=364 nm and a broad
ill-defined band at lꢀ418 nm.
the weak solvatochromic effect observed in emission; m(S1) is
therefore estimated to be 9.3 D.
The case of SIA-TXO2 is very similar to that of SIA-F, due to
the weak acceptor strength of the TXO2 fragment, with a shift
of the absorption bands of less than 2 nm and a shift of the
emission bands of less than 10 nm; however, there is a de-
crease in the emission QYs, which is halved upon going from
that in a non-polar solvent (40% in cyclohexane) to a more
polar one (20% in acetonitrile; see Table S6 in the Supporting
Information). The dipole moment of SIA-TXO2 in the ground
state, m(S0)=5.7 D, obtained through DFT calculations is much
higher than that of SIA-F (m(S0)=0.6 D). For SIA-TXO2, a small
Dm of 6.23 D is therefore calculated through Lippert–Mataga
formalism, and m(S1) is therefore estimated to be 11.9 D.
Hence, there is only a very weak solvatochromic effect, which
translates into weak photoinduced ICT within SIA-TXO2. This
feature confirms the significant overlap between the orbitals
involved in the transitions responsible for these emission pro-
cesses (HOMO/LUMO+1 transition only involving the IA frag-
ment).
As observed for the other molecules, the absorption spectra
of SIA-DAF are not dependent on the solvent polarity and
follow exactly the same trend as those exposed above for SIA-
TXO2 and SIA-F (cyclohexane: lmax =354.5 nm and acetonitrile:
lmax =353 nm). The dipole moment at the ground state ob-
tained through DFT calculations (m(S0)=4.1 D) is intermediate
between those of SIA-F and SIA-TXO2. However, the emission
spectra of SIA-DAF display peculiar behaviour. Indeed, from cy-
clohexane to ethyl acetate, only a very weak redshift of 10 nm
is detected (cyclohexane: lmax =360 nm and ethyl acetate:
lmax =370 nm). This feature is also indicative of weak photoin-
duced ICT in these solvents, in accordance with transitions be-
tween molecular fragments only involving the IA unit. This is
consistent with the conclusions drawn from TD-DFT results
and absorption spectra. However, in acetonitrile, a dual emis-
sion is observed. Indeed, the first structured emission is clearly
observed at l=360 nm, which is at exactly the same wave-
length as that observed in cyclohexane. In addition, a new and
very broad emission band between l=400 and 550 nm (cen-
tred at l=453 nm) is also recorded in acetonitrile. The QYs
also drop from 38% in cyclohexane to 5% in toluene, 1% in
THF, 1.5% in ethyl acetate and 1% in acetonitrile (see Table S6
in the Supporting Information). Hence, we believe that the first
emission corresponds to a locally excited state of the SIA unit
and the second broad band to a photoinduced ICT excited
state from the two fragments: IA (HOMO) to DAF (LUMO).
For SIA-DAF, the Lippert–Mataga calculations were per-
formed separately for these two excited states. In addition,
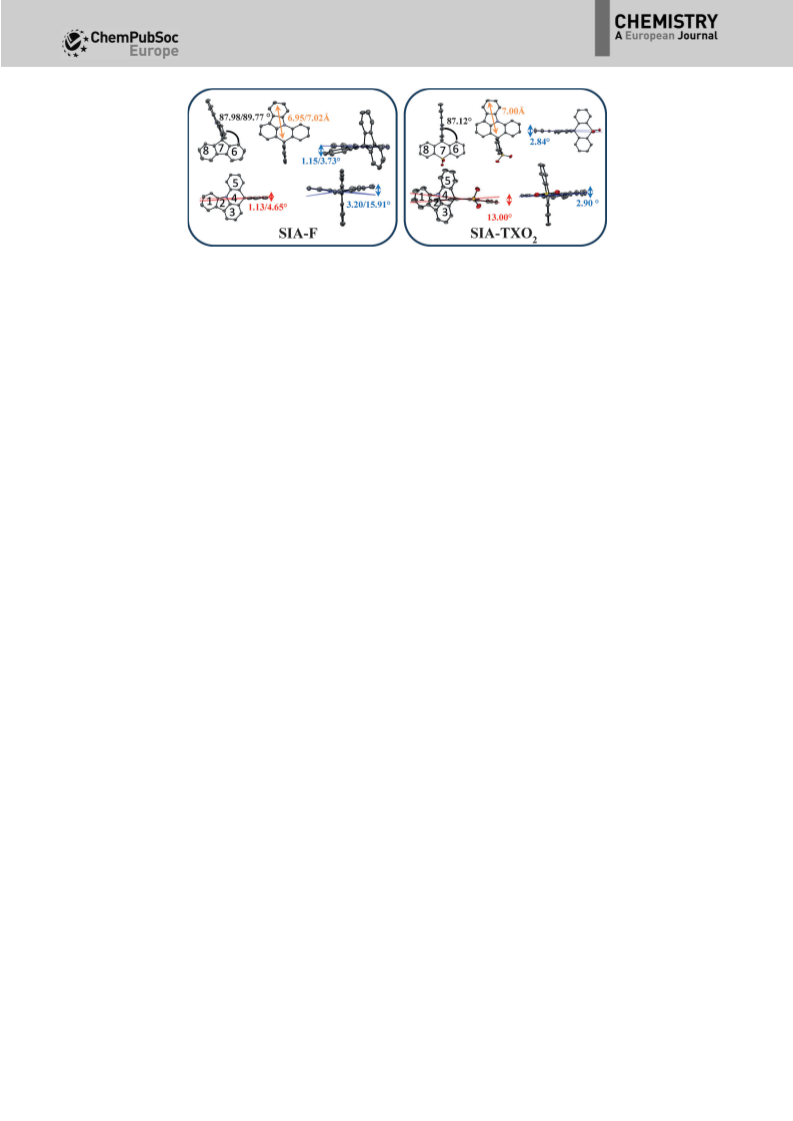
and in the absence of the crystallographic structure of SIA-
DAF, the radius of this molecule is expected to be 7.0 ꢃ, similar
to that of SIA-F and SIA-TXO2. Because the radius of these
molecules is directed by the SIA core (Figure 1), this estimation
is expected to be correct. For the locally excited state, the
dipole moment at the excited state, m(S1)LE, is 9.08 D, which is
similar to that calculated for SIA-F (9.26 D) and shows that the
acceptor core has no influence on the emissive properties of
this locally excited state (SIA unit). The ICT excited state,
Studying the absorption and emission spectra of bipolar
compounds in different polarity solvents is a key point to
assess the intensity of charge transfer and the polarity of the
excited states (Figure 7). Thus, we first note that the absorp-
tion maxima of SIA-F are almost insensitive to the dielectric
constant of the environment, with only a slight blueshift of the
maximum from cyclohexane (lmax =355 nm) to acetonitrile
(lmax =352.5 nm). This very weak effect is due to a slight gap
extension caused by stabilisation of the ground state in aceto-
nitrile. As classically observed, the emission spectra are more
influenced by the polarity of the solvent, although this influ-
ence remains modest herein. Indeed, we note that the emis-
sion of SIA-F is redshifted by only 10 nm from cyclohexane
(lmax =361 nm) to acetonitrile (lmax =371 nm). In addition, the
QYs are not affected by the solvent polarity (33% in cyclohex-
ane, 32% in acetonitrile, see Table S6 in the Supporting Infor-
mation). These data indicate that SIA-F displays a very weak
photoinduced ICT due to the significant mixing of both HOMO
and LUMO levels (Figure 6). Using Lippert–Mataga formalism
(see the Supporting Information for details; the radius of the
molecule has been estimated from crystallography results),
one can evaluate the dipole moment difference, Dm, between
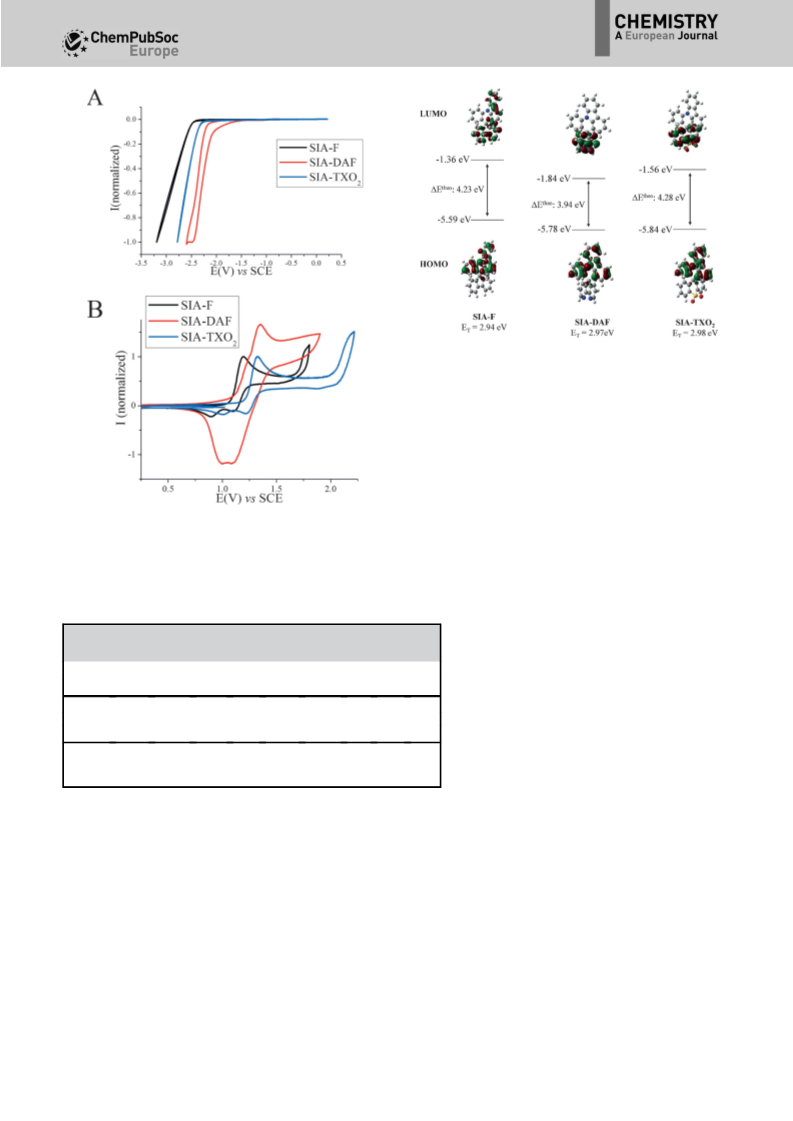
the ground and first excited state. For SIA-F (dipole moment
at the ground state obtained from DFT calculations: m(S0)=
0.6 D), a small Dm of 8.66 D is calculated in accordance with
Chem. Eur. J. 2016, 22, 1 – 15
9
ꢂ 2016 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
&
&
These are not the final page numbers! ÞÞ

 Thiery, Sébastien
Thiery, Sébastien
 Tondelier, Denis
Tondelier, Denis
 Geffroy, Bernard
Geffroy, Bernard
 Jeannin, Olivier
Jeannin, Olivier
 Rault-Berthelot, Jo?lle
Rault-Berthelot, Jo?lle
 Poriel, Cyril
Poriel, Cyril